Targeted therapy is emerging pharmacotherapy for cancer. It is a type of precision cancer treatment. Targeted therapy used to treat breast cancer, bladder cancer, colorectal cancer kidney cancer, leukemia, liver cancer, lung cancer, lymphoma and prostate cancer. Targeted therapies are made to find and attack specific areas or substances in cancer cells.
The global targeted therapy market is estimated to be valued at US$ 98.6 Bn in 2021 and is expected to exhibit a CAGR of 9.0% during the forecast period (2021-2028).
Figure 1.Global Targeted Therapy Market in Terms of Value (US$ Billion), By Region, 2021
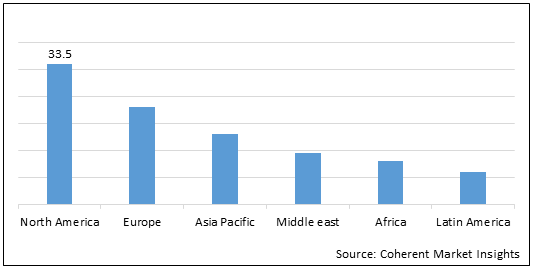
To learn more about this report, Request Free Sample
Strong focus on research and development by market players is estimated to drive the global targeted therapy market growth during the forecast period.
For instance, in 1970, Tamoxifen the first targeted cancer therapy was approved by U.S. Food and Drug Administration. The discovery of oncogenes and tumor suppressor genes, and the completion of human genome sequencing fueled some major advances in treating the cancer. Newly emerging biological and genetic information rapidly prompted the introduction of a large number of new targeted cancer therapies. Thus, key players are investing in the research and development and thereby it is expected to drive the market growth. For instance, Boehringer Ingelheim a pharmaceutical companies announced the initiation of a phase II clinical trial of BI 764198, an inhibitor of TRPC6, a receptor-operated cation channel. This therapy may provide the first potential treatment for COVID-19 related respiratory distress syndrome.
Targeted Therapy Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 98.6 Bn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 9.0% | 2028 Value Projection: | US$ 165.4 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Amgen Inc., GlaxoSmithKline plc, Sanofi, Takeda Pharmaceutical Company Limited, Merck & Co., Inc., Novartis AG, Pfizer Inc., F. Hoffmann-La Roche Ltd, AstraZeneca, Seagen Inc., Bayer AG, and Bristol Myers Squibb |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. Global Targeted Therapy Market Share (%), By Type, 2021
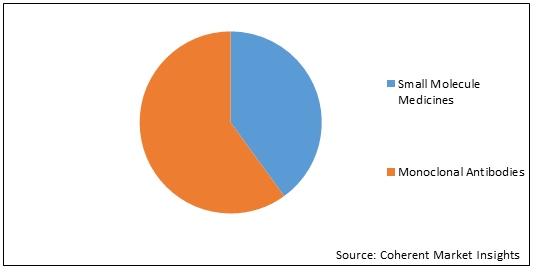
To learn more about this report, Request Free Sample
Strategic development by key market players is anticipated to propel the global targeted therapy market growth over the forecast period.
Strategic development such as acquisition, collaboration, partnership, and others to strengthen the market position is likely expected to boost the growth of market. For instance, in May 2021, Xilio Therapeutics, a biotechnology company, entered into a clinical trial collaboration and supply agreement with Merck outside the U.S. and Canada, to evaluate XTX101, Xilio’s engineered tumor-selective Fc-enhanced, anti-CTLA-4 monoclonal antibody (mAb) product candidate, in combination with KEYTRUDA (pembrolizumab), Merck’s anti-PD-1 (programmed death receptor-1) therapy.
Global Targeted Therapy Market – Impact of Coronavirus (COVID-19) Pandemic
COVID-19 causes several challenges to cancer practice, especially to ensure continuity of treatment during this period. It has significantly affected healthcare systems and economies within a short timeframe. Various diagnostic & research centres had to slow their activity due to public health measures. Thus, the emerging cancer research was affected due to the COVID-19.
However, targeted therapy is being evaluated in COVID-19 treatment. Thus, by encouraging clinicians to pursue targeted therapy agents during the pandemic is likely expected to create opportunity during the forecast period.
Global Targeted Therapy Market: Restraint
Cost is a major factor influencing the selection of targeted therapy products. The drugs prescribed in targeted therapy treatment are often prohibitively expensive. Thus, higher cost of therapy is likely to hamper the growth of market. For instance, monthly averages of targeted therapy is US$ 5,000 to US$ 10,000 and annual totals over US$ 100,000.
Key Players
Major players operating in the global targeted therapy market include Amgen Inc., GlaxoSmithKline plc, Sanofi, Takeda Pharmaceutical Company Limited, Merck & Co., Inc., Novartis AG, Pfizer Inc., F. Hoffmann-La Roche Ltd, AstraZeneca, Seagen Inc., Bayer AG, and Bristol Myers Squibb.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients