Severe Acute Respiratory Syndrome (SARS) Treatment Market is estimated to be valued at USD 131.30 Bn in 2026 and is expected to reach USD 177.4 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.4% from 2026 to 2033.
The Severe Acute Respiratory Syndrome (SARS) treatment market is witnessing strong growth, driven by rising demand for antiviral therapies, supportive care, and innovative drug classes. Advancements in nucleoside analogues, protease inhibitors, and immunomodulatory approaches are enhancing treatment effectiveness. Increasing global healthcare investment, heightened preparedness for respiratory outbreaks, and ongoing research collaborations are expected to sustain momentum and expand therapeutic options in the coming years.
|
Current Event |
Description and its Impact |
|
China's Healthcare Policy Reforms and Respiratory Disease Preparedness |
|
|
Global Supply Chain Disruptions and Pharmaceutical Manufacturing |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of drug class, the antivirals segment is expected to hold 40% share of the market in 2026, due to their direct action against viral replication. In clinical settings, drugs like Remdesivir, Lopinavir/Ritonavir, and Ribavirin are extremely widespread. Since they work to lower viral load and improve patient outcomes, they are the most important part of SARS treatment, especially for SARS-CoV2 cases in 2026.
For instance, in September 2025, Shionogi announced that the FDA had accepted its New Drug Application for Ensitrelvir. This is the first oral drug that can help prevent COVID-19 after exposure. This milestone marks an important advancement forward in antiviral treatment. It gives people more options for preventing SARS-CoV-2 and makes the world more ready for respiratory outbreaks by making therapeutic solutions more accessible and easier to use.
In terms of indication, the SARS‑CoV2 segment is expected to lead with 65% share of the market in 2026, driving most treatment demand. Since there are so many COVID-related SARS cases around the world, researchers intend to continue working on treatments that work against this strain. Ongoing mutations and variants make it even more important to have effective antiviral and monoclonal antibody treatments. This is expected to maintain SARS-CoV2 at the top of the market in 2026.
For instance, in December 2025, the WHO started a global plan to help countries deal with coronaviruses, such as SARS-CoV2, which causes COVID-19. This program improves surveillance, preparedness, and treatment strategies, which directly helps the Severe Acute Respiratory Syndrome (SARS) Treatment Market by making the world more resilient and making sure that future outbreaks are dealt with quickly.
In terms of route of administration, the intravenous segment is projected to account for 55% share in 2026, owing to severe SARS patients require rapid drug delivery in hospital settings. IV antivirals, corticosteroids, and monoclonal antibodies work right away to help. This is the best way to give critical care as it allows for controlled dosing and faster absorption. It will be the main way to give it in 2026.
In terms of distribution channel, the hospital pharmacies segment is projected to capture 50% share of the market in 2026, reflecting the acute nature of SARS treatment. Most patients are treated in hospitals, where they can get antivirals, corticosteroids, and monoclonal antibodies right away. In 2026, hospital pharmacies are going to be the most important way for people to get their medications due to they have reliable supply chains, strict monitoring, and drugs that are always available.
To learn more about this report, Download Free Sample
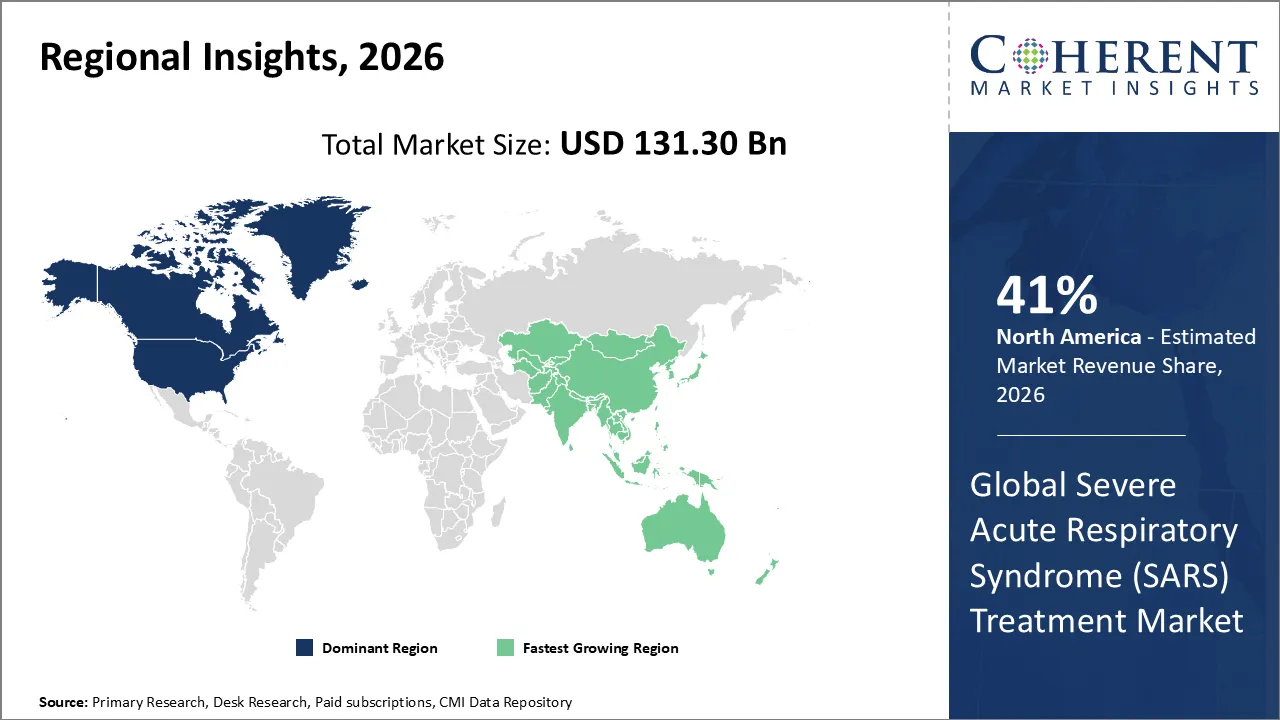
North America is dominating the Severe Acute Respiratory Syndrome (SARS) treatment market with 41% share in 2026, due to they have strong healthcare systems, well-prepared governments, and quick adoption of new treatments. North America has a lot of pharmaceutical companies and biotech innovators, and there exists a large number of demands for monoclonal antibodies and antivirals. The government is also stockpiling a significant number of these drugs and getting ready for a pandemic. In light of this, the region has the largest market share in the world, setting standards for treatment protocols and regulatory approvals that affect the whole industry.
For instance, in December 2025, Atea Pharmaceuticals finished signing up patients in North America for bemnifosbuvir, an oral antiviral that targets SARS-CoV-2. This milestone makes the Severe Acute Respiratory Syndrome (SARS) Treatment Market opportunity stronger by adding more treatment options, helping to prepare for pandemics, and making North America a key area for developing global antiviral strategies.
Asia Pacific is anticipated to be the fastest growing region. An increasing number of patients are at risk for respiratory infections, the healthcare system is getting larger, the government is putting an extensive amount of revenue into being ready for a pandemic, and the pharmaceutical industry is getting larger as more clinical trials are happening. In spite of this, Asia-Pacific is likely to grow faster than other areas, becoming an important center for low-cost treatments and new ideas.
For instance, in September 2025, Mongolia continues to move forward with plans for a Respiratory Infections Surveillance Collaboratory to improve monitoring, early detection, and readiness for respiratory diseases. With help from WHO, the initiative will bring together health authorities, researchers, and partners to make people more resilient, keep an eye on viruses like influenza, RSV, and coronaviruses, and improve the ability of public health to respond.
In 2026, the U.S. is going to require more Severe Acute Respiratory Syndrome (SARS) treatment to address better healthcare infrastructure, more research and development in pharmaceuticals, being ready for new respiratory threats, and putting money into antivirals and biologics. The U.S. is a major demand center due to rising awareness, government programs, and lessons learned from the pandemic.
For instance, in April 2025, Researchers at the University of Arizona Health Sciences started a business to offer new ways to treat asthma and COPD. The company wants to turn research into real-world care by building on what it learns in the lab. It wants to improve treatments for chronic respiratory diseases and find new ways to deal with major global health threats by focusing on patients.
China's need for Severe Acute Respiratory Syndrome (SARS) treatment in 2026 is due to its large population, rising healthcare spending, and readiness for respiratory threats. China is a key growth market in Asia for antivirals and biologics due to government programs, the growth of the pharmaceutical industry, and lessons learned from past outbreaks.
For instance, in July 2025, GSK and China's Hengrui Pharma made deals to work together to make new medicines for respiratory, immunology, and cancer. GSK is paying $500 million up front for HRS-9821, a PDE3/4 inhibitor for COPD, and up to $12 billion in milestones. This will strengthen respiratory pipelines around the world, especially in Europe and Asia.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 131.30 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.4% | 2033 Value Projection: | USD 177.4 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Gilead Sciences Inc., Hetero labs Ltd., Cipla Ltd., Cadila healthcare Ltd., Ipca laboratories, GlaxoSmithKline plc., Novartis, Pfizer Inc., Roche Holding AG, AbbVie, Takeda Ltd., Johnson & Johnson, Boehringer Ingelheim, Eli Lily & Co., and AstraZeneca plc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The growing prevalence of SARS‑CoV2 continues to be the primary driver of the Severe Acute Respiratory Syndrome (SARS) Treatment Market size. Infections are on the rise, and new variants are appearing. Therefore, antivirals, monoclonal antibodies, and supportive therapies are going to be in high demand. This should result in steady growth in the market and better global healthcare readiness through 2026.
Advancements in antiviral and biologic therapies are significantly boosting the Severe Acute Respiratory Syndrome (SARS) Treatment Market share. New drugs like Remdesivir, Lopinavir/Ritonavir, and monoclonal antibodies like REGEN COV and Sotrovimab make treatments more effective. These new ideas improve clinical outcomes, give more treatment options, and keep the global market growing.
Global preparedness investments are a crucial driver in the Severe Acute Respiratory Syndrome (SARS) Treatment Market forecast. Governments and healthcare systems all over the world are setting aside money for pandemic readiness, building up hospital infrastructure, and stockpiling antivirals. These programs make treatment providers more resilient, make sure they can respond quickly, and open up long-term growth opportunities.
Diversified routes of administration are fueling Severe Acute Respiratory Syndrome (SARS) Treatment Market demand. Intravenous delivery remains very important for severe cases, but the rise of oral antivirals and new inhalation therapies makes treatment more accessible, easier for patients to follow, and more flexible. This expansion makes it easier for healthcare systems all over the world to adopt it and helps them keep growing.
The Severe Acute Respiratory Syndrome (SARS) treatment market value is getting increasing attention from drug companies and healthcare systems. This is since there is still a need to deal with SARS-related coronaviruses and get ready for future respiratory outbreaks. There remain many different ways to treat patients, including antiviral agents, corticosteroids, monoclonal antibodies, and supportive care like oxygen management. This demonstrates that there are many different ways to treat patients, with targeted drugs and symptom control working together.
Antiviral therapies are an important component of treatment plans, and research and development continues to go on to find ways to stop viruses from replicating. Immunotherapies like monoclonal antibodies are becoming more popular as they may be able to neutralize virus particles and lower the viral load in patients who are at high risk. Corticosteroids, on the other hand, are commonly used to treat inflammation in severe respiratory cases.
North America always leads in the adoption of new treatments and therapeutic innovations, due to its advanced healthcare infrastructure and large investments in research. Europe is not far behind, with a lot of public health programs and clinical trials going on. The Asia Pacific region is becoming a lively place for SARS therapy research as healthcare funding is going up and clinical capabilities are growing.
Pharmaceutical companies, research institutions, and government agencies are working together to shape the development pipeline. They are using new technologies like broad-spectrum antiviral agents, mRNA platforms, and artificial intelligence-driven drug discovery models to speed up the search for promising therapeutic candidates.
Overall, the SARS treatment market's path shows that people are still putting capital into both established and new types of treatments. This is since they want to meet short-term clinical needs while also planning for the long term.
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients