Carcinoid Syndrome Diarrhea Treatment Market Size and Forecast – 2026 – 2033
The Global Carcinoid Syndrome Diarrhea Treatment Market size is estimated to be valued at USD 450 million in 2026 and is expected to reach USD 740 million by 2033, exhibiting a compound annual growth rate (CAGR) of 7.2% from 2026 to 2033.
Global Carcinoid Syndrome Diarrhea Treatment Market Overview
The carcinoid syndrome diarrhea treatment market is driven by the rising prevalence of neuroendocrine tumors and increasing awareness regarding carcinoid syndrome symptoms and diagnosis. Growing demand for targeted therapies and advanced somatostatin analogs is supporting market expansion. Improved healthcare infrastructure, higher screening rates, and increasing access to specialized oncology treatments are further boosting demand. Pharmaceutical companies are investing in research and development to introduce more effective therapies with fewer side effects. Additionally, favorable reimbursement policies and expanding clinical trials for novel drugs are accelerating market growth. North America dominates the market due to advanced healthcare systems, while Asia-Pacific is witnessing gradual growth with improving cancer care services.
Key Takeaways
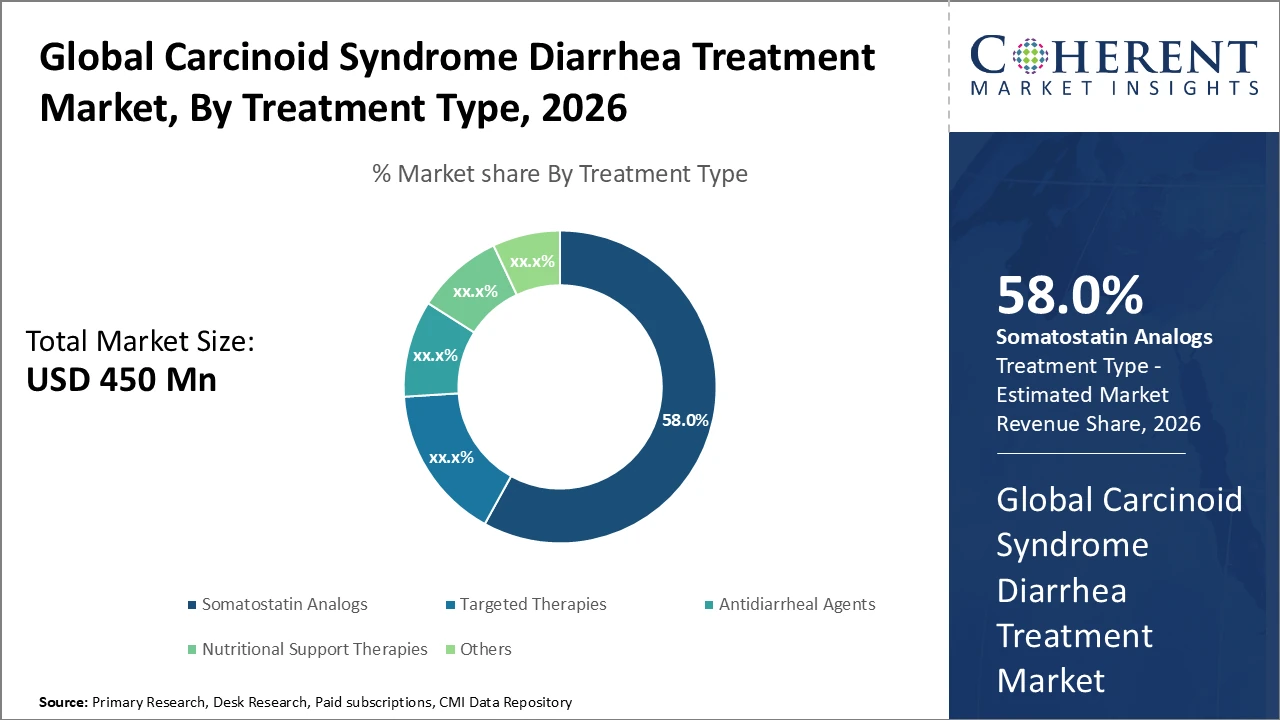
The somatostatin analog segment continues to dominate therapeutic interventions, generating over 58% of market revenue in 2026, driven by innovation in long-acting formulations.
Injectable administration remains the preferred route, capturing nearly 60% of the market, supported by clinical preference and improved bioavailability.
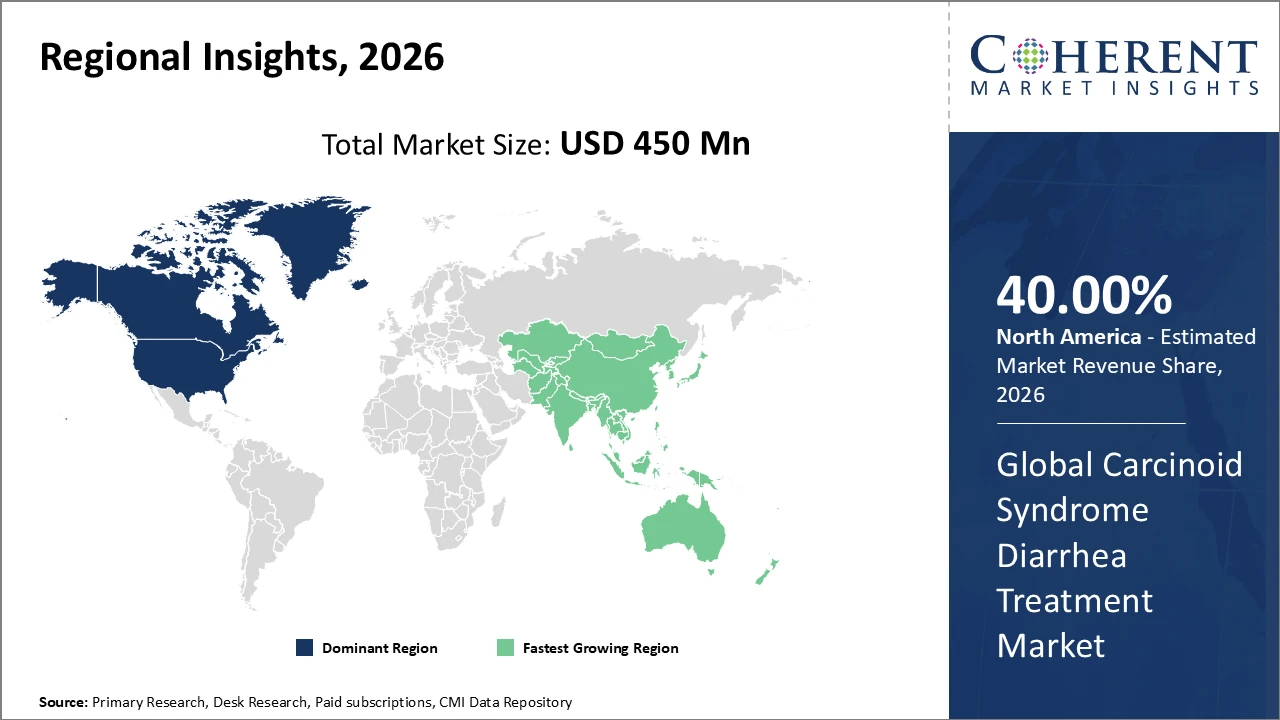
North America holds the largest industry share, accounting for approximately 40% of revenue, supported by high diagnosis rates and advanced healthcare infrastructure.
Asia Pacific exhibits the fastest market growth, with a CAGR exceeding 9%, due to expanding healthcare access, increasing oncology awareness, and favorable government policies.
Carcinoid Syndrome Diarrhea Treatment Market Segmentation Analysis
To learn more about this report, Download Free Sample
Carcinoid Syndrome Diarrhea Treatment Market Insights, By Treatment Type
Somatostatin analogs dominate the Carcinoid Syndrome Diarrhea Treatment market due to their proven effectiveness in controlling hormone-related diarrhea and the availability of long-acting formulations that improve patient adherence. The targeted therapies segment is the fastest growing, fueled by advances in molecular research that enable precise tumor pathway targeting, enhancing symptom control beyond standard treatments. Antidiarrheal agents offer symptomatic relief but exhibit slower growth because of limited efficacy as standalone therapies. Nutritional support and other adjunctive therapies primarily focus on improving patient quality of life during and after treatment, complementing core therapeutic approaches and supporting comprehensive symptom management.
Carcinoid Syndrome Diarrhea Treatment Market Insights, By Route of Administration
Injectable routes dominate the Carcinoid Syndrome Diarrhea Treatment market due to superior bioavailability and clinical preference for managing hormone-related diarrhea. Long-acting injectable formulations reduce dosing frequency, significantly improving patient adherence and convenience. The oral segment is the fastest growing, driven by the development of novel oral somatostatin analogs that offer ease of administration without compromising therapeutic efficacy. Transdermal treatments remain a niche segment with limited market share but provide an emerging alternative for patients who prefer to avoid injections.
Carcinoid Syndrome Diarrhea Treatment Market Insights, By End User
Hospitals continue to dominate the Carcinoid Syndrome Diarrhea Treatment market, holding the highest share due to their capacity to provide integrated care, including diagnosis, treatment, and continuous monitoring of patients. Specialty clinics represent the fastest-growing subsegment, driven by increasing focus on neuroendocrine tumors and the delivery of tailored therapeutic regimens. Homecare settings are gaining adoption as long-acting injectable therapies become more patient-friendly, supported by telemonitoring and remote care technologies that enhance treatment adherence.
Carcinoid Syndrome Diarrhea Treatment Market Trends
The Carcinoid Syndrome Diarrhea Treatment market is increasingly focused on precision medicine, with 2025 clinical evidence showing that combination therapies tailored to individual molecular profiles improved symptom management outcomes by nearly 30%.
Digital health technologies, including telehealth platforms, are being integrated to enable remote monitoring of treatment adherence, first widely adopted across European markets in 2024.
The trend of home administration, especially for injectable therapies, has enhanced patient compliance in North America by up to 25%, reflecting a shift toward patient-centric care models.
Overall, the market is being driven by novel treatment approaches and digital innovation.
Carcinoid Syndrome Diarrhea Treatment Market Insights, By Geography
To learn more about this report, Download Free Sample
North America Carcinoid Syndrome Diarrhea Treatment Market Analysis and Trends
In North America, the Carcinoid Syndrome Diarrhea Treatment market is dominated by a well-established healthcare ecosystem, advanced research infrastructure, and high awareness of neuroendocrine tumor management. The region accounts for approximately 40% of the market, supported by strong production capacity, comprehensive reimbursement mechanisms, and widespread access to innovative therapies. Government incentives promoting rare disease treatments have further stimulated clinical trial activity and accelerated drug approvals. Key players such as Novartis and Ipsen lead the market, leveraging advanced somatostatin analogs and targeted therapies. These factors collectively strengthen North America’s position as a major revenue contributor and innovation hub in the global market.
Asia Pacific Carcinoid Syndrome Diarrhea Treatment Market Analysis and Trends
Asia Pacific demonstrates the fastest growth in the Carcinoid Syndrome Diarrhea Treatment market, with a CAGR exceeding 9%, fueled by rapid development of healthcare infrastructure, expanding oncology awareness programs, and enhanced treatment affordability through supportive government initiatives. China and India play a central role in this growth, driven by increasing import volumes of pharmaceutical agents and rising rates of early diagnosis for neuroendocrine tumors. Strategic collaborations between multinational companies and regional healthcare providers have further accelerated the adoption of advanced therapeutics. These factors collectively position Asia Pacific as a key emerging market with significant potential for revenue expansion and treatment accessibility.
Carcinoid Syndrome Diarrhea Treatment Market Outlook for Key Countries
USA Carcinoid Syndrome Diarrhea Treatment Market Analysis and Trends
The USA continues to be a key driver of the North American Carcinoid Syndrome Diarrhea Treatment market, contributing over 70% of regional revenue in 2026. This leadership is supported by early and accurate diagnosis enabled by advanced biomarker technologies, which increase demand for effective therapies. Between 2024 and 2026, adoption of long-acting somatostatin analogs rose by 15%, reflecting strong clinical endorsement from major academic and specialty centers. Additionally, comprehensive insurance coverage, coupled with government incentives for rare disease drug approvals, further supports market expansion. These factors collectively position the USA as the dominant and most influential market in the region.
Germany Carcinoid Syndrome Diarrhea Treatment Market Analysis and Trends
Germany’s Carcinoid Syndrome Diarrhea Treatment market is a key contributor in Europe, driven by extensive hospital networks specializing in neuroendocrine tumor and carcinoid syndrome management. Government-backed reimbursement policies have significantly improved patient access, resulting in an 18% increase in therapy uptake in 2025. Local companies have adopted value-based pricing strategies to further enhance affordability and accessibility of novel treatments. Strategic collaborations between pharmaceutical manufacturers and healthcare institutions have accelerated the adoption of advanced therapies, supporting more comprehensive patient care. These factors collectively strengthen Germany’s position as a leading European market with growing revenue potential in carcinoid syndrome management.
Analyst Opinion
Treatment Modalities Expansion: Innovative therapies such as somatostatin analogs and targeted treatments accounted for over 65% of market revenue in 2024. The approval of novel long-acting somatostatin analogs in 2025 improved patient adherence by 18%, supporting market growth.
Patient Diagnosis Rates: Advances in biomarker discovery and imaging techniques in 2026 increased identifiable carcinoid syndrome cases by 12%, expanding the patient pool and driving demand for effective diarrhea treatments.
Geographic Penetration and Treatment Accessibility: Exports of specialized drugs from North America to Asia Pacific grew by 22% in 2025, improving cross-border healthcare access and boosting market revenue.
Pricing and Reimbursement Policies: Expanded insurance coverage and strategic pricing in European regions led to a 15% rise in reimbursements in 2024, enhancing market penetration and revenue for key players.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 450 million |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.2% | 2033 Value Projection: | USD 740 million |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Ipsen Pharma, Novartis AG, Pfizer Inc., Sanofi SA, AstraZeneca PLC, Mylan N.V., Incyte Corporation, Cipla Limited, Leo Pharma A/S, Lundbeck A/S | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Carcinoid Syndrome Diarrhea Treatment Market Growth Factors
The expanding patient base, driven by higher carcinoid tumor diagnosis rates, is a key driver of global demand for Carcinoid Syndrome Diarrhea treatments. Rising prevalence of carcinoid syndrome-related diarrhea has strengthened market dynamics, particularly across North America and Europe, contributing to growing industry share. Advances in drug development, including novel somatostatin analogs and targeted biologics, have enhanced treatment efficacy, boosting revenue. Additionally, improved reimbursement policies and government initiatives in emerging markets, especially Asia Pacific, have increased treatment accessibility. The surge in clinical trials between 2024 and 2026 for innovative therapies further reinforces growth, creating broader opportunities and expanding the overall market potential.
Carcinoid Syndrome Diarrhea Treatment Market Development
In July 2025, National Health Mission launched the “Stop Diarrhea Campaign-2025” across Tripura to eliminate child deaths caused by diarrhea.
Key Players
Leading Companies of the Market
Ipsen Pharma
Novartis AG
Pfizer Inc.
Sanofi SA
Mylan N.V.
AstraZeneca PLC
Incyte Corporation
Cipla Limited
Leo Pharma A/S
Lundbeck A/S
Competitive strategies in the Carcinoid Syndrome Diarrhea Treatment market focus on research collaborations, product lifecycle expansions, and strategic acquisitions. For instance, Ipsen’s acquisition initiatives in 2025 strengthened its therapeutic pipeline, leading to a 20% increase in market share. Similarly, Novartis emphasized innovative drug delivery mechanisms, which boosted revenue from its injectable products by 28% between 2024 and 2026, demonstrating strong business growth. These strategic approaches highlight how leading companies leverage innovation, portfolio enhancement, and targeted investments to consolidate their market positions, drive revenue expansion, and maintain a competitive edge in the rapidly evolving global treatment landscape.
Carcinoid Syndrome Diarrhea Treatment Market Future Outlook
The future of the Carcinoid Syndrome Diarrhea Treatment market is expected to be shaped by the growing prevalence of neuroendocrine tumors and advancements in targeted therapies. Innovations in somatostatin analogs, peptide receptor radionuclide therapy (PRRT), and combination treatments are likely to enhance symptom control and improve patient quality of life. Personalized medicine approaches, including biomarker-driven therapies, will enable more effective and tailored treatment strategies. Additionally, increasing awareness among healthcare providers, early diagnosis, and supportive care options are expected to drive market growth. Overall, the market is poised for expansion, with a focus on improved efficacy, patient adherence, and integrated therapeutic solutions.
Carcinoid Syndrome Diarrhea Treatment Market Historical Analysis
The Carcinoid Syndrome Diarrhea Treatment market has evolved significantly over the past decade, transitioning from basic symptomatic management to targeted therapeutic approaches. Early treatments relied heavily on short-acting somatostatin analogs to control hormone-related diarrhea, providing temporary relief with frequent dosing requirements. Over time, the introduction of long-acting formulations improved patient compliance and symptom control. Advances in peptide receptor radionuclide therapy (PRRT) and targeted therapies further expanded treatment options, addressing both tumor progression and symptom management. Increased awareness of neuroendocrine tumors, improved diagnostic techniques, and growing investment in innovative therapies have collectively driven historical market growth and shaped the foundation for future expansion.
Sources
Primary Research Interviews:
Medical researchers, R&D managers, and product developers from companies manufacturing somatostatin analogs, PRRT agents, and other carcinoid syndrome diarrhea therapies.
Clinicians, oncologists, and gastroenterologists treating patients with neuroendocrine tumors experiencing carcinoid syndrome diarrhea.
Industry experts from firms providing targeted therapies, AI-assisted treatment monitoring, and supportive care solutions for symptom management.
Magazines:
Oncology Times – Updates on neuroendocrine tumor treatments, symptom management, and therapy advancements.
Clinical Oncology News – Trends in patient care, therapeutic innovations, and drug adoption.
Pharmaceutical Executive – Market developments, product launches, and innovations in gastrointestinal and neuroendocrine therapies.
Journals:
Journal of Clinical Oncology – Research on carcinoid syndrome management, PRRT efficacy, and clinical outcomes.
Neuroendocrinology – Studies on hormone-related tumor symptoms, treatment response, and patient quality of life.
Digestive Diseases and Sciences – Insights on gastrointestinal symptom management and therapeutic interventions.
Newspapers:
The Wall Street Journal – Company strategies, mergers, and investments in carcinoid syndrome therapies.
Financial Times – Market trends, regulatory updates, and global demand for neuroendocrine tumor treatments.
Reuters – News on clinical trials, product launches, and healthcare advancements.
Bloomberg – Corporate performance and global market insights in oncology and specialty therapeutics.
Associations:
European Society for Medical Oncology (ESMO) – Guidelines for neuroendocrine tumor management and treatment protocols.
American Society of Clinical Oncology (ASCO) – Standards for carcinoid syndrome treatment and patient care recommendations.
World Health Organization (WHO) – Global guidelines for rare tumor management and symptom control therapies.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients