Wearable Injector Market Size and Forecast – 2026 – 2033
The Wearable Injector Market size is estimated to be valued at USD 1.99 billion in 2026 and is expected to reach USD 4.85 billion by 2033, exhibiting a compound annual growth rate (CAGR) of 13.6% from 2026 to 2033.
Global Wearable Injector Market Overview
Drugs are mostly viscous and has various formulations (intravenous, intramuscular, and subcutaneous) that needs to be absorbed into digestive tract, which raise multiple drug delivery issue with traditional drug delivery system. Moreover, such issues not only raise costs but also decrease satisfaction and compliance of patients. Wearable injectors or bolus injectors are the new class for drug delivery device designed for subcutaneous delivery. It has capacity to hold the drug volume of up to 2.5 ml.
Wearable injectors are easy to handle, low cost, disposable, and allow self-administration. Advancement in injector technology over traditional drug delivery system include single button to release the drug and after completion of drug in body, the needle is retracted and gets locked automatically. Thus, it’s safe and convenient disposable system benefits people who has needle phobia. Furthermore, it allows audible and visual signals, which is expected to fuel the growth of wearable injector market during forecast period.
Key Takeaways
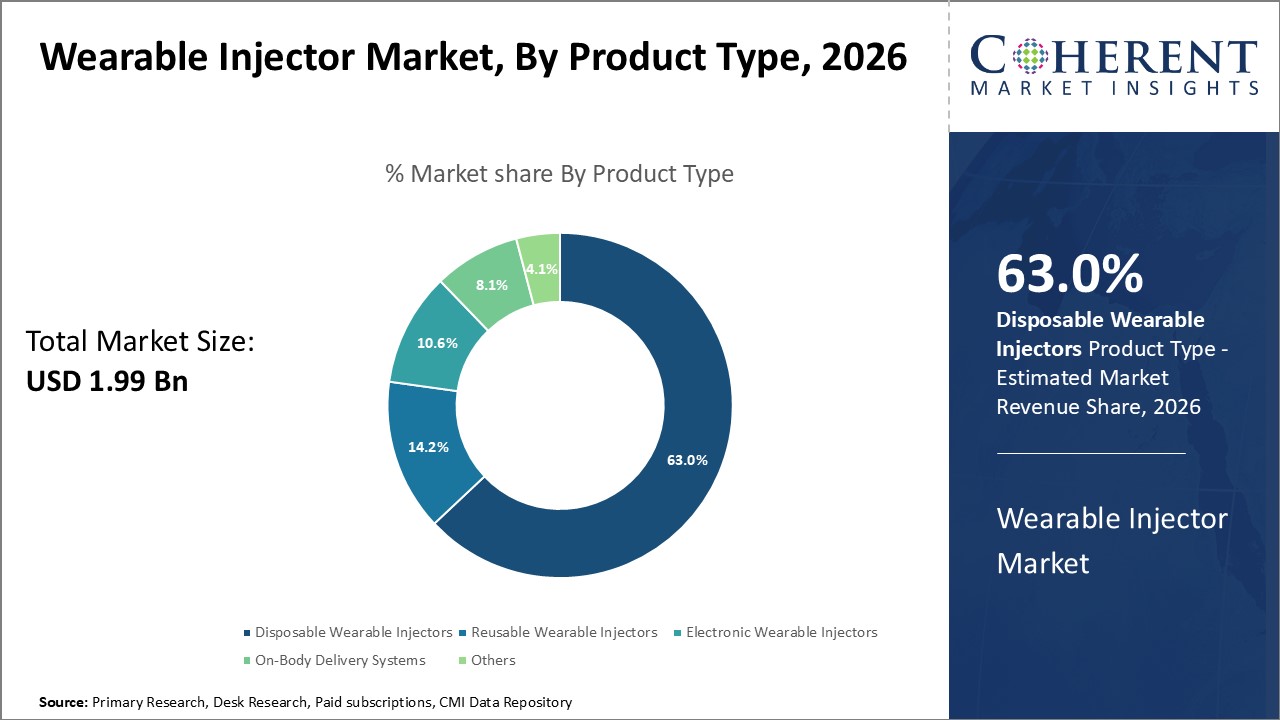
The Disposable Wearable Injectors segment dominates with a market share of 63%, driven by ease of use and cost efficiency. Autoimmune disorder applications showcase robust growth opportunities based on rising prevalence and therapeutic complexity.
Hospitals and clinics remain the leading end users, accounting for over 56% market revenue share, whereas home healthcare is emerging rapidly owing to increasing chronic disease management.
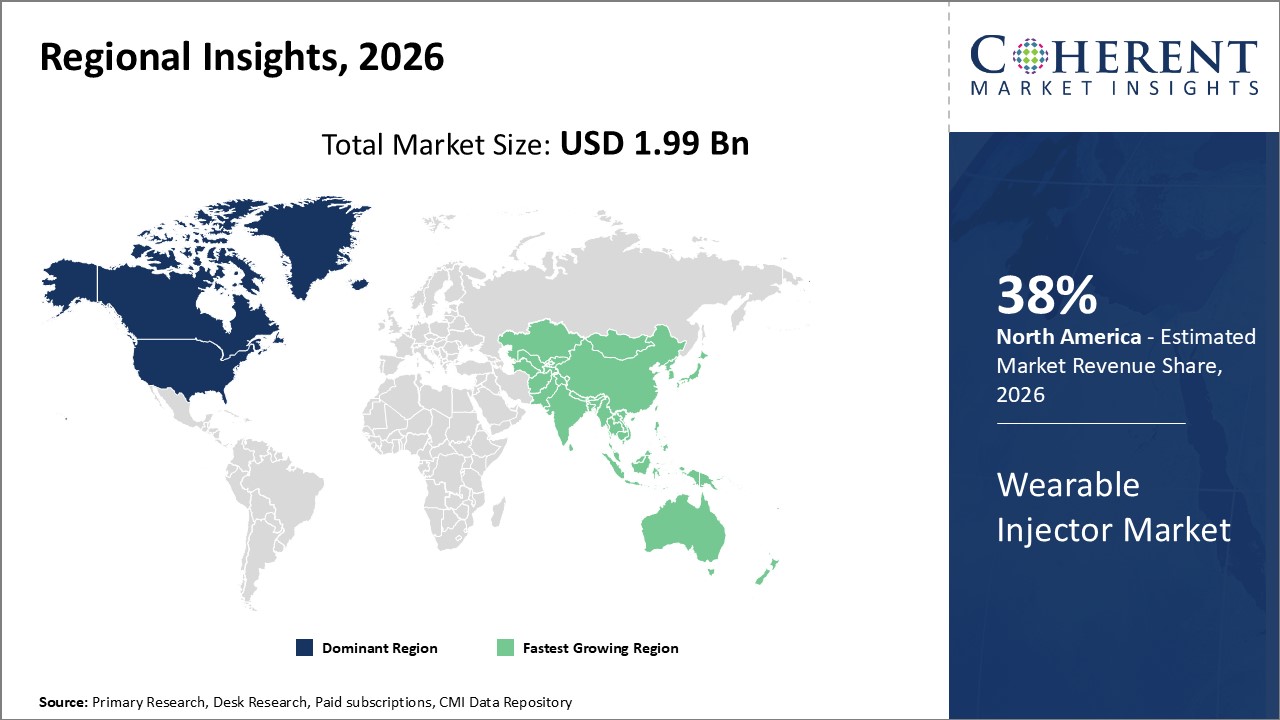
North America holds the largest market share at approximately 38%, attributed to advanced healthcare infrastructure and strong reimbursement policies. Europe's growing regulatory support and Asia Pacific's expanding healthcare investment contribute to dynamic regional market growth.
Wearable Injector Market – Segmentation Analysis
To learn more about this report, Request Free Sample
Wearable Injector Market Insights, By Product Type
In terms of product type, Disposable Wearable Injectors dominate the market share with 63%. This dominance stems from their convenience, cost-effectiveness, and suitability in single-use biologic therapies, which are expanding rapidly due to evolving treatment protocols. This segment is gaining traction due to rising demand for easy-to-use, cost-effective, and hygienic injection solutions that reduce the risk of contamination and improve patient compliance. Disposable wearable injectors offer advantages like portability, reduced need for healthcare visits, and better integration with digital health technologies for remote monitoring.
Wearable Injector Market Insights, By Application
In terms of application, Oncology being the dominating segment as the applications leverage wearable injectors to facilitate complex and prolonged treatment regimens, improving patient compliance and outcomes, reflected by a 20% increase in device prescriptions in recent oncology care protocols. This is primarily due to the increasing prevalence of cancer and the need for precise, large-volume drug delivery required for oncology treatments like chemotherapy, biologics, and immunotherapy. Wearable injectors offer continuous, self-administered subcutaneous injections that improve treatment adherence and reduce hospital visits for cancer patients.
Wearable Injector Market Insights, By End User
In terms of end user, Hospitals & Clinics dominates the market share due to institutional purchases and early technology adoption, accounting for over 55% revenue share. This segment benefits from close monitoring requirements and integration with electronic medical records. The growing need for precise medicine delivery for long-term illnesses such diabetes, cancer, autoimmune diseases, and cardiovascular diseases helps this market. Because wearable injectors are accurate, convenient, and can deliver huge amounts of biologics and chemotherapeutic drugs to patients with little discomfort, hospitals and clinics use them.
Wearable Injector Market Trends
The wearable injector market is witnessing rapid evolution with digital integration becoming a fundamental theme. Connected devices that enable remote monitoring and real-time dose tracking have gained traction, supported by healthcare providers’ need for data-driven outcomes. The shift towards disposable yet highly sophisticated injectors also denotes increased patient convenience without compromising safety, as observed in a 30% surge in disposable injector adoption in 2025 across European markets.
Moreover, partnerships between pharmaceutical companies and technology providers are fostering innovation in wearable injectors tailored for next-generation biologics and biosimilar. Asia Pacific's growing healthcare infrastructure combined with digital health initiatives has propelled early adoption trends significantly, indicating a future shift in global market shares.
Wearable Injector Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Wearable Injector Market Analysis and Trends
In North America, the dominance in the wearable injector market is underpinned by advanced biopharmaceutical research, strong payer support, and high patient awareness. The region commands around 38% market share, bolstered by industry leaders such as West Pharmaceutical Services and Phillips Healthcare. Regulatory streamlining by the FDA has facilitated faster device approvals, driving accelerated market revenue growth.
Asia Pacific Wearable Injector Market Analysis and Trends
Asia Pacific, however, is growing at the quickest rate, with a CAGR of more than 16% anticipated over the course of the forecast period. The market has expanded as a result of government initiatives supporting digital health and investments in healthcare infrastructure. Significant manufacturing and distribution contributions from businesses like Ypsomed and Nemera have further expedited expansion, particularly in China, India, and Southeast Asia.
Wearable Injector Market Outlook for Key Countries
United States Wearable Injector Market Analysis and Trends
The USA’s market is a critical driver of global wearable injector industry growth due to its well-established healthcare ecosystem and high adoption rates of advanced medical devices. In 2025, over 65% of all wearable injectors deployed worldwide were utilized in the U.S. market, driven by rising chronic diseases and a strong preference for autonomous drug delivery. Companies such as West Pharmaceutical Services and Phillips Healthcare have heavily invested in domestic manufacturing capabilities and regulatory compliance, resulting in reduced time-to-market and expanding their footprints in clinical and homecare settings.
Germany Wearable Injector Market Analysis and Trends
Germany represents the key European country for wearable injector innovation and adoption, with intensive R&D investments and supportive healthcare reimbursement frameworks. In 2025, German healthcare facilities reported a 30% increase in wearable injector integration for autoimmune and oncology therapies. Local players like Gerresheimer AG and international market companies collaborate to accelerate product launches and clinical acceptance, positioning Germany as a pivotal market within Europe’s overall wearable injector industry.
Analyst Opinion
Increasing production capacity from key manufacturers has significantly propelled the market size, with leading contract manufacturing organizations scaling up output by nearly 20% in 2025 to meet growing demand. For instance, manufacturing expansions in sterile injectable facilities in North America boosted supply-side robustness.
Demand for wearable injectors surged notably across therapeutic areas like oncology and autoimmune disorders, where patient compliance is critical. In 2026, clinical adoption increased by over 26% year-over-year as hospitals started integrating these devices into patient care protocols.
Market pricing strategies have demonstrated a gradual stabilization after initial premium pricing phases, with competitive price adjustments enabling wider accessibility. In Europe, for example, average user costs dropped by 8% in 2025, facilitating broader insurance reimbursements.
Imports of advanced wearable injectors in developing economies grew by 19% in 2025, underpinning heightened demand and cross-border market penetration. This marked an expansion of deployment across emerging markets seeking improved drug delivery methods.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.99 billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 13.6% | 2033 Value Projection: | USD 4.85 billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | West Pharmaceutical Services Inc., Philips Healthcare, Ypsomed Holding AG, SHL Group, Sensile Medical AG, Owen Mumford Ltd., Nemera, Enable Injections, Haselmeier, Unilife Corporation | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Wearable Injector Market Growth factors
The surge in chronic diseases such as diabetes and autoimmune disorders has markedly escalated the demand for wearable injectors due to their precision and ease-of-use in homecare settings. In 2025 alone, diabetes patients using wearable injection devices increased by 22% globally, reflecting a strong market driver. Advances in sensor integration and IoT-based connectivity have also enhanced device functionality, leading to better patient monitoring and adherence.
The rise of personalized medicine and biologic therapies requiring complex dosing regimens has driven demand for sophisticated injectable platforms, as seen in oncology treatments where wearable injectors have improved quality of life metrics significantly. Additionally, favorable regulatory pathways introduced in key markets like North America and Europe in 2023 streamlined approvals, mitigating market restraints and stimulating business growth.
Wearable Injector Market Development
In July 2026, A trial employing the BD Libertas wearable injector for subcutaneous biologic delivery has been announced by Becton, Dickinson and Company (BD). Following successful outcomes from over 50 pre-clinical and clinical tests conducted by BD, the injector was chosen to be used in this first pharmaceutical-sponsored combo product trial.
In October 2025, The NeoFlexTM line of coated plungers from Datwyler, a top supplier of premium, system-critical elastomer components, has expanded to include additional sizes for containers intended for wearable technology and high-volume auto-injectors. In Milan, Italy, the business debuted its NeoFlex Prefilled Syringe 10ml and NeoFlex Cartridge Wearable 10ml at CPHI Worldwide (Booth 20B48).
Key Players
West Pharmaceutical Services Inc.
Ypsomed Holding AG
SHL Group
Sensile Medical AG
Owen Mumford Ltd.
Nemera
Enable Injections
Haselmeier
Unilife Corporation
Several market players have initiated strategic alliances to advance their product portfolios and scale manufacturing capabilities. For example, West Pharmaceutical Services expanded its sterile injectable solutions infrastructure in the U.S., resulting in a 15% capacity boost and accelerated time-to-market. Ypsomed’s acquisition of enabling technology firms in 2025 has enhanced their electronic wearable injector pipeline, significantly contributing to innovation-led business growth.
Wearable Injector Market Future Outlook
Due to a number of important aspects, the wearable injector market has a very bright future. There is a growing need for sophisticated drug delivery systems that facilitate precise, reliable, and practical medicine administration due to the rising incidence of chronic illnesses like diabetes, cancer, and autoimmune disorders. Rapid adoption of patient self-administration and home-based healthcare is lowering hospital stays and medical expenses.
Device usability, safety, and real-time treatment adherence monitoring are being improved by technological advancements such as miniaturization, wireless connectivity, intelligent sensor integration, and digital health platforms. Additionally, the range of applications for wearable injectors is increased by the development of customized medicine and biologics. While Asia Pacific is predicted to expand quickly due to aging demographics and growing government backing, regional markets like North America are leading because of their strong healthcare infrastructure. Overall, the market's continued expansion and innovation will be shaped by a confluence of changing care models, technology advancements, and pressures on healthcare demand. The market's position in managing chronic diseases and providing outpatient care will be further reinforced by regulatory backing and integration with remote monitoring systems.
Historical Developments
In 2015, Unilife Corporation introduced Imperium, which was an instant patch pump used for insulin. Imperium was a subcutaneous insulin infusion, disposable, prefilled, and multi-day wearable insulin pump. It had various features that included data connectivity for apps to constantly remind and update wireless connectivity such as Bluetooth LE. It also provided real-time access to healthcare providers to track the insulin therapy of each patient.
In 2014, Enable Injections LLC signed an agreement with CSL Behring to develop novel drug delivery devices. Enable Injectors intended to improve comfort, convenience, and compliance administrative devices for the patients with rare diseases.
Enable Injection LLC and Flex entered an agreement to develop and manufacture wearable medical devices in 2016. Enable and Flex aimed to develop subcutaneous pain-free injection technology with features such as Bluetooth connectivity, monitoring systems with smartphone apps, mix automation, and passive warming systems.
Unilife Corporation entered an agreement with Amgen to develop customized wearable injectors in 2016. Unilife granted exclusive rights to Amgen for Unilife’s wearable injectors, with only selected drug classes being used for wearable injector devices. Furthermore, Unilife also granted some non-exclusive rights to Amgen for its delivery system of certain therapeutic areas that included inflammation, nephrology, oncology, bone health, neuroscience, and cardiovascular.
Sources
Primary Research interviews:
Biotechnology firms
Medical device manufacturers
Hospital procurement managers
Databases:
Scopus
ScienceDirect
SpringerLink
Magazines:
PharmaVOICE
MedTech Outlook
Drug Development & Delivery
Journals:
International Journal of Pharmaceutics
Drug Delivery and Translational Research
Advanced Drug Delivery Reviews
Newspapers:
Economic Times (Healthcare & Pharma)
Business Standard
Mint (Pharma & Biotech)
Associations:
Biotechnology Innovation Organization (BIO)
American Association of Pharmaceutical Scientists (AAPS)
European Federation of Pharmaceutical Industries and Associations (EFPIA)
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients