Cytomegalovirus Infection Treatment And Diagnosis Market Size and Forecast – 2026 – 2033
The Cytomegalovirus Infection Treatment & Diagnosis market is projected to grow from an estimated USD 1.5 Bn in 2026 to about USD 2.7 Bn by 2033, expanding at a CAGR of 7.5 % over 2026–2033, reflecting strong demand for diagnostics and therapies for CMV infections worldwide.
Global Cytomegalovirus Infection Treatment And Diagnosis Market Overview
Cytomegalovirus (CMV) infection, caused by a common herpesvirus, can lead to severe complications in immunocompromised individuals, transplant recipients, and newborns. Diagnosis involves serological tests, PCR-based assays, and viral culture to detect active or latent infections. Treatment primarily includes antiviral medications such as ganciclovir, valganciclovir, foscarnet, and cidofovir, tailored to patient risk and infection severity. The market for CMV diagnosis and treatment is expanding due to rising awareness, increasing immunocompromised populations, and technological advancements in rapid and accurate diagnostics. Ongoing research aims to improve therapeutics and develop effective vaccines to prevent CMV infections globally.
Key Takeaways
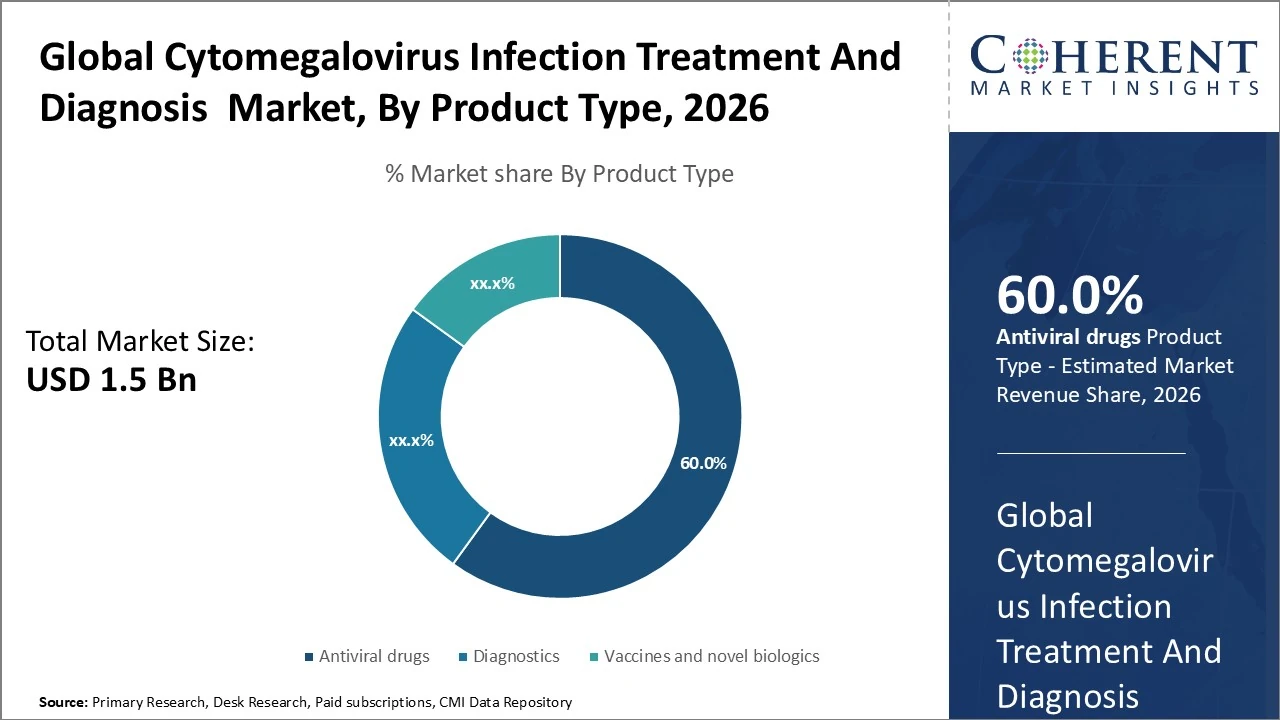
The global Cytomegalovirus infection treatment and diagnosis market is dominated by Antiviral drugs, holding an estimated 60% share.
In the Cytomegalovirus infection treatment and diagnosis market, molecular diagnostic technologies, especially PCR‑based assays, lead with roughly 48 % share.
The CMV infection treatment and diagnosis market is led by Hospitals, capturing the largest share at around 50 %.
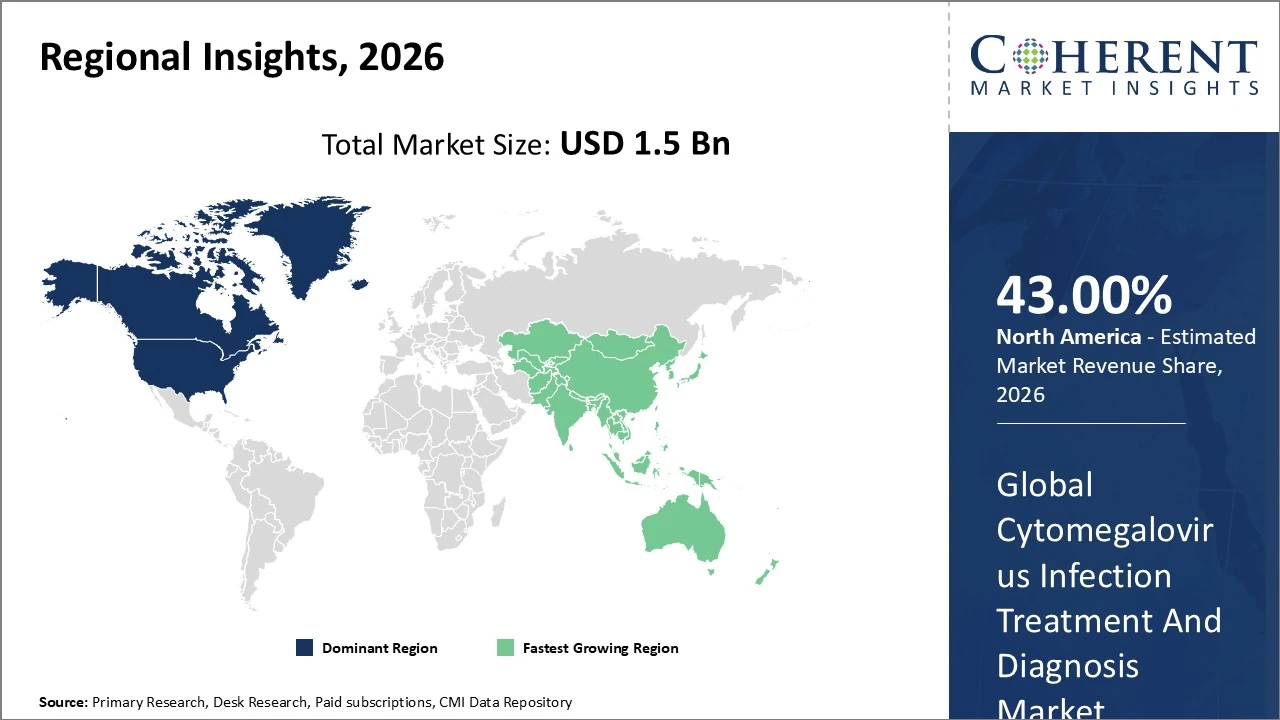
North America dominates the Cytomegalovirus infection treatment and diagnosis market, accounting for around ~43 % of global revenue.
In Asia Pacific, the cytomegalovirus infection treatment and diagnosis market holds roughly ~20 % of global revenue.
The United States market contributes an estimated ~25 % of the global CMV treatment market.
Cytomegalovirus Infection Treatment And Diagnosis Market Segmentation Analysis
To learn more about this report, Request Free Sample
Cytomegalovirus Infection Treatment And Diagnosis Market Insights, By Product Type
Antiviral drugs dominate, holding an estimated 60% share due to widespread use of ganciclovir, valganciclovir, and other polymerase inhibitors in clinical settings. Diagnostics account for approximately 25 % of the market, driven by rising demand for PCR and serology tests for early detection and monitoring. Vaccines and novel biologics represent the remaining share (~15 %), with growth potential as preventive solutions advance through development and clinical evaluation.
Cytomegalovirus Infection Treatment And Diagnosis Market Insights, By Technology
In the Cytomegalovirus infection treatment and diagnosis market, molecular diagnostic technologies, especially PCR‑based assays, lead with roughly 48 % share due to high sensitivity and rapid detection in high‑risk groups. Serological methods (e.g., ELISA) follow with about 35 % share, widely used for screening and immune status evaluation. Antigen and culture techniques comprise the remainder (17 %), mainly in specialized labs. On the treatment side, traditional antiviral therapies using advanced drug‑discovery platforms dominate technology adoption, while emerging biologics and immunotherapies are gaining traction with increasing R&D investment and innovation.
Cytomegalovirus Infection Treatment And Diagnosis Market Insights, By End-User
The CMV infection treatment and diagnosis market is led by Hospitals, capturing the largest share at around 50%, due to inpatient management, transplant care, and integrated diagnostic services in clinical settings. Specialty clinics and outpatient centers follow with roughly 25 %, driven by oral therapies and focused CMV monitoring programs. Diagnostic laboratories contribute about ~15 %, reflecting their role in PCR and serological testing for early detection and viral load monitoring. Other end‑users such as research institutes and ambulatory surgical centers make up the remaining share, supporting innovation and niche services.
Cytomegalovirus Infection Treatment And Diagnosis Market Trends
Molecular technologies like PCR, digital PCR, and point‑of‑care assays are improving early detection and viral load monitoring, accelerating clinical decision‑making and patient management.
Next‑generation antivirals, immunotherapies (e.g., virus‑specific T‑cells), and combination regimens are emerging to enhance efficacy, reduce toxicity, and address resistance challenges.
Increased emphasis on prophylactic treatments and vaccine development reflects a preventive market focus, aiming to reduce incidence in high‑risk groups like transplant recipients and pregnant women.
Cytomegalovirus Infection Treatment And Diagnosis Market Insights, By Geography
To learn more about this report, Request Free Sample
North America Cytomegalovirus Infection Treatment And Diagnosis Market Analysis and Trends
North America dominates the Cytomegalovirus infection treatment and diagnosis market, accounting for around 43% of global revenue due to advanced healthcare infrastructure, high organ transplant volumes, extensive antiviral R&D, and strong adoption of molecular diagnostics like PCR testing. The U.S. leads regionally, driving most demand with proactive screening programs and favorable reimbursement policies, contributing significantly to early detection and treatment uptake. Growth trends include integration of rapid diagnostic technologies and expanded use of prophylactic antivirals in high‑risk patients. Investments in novel therapies and heightened clinician awareness are expected to sustain North America’s market leadership through the forecast period.
Asia Pacific Cytomegalovirus Infection Treatment And Diagnosis Market Analysis and Trends
In Asia Pacific, the cytomegalovirus infection treatment and diagnosis market holds roughly 20 % of global revenue in 2026, driven by expanding healthcare infrastructure, rising organ transplant procedures, and improved CMV awareness in China, Japan, and India. Diagnostic uptake, particularly PCR testing, is increasing as regional capabilities improve. The market is also the fastest‑growing globally, with projected CAGR of ~9 % through the forecast period, supported by rising healthcare spending and government initiatives to enhance infectious disease management. However, variability in access and affordability across emerging economies continues to influence penetration rates.
Cytomegalovirus Infection Treatment And Diagnosis Market Outlook for Key Countries
USA Cytomegalovirus Infection Treatment And Diagnosis Market Analysis and Trends
The United States market leads globally due to advanced healthcare infrastructure, strong R&D, and high clinical adoption of CMV diagnostics and therapies, contributing an estimated ~25 % of the global CMV treatment market. U.S. demand is driven by transplant patient care, rising immunocompromised populations, and FDA approvals of novel treatments like maribavir for resistant post‑transplant CMV infections. Adoption of molecular PCR diagnostics and prophylactic protocols is expanding, improving early detection and management. While antiviral drugs dominate, innovation in biologics and precision therapies is accelerating, with increasing clinical investment sustaining market growth.
Germany Cytomegalovirus Infection Treatment And Diagnosis Market Analysis and Trends
In Germany, the cytomegalovirus (CMV) market is evolving with steady growth in diagnostics and antiviral therapy adoption. The country’s well‑structured healthcare system emphasizes evidence‑based CMV management and national guidelines for diagnosis and treatment, particularly in pregnancy and neonatal care. CMV seroprevalence among adults is significant, with about ~57 % carrying antibodies, influencing diagnostic demand. PCR and serological testing are widely used for early detection, especially in immunocompromised patients and transplant recipients. Therapeutic uptake, including antivirals, is supported by reimbursement policies, while research and clinical initiatives drive innovation in preventive and treatment strategies, sustaining moderate market expansion.
Analyst Opinion
Analysts expect steady expansion at a CAGR of 7.5 % (2026–2033), driven by rising immunocompromised populations and increasing transplant procedures globally.
Molecular assays like PCR dominate, with analysts noting a shift toward rapid, point-of-care diagnostics for early detection and monitoring.
Ganciclovir, valganciclovir, and foscarnet are projected to maintain the largest revenue share, though new biologics and immunotherapies are gaining attention.
Analysts highlight U.S. infrastructure, high transplant rates, and favorable reimbursement policies as sustaining regional dominance.
Analysts identify preventive vaccines and prophylactic therapies as key long-term growth drivers, particularly in Asia Pacific and emerging markets.
Market Scope
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.5 Billion |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.5% | 2033 Value Projection: | USD 2.7 Billion |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: | Gilead Sciences, F. Hoffmann-La Roche, GlaxoSmithKline, Merck & Co., ViiV Healthcare, Pfizer | ||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Cytomegalovirus Infection Treatment And Diagnosis Market Growth Factors
The Cytomegalovirus Infection Treatment and Diagnosis market is growing due to several key factors. Rising numbers of immunocompromised patients, including transplant recipients and HIV-positive individuals, increase demand for effective treatment and monitoring. Technological advancements in diagnostics, particularly PCR and rapid point-of-care tests, enable early detection and improved patient outcomes. Increasing awareness among clinicians and patients about CMV risks drives proactive screening and therapy. Expanding healthcare infrastructure in emerging markets and favorable government initiatives support access to CMV diagnostics and antivirals. Additionally, ongoing R&D in novel antivirals, biologics, and preventive vaccines fuels market expansion globally.
Cytomegalovirus Infection Treatment And Diagnosis Market Development
In March 2025, Zydus launched ANVIMO (Letermovir) for the prevention of cytomegalovirus infection in haematopoietic stem cell and kidney transplant patients, available in 240 mg and 480 mg doses.
Key Players
Leading Companies of the Market
Gilead Sciences
F. Hoffmann-La Roche
GlaxoSmithKline
Merck & Co.
ViiV Healthcare
Pfizer
Key players in the Cytomegalovirus Infection Treatment and Diagnosis market include Gilead Sciences, F. Hoffmann-La Roche, GlaxoSmithKline, Merck & Co., ViiV Healthcare, Pfizer. These companies lead in antivirals, diagnostics, and emerging CMV therapies, driving innovation, market share, and global adoption of CMV management solutions.
Cytomegalovirus Infection Treatment And Diagnosis Market Future Outlook
The future outlook of the Cytomegalovirus Infection Treatment and Diagnosis market is highly promising, with strong growth expected through 2033. Increasing prevalence of immunocompromised patients, organ transplants, and congenital CMV cases will drive demand for effective diagnostics and antiviral therapies. Technological advancements, including rapid PCR testing, point-of-care diagnostics, and AI-enabled monitoring, will enhance early detection and patient management. Investment in novel antivirals, biologics, and vaccine development is anticipated to expand treatment options. Geographically, North America and Asia Pacific are poised for significant growth, supported by healthcare infrastructure, awareness campaigns, and proactive government initiatives, ensuring sustained market expansion.
Cytomegalovirus Infection Treatment And Diagnosis Market Historical Analysis
The Cytomegalovirus Infection Treatment and Diagnosis market historical analysis shows steady growth over the past decade, driven by increasing awareness of CMV-related complications in immunocompromised patients, transplant recipients, and newborns. Historically, antiviral therapies such as ganciclovir, valganciclovir, and foscarnet dominated treatment, while serological and PCR-based diagnostics became standard for early detection and monitoring. North America led the market due to advanced healthcare infrastructure, followed by Europe with high adoption of diagnostics and treatment protocols. Investments in R&D, gradual introduction of novel therapeutics, and growing screening initiatives historically shaped market dynamics and laid the foundation for current growth trends.
Sources
Primary Research Interviews:
Hospital & Clinic Administrators
Healthcare Professionals
Pharmaceutical & Biotech Executives
Diagnostic Laboratory Managers
Databases:
PubMed / Medline
ClinicalTrials.gov
World Health Organization
Journals:
Journal of Clinical Virology
Clinical Infectious Diseases
The Journal of Infectious Diseases
Transplantation
Newspapers:
The Wall Street Journal
Financial Times
The Guardian
The New York Times
Associations:
American Society for Microbiology (ASM)
Infectious Diseases Society of America (IDSA)
American Society of Transplantation (AST)
European Society of Clinical Microbiology and Infectious Diseases (ESCMID)
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients