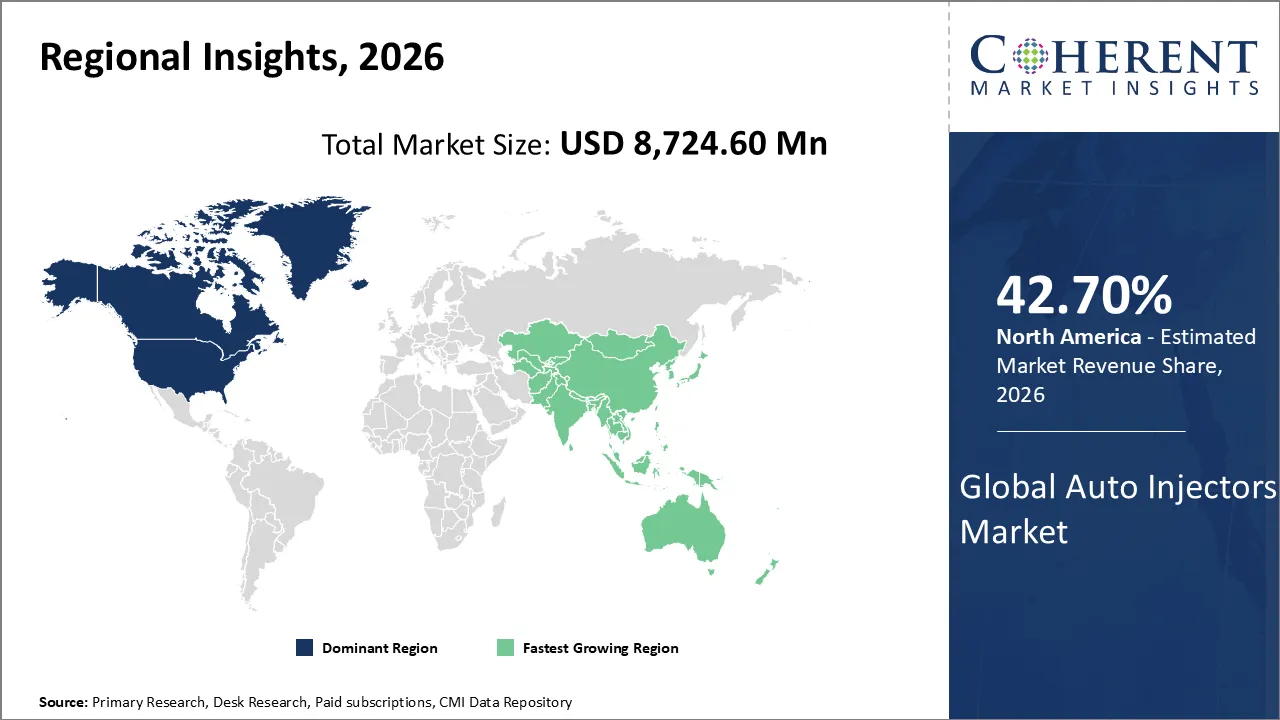
Auto injectors market is estimated to be valued at USD 8,724.60 Mn in 2026 and is expected to reach USD 27,660.0 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 18% from 2026 to 2033.
Auto-injector is a medical device which is used for injecting oneself with a single, preloaded dose of a drug. It is consists of a spring-loaded syringe which gets activated when the device is pushed firmly against the body. Auto-injector devices are useful for the rapid administration of a particular drugs and antidotes. An auto-injector has a drug cartridge with an embedded needle for subcutaneous or intramuscular injection, which is usually painless. Then, drugs are slowly delivered by the auto-injector across a large area in the muscle, which increases the drug absorption and the drug effects. Epinephrine auto-injector is a type of auto-injector widely used for people at risk of anaphylaxis. They can also be used by those who have not been medically trained. Auto-injectors are often used in the military to protect personnel from chemical warfare agents.
|
Current Event |
Description and its Impact |
|
Rising Global Diabetes and Anaphylaxis Prevalence |
|
|
Regulatory and Safety Developments |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of product type, the prefilled auto-injectors segment is expected to lead the market with 64% share in 2026, because they are convenient, safe, and dependable. Patients prefer them for emergencies, especially when they have anaphylaxis, as they eliminate preparation steps and they lower the risk of dosing errors. Pharmaceutical companies also offer prefilled formats for biologics, which helps them become popular in all therapeutic areas and stay at the top of the market in 2026.
For instance, in October 2025, Ypsomed's release of YpsoLoop is directly related to prefilled auto-injectors, which brings sustainability to the most popular type of auto-injector. It is designed to be circular, which reduce on waste while keeping safety and convenience. This new idea makes prefilled auto-injectors increasingly popular by making them easy for patients to use and sustainable. This makes them useful in both healthcare and environmental responsibility.
In terms of disease indication, the anaphylaxis segment is expected to hold 39% share of the market in 2026, due to the rising prevalence of severe allergic reactions worldwide. Epinephrine auto-injectors are life-saving devices that are commonly prescribed and kept in schools, workplaces, and homes. Since they are so important in emergency care, they will always be in high demand. This means that anaphylaxis will be the most prevalent health indication segment in 2026.
In terms of distribution channel, the retail pharmacies segment is projected to account for the largest share of the market in 2026, because they may provide them to patients immediately, like epinephrine auto-injectors. They are the main channel as they are widely available, pharmacists can be trusted to help, and insurance covers them. While online sales grow rapidly, retail pharmacies remain the most significant distribution hub in 2026 for auto-injectors.
For instance, in October 2025, the release of Brekiya, the first DHE auto-injector for migraine and cluster headaches, by Amneal expands the uses of auto-injectors beyond anaphylaxis. As a prescription drug, it will mostly be distributed through retail pharmacies, making it easy for patients to get. This new idea makes the auto-injector market stronger while also meeting needs in neurological treatment that haven't been met before.
To learn more about this report, Download Free Sample
North America is expected to dominate the auto-injectors market with 42.70% share in 2026, due to chronic diseases are becoming more common, biologics are growing more popular, strong healthcare infrastructure, and favorable reimbursement policies. Patient preference for self-administration, the presence of major pharmaceutical companies, and widespread knowledge of anaphylaxis and autoimmune conditions all contribute to regional dominance.
For instance, in January 2026, MGS launched the A.i.r. platform in Germantown, Wisconsin, USA, introducing auto-injectors more design options. This new idea helps drug companies and healthcare providers by enabling them make solutions that are tailored to each patient. The platform makes the global auto-injector market stronger by combining ease of use and long-term reliability to meet changing therapeutic needs in many regions and for many conditions.
Asia Pacific is anticipated to be the fastest growing region, due to rising chronic disease prevalence, expanding healthcare infrastructure, and growing middle-class affordability. The region is growing the fastest in the world as greater numbers of individuals are aware of self-administration, the government is promoting home-based care, and biologics can be made at low costs in countries like India and China.
For instance, in August 2025, Hanmi Pharmaceutical plans to release an auto-injector version of Rolontis in South Korea. The device has a hidden needle that makes it easier for patients to use, cuts down on hospital visits, and allows neutropenia patients to give themselves care. This new use of auto-injectors in oncology improves adherence and makes treatment easier for cancer patients.
The U.S. auto-injectors market is growing as more individuals are getting anaphylaxis, diabetes, and autoimmune disorders. Growth is driven by a strong demand for biologics, better healthcare infrastructure, and good reimbursement policies. Patients want to give themselves their own medications, and leading pharmaceutical companies are always launches with new ways to do this. This keeps the U.S. market strong.
For instance, in September 2025, Sharp Services invested $20 million on U.S. facilities to improve their ability to assemble auto-injectors and pens. This strategic move strengthens domestic manufacturing, makes the supply chain more resilient, and helps pharmaceutical companies with production that can grow. The investment increases innovation and accessibility, which strengthens the United States' position in the global auto-injector market.
China's auto-injectors market will be extremely active in 2026 because diabetes, allergies, and autoimmune disorders are becoming more common, and the country's healthcare system is growing quickly. More affordable middle-class goods, government support for self-care, and local biologics manufacturing all make things easier to get. More people are aware of the benefits of easy self-administration, which leads to strong adoption. This makes China a key growth engine.
For instance, in July 2025, using its auto-injector platform, Ypsomed launched the first dual GCG/GLP-1 receptor agonist for treating obesity in China. This milestone expands new uses for auto-injectors in metabolic care, making it easy for people to give themselves injections. The new technology makes it easier for patients to follow their treatment plans, strengthens China's position in advanced drug delivery, and shows that Ypsomed is the world's leader in device solutions.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8,724.60 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 18% | 2033 Value Projection: | USD 27,660.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Becton, Dickinson and Company, Sanofi S.A., Takeda Pharmaceutical Company Limited, Xeris Biopharma Holdings Inc., BlackHagen Design, GSK plc., Genentech, Inc., AstraZeneca, Owen Mumford Ltd., Stevanato Group, Jabil Inc., Pfizer, Inc., Mylan N.V., Novartis AG, Bayer AG, Janssen Pharmaceuticals, Inc., Antares Pharma, Eli Lilly and Company, Amgen Inc., Rafa Laboratories Ltd., Halozyme, Inc., Biogen, and Teva Pharmaceutical Industries Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
A significant reason for people to starting to using auto-injectors is due to the fact a rising number of individuals are getting chronic diseases like diabetes and rheumatoid arthritis. Patients need to take insulin and biologics often and exactly, so auto-injectors are a useful option. Also, these devices help people with multiple sclerosis and obesity get the right amount of medicine at the right time. The Auto-injectors Market share continues to surge as more patients choose to give themselves injections, resulting in many opportunities for manufacturers and healthcare providers around the world to grow their businesses.
There is a growing incidence rate of severe allergic reactions, which has greatly increased the need for epinephrine auto-injectors. In an emergency, these devices can save lives as they are quick, dependable, and easy to use for patients and caregivers. Rising awareness regarding anaphylaxis, and more easy-to-use devices are becoming available. As a result, the auto injectors market demand is rising quickly. Both healthcare systems and individuals want easy access to these important tools for immediate help during allergic reactions.
The combination of digital health features like dose tracking, Bluetooth connectivity, and reminders are influencing the ways that patients stick to their treatment plans for chronic diseases. These improvements make it easier for patients to keep track of their treatment schedules, which means fewer missed doses and better results. Healthcare providers also benefit from sharing data in real time, which makes care more personalized. The Auto-injectors Market forecast predicts strong growth because digital-enabled devices are becoming more popular with medical professionals and patients around the world.
The market for auto-injectors keeps growing as a growing number of healthcare settings need devices that allow patients give themselves drugs. Recent market studies show that the sector makes an extensive amount of funds around the world, thanks to the widespread use of auto-injectors for managing chronic diseases, treating emergencies like anaphylaxis, and other medical uses. The largest product category is disposable auto-injectors, as they are easy to use, do not require much training, and work with many biologic therapies. Smart and connected injectors that keep track of dosages are becoming popular new technologies. They are a response to the needs of patients and providers for better compliance and remote monitoring.
Data on therapeutic applications show that rheumatoid arthritis and diabetes are two of the main groups that use auto-injectors, supported biologic and subcutaneous drugs are common in these areas of treatment. Epinephrine auto-injectors and other devices for treating anaphylaxis are still essential for being ready for emergencies. They are also becoming more common in public health guidelines and educational campaigns.
North America is the leading market with the highest adoption rates, due to its well-established healthcare system and the fact that many patients prefer to receive care at home. The Asia-Pacific markets are growing because more individuals are becoming aware of them, healthcare services are expanding, and more people are getting chronic diseases. Home care settings are the most prevalent settings for individuals to use the product. Many patients and caregivers prefer the ease of self-administered injections to clinic-based services.
Developments in demographics, an increase in the need for chronic disease treatment, and technological improvements that make auto-injectors easier to use, safer, and more likely to be used by patients all affect the overall demand for them.
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients