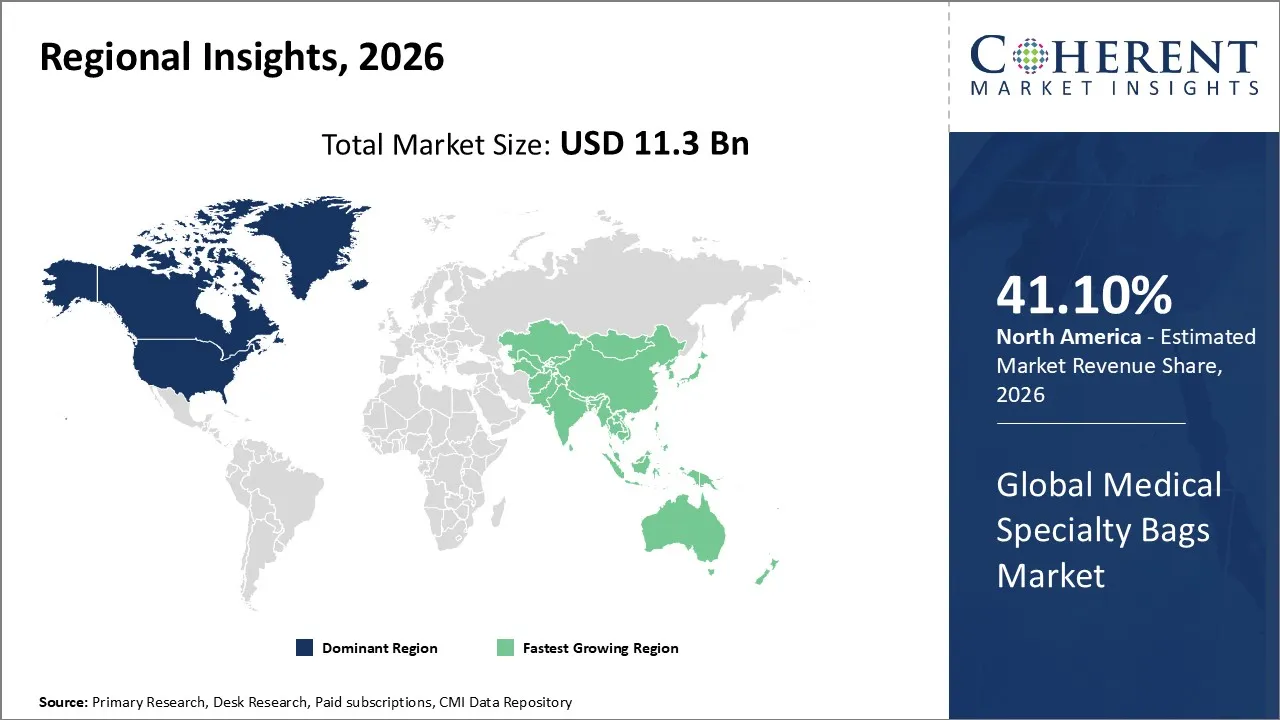
The Medical Specialty Bags Market is estimated to be valued at USD 11.3 Bn in 2026 and is expected to reach USD 14.7 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.3% from 2026 to 2033.
The Medical Specialty Bags Market serves healthcare by providing specialized bags that contain, collect, drain, and transport fluids safely in hospitals, clinics, and home care settings. It offers ostomy, urinary, IV fluid, blood, and other collection bags that ensure sterility and enhance patient care. Market growth results from the increasing prevalence of chronic diseases, a growing aging population, more surgical procedures, continuous improvements in bag materials and design, and a stronger emphasis on infection control and safety.
|
Current Events |
Description and its impact |
|
Geopolitical Developments Affecting Medical Trade and Supply Chains |
|
|
Technological Advancements in Medical Bag Materials |
|
|
Nanolevel Industry and Market-Specific Events |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Ostomy Collection Bags hold the largest market share of 36.8% in 2026. Rising cases of gastrointestinal disorders and cancers are increasing the number of patients undergoing colostomy, ileostomy, and urostomy procedures, driving demand for ostomy collection bags in the Medical Specialty Bags Market. Healthcare providers and patients actively adopt these bags due to growing awareness of ostomy care and improvements in design that enhance comfort, prevent leaks, and control odor. The expanding elderly population, improved healthcare access, and emphasis on home care further encourage consistent use of high-quality ostomy bags, fueling market growth. For instance, in June 2025, Northern Ireland’s Eakin Healthcare launched the world’s largest ostomy bag production line in Cardiff, investing £9 million as part of a £60 million expansion.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 41.10% in 2026. In North America, healthcare providers and patients actively drive demand for advanced fluid containment and drainage solutions, such as IV, ostomy, and urinary bags, supported by a well‑developed healthcare infrastructure and strict regulatory standards. They increasingly choose single‑use, high-quality specialty bags to improve safety and minimize infection risks, while continuous innovations in materials and design enhance usability. The region’s rising chronic disease burden, expanding home healthcare services, and supportive reimbursement policies further encourage widespread adoption across hospitals, clinics, and outpatient care settings.
In the Asia Pacific region, hospitals, clinics, and home healthcare providers are actively increasing demand for medical specialty bags used in surgeries, infusion therapy, and chronic care, driven by expanding healthcare infrastructure in China, India, and Southeast Asia. Providers are adopting sterile, single‑use solutions more widely as awareness of hygiene and safety rises. The growing number of patients with chronic conditions and the expanding home healthcare sector are prompting local manufacturers to produce and distribute specialty medical bags more broadly across diverse care settings.
In the United States, hospitals, clinics, and outpatient providers are actively increasing their use of advanced medical specialty bags for fluid management, waste collection, and sterile transport as they expand services. Healthcare facilities are adopting high‑quality, single‑use solutions to enhance patient safety, while continuous improvements in bag materials and design address diverse clinical requirements. Regulatory compliance and supportive reimbursement policies further motivate providers to implement these specialty products more widely, reinforcing their growing presence across the country’s healthcare system.
In China, hospitals, clinics, and healthcare providers are actively increasing demand for medical specialty bags as the country expands healthcare infrastructure and improves access to quality care. Government programs aimed at enhancing patient outcomes and promoting modern medical technologies are prompting manufacturers to design and launch bags with advanced materials and features. Rising individual healthcare spending and growing awareness of hygiene and safety are encouraging providers to adopt sterile, single‑use specialty bags widely across hospitals, clinics, and home care settings. For instance, Gelesis partnered with China Medical System Holdings (CMS) to commercialize Plenity in China, leveraging CMS’s expertise in sales and marketing of specialty pharma products.
Manufacturers are increasingly focusing on developing medical specialty bags using lightweight, durable, and biocompatible materials. Innovations such as leak-proof adhesives, odor control layers, and flexible yet strong polymers enhance patient comfort and usability. This trend addresses patient demands for safer, more ergonomic bags while meeting stringent hospital standards. Improved designs also facilitate handling, reduce waste, and support both hospital and home care environments, driving broader adoption across diverse healthcare settings.
The expansion of home healthcare services is reshaping demand for specialty bags. Patients increasingly rely on portable, easy-to-use bags for IV fluids, ostomy care, and urinary collection, allowing safe management outside hospitals. Providers are offering solutions tailored for home use, emphasizing simplicity, hygiene, and reliability. This shift not only reduces hospital stays but also encourages manufacturers to innovate products that support patient independence and continuous monitoring in home-based treatment programs.
Developing specialty bags with enhanced comfort, odor control, skin-friendly adhesives, and modular designs presents growth potential. Incorporating technology, such as smart monitoring systems or easy‑to-use home care kits, can differentiate products and increase adoption. Continuous innovation that aligns with patient needs, ergonomic designs, and safety standards allows companies to capture more market share while improving compliance, usability, and patient satisfaction across ostomy, urinary, IV, and drainage bag segments.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 11.3 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.3% | 2033 Value Projection: | USD 14.7 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Braun Melsungen AG, Baxter International, Inc., Coloplast A/S, ConvaTec, Inc , Hollister Incorporated, Medline Industries Inc , Nolato AB, BD, Macopharma SA, Terumo Corporation, Smiths Medical, Fresenius SE & Co. KGaA, HLL Lifecare Limited, Ostique, Grifols, and AdaptHealth LLC |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients