Global Acne Medication Market Size and Forecast: 2026-2033
The global acne medication market is expected to grow from USD 9,027.9 Mn in 2026 to USD 12,658.5 Mn by 2033, registering a compound annual growth rate (CAGR) of 4.9% from 2026 to 2033. The global acne medication market is poised for significant expansion, fueled by the soaring prevalence of acne, particularly among adolescents and young adults.
According to Global Burden of Disease (GBD) estimates, acne affects approximately 9.4% of the global population, making it one of the most prevalent skin disorders worldwide. Additionally, as per the data published by the National Library of Medicine, acne vulgaris is commonly observed in adolescents and young adults. Its prevalence rates is estimated to range from 35% to over 90% among adolescents.
(Source: MDPI, National Library of Medicine)
Key Takeaways of the Global Acne Medication Market
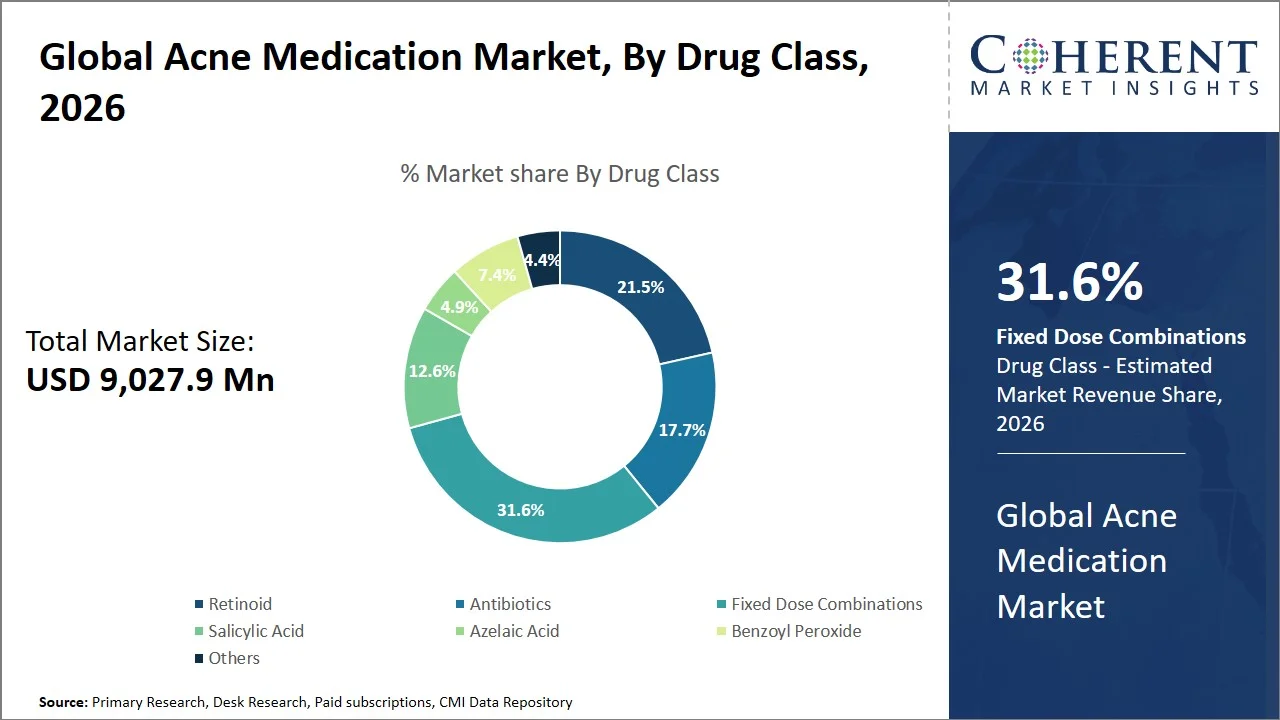
- Fixed dose combinations are projected to hold 31.6% of the global acne medication market share in 2026, making it the dominant drug class segment, with high penetration in North America and Europe attributed to high clinical acceptance towards multi-mechanism approaches, and regulatory support for combination products. For instance, the Cabtreo approved by the U.S. Food and Drug Administration represents growing use of triple combination therapy in the U.S., improving treatment effectiveness and compliance. (Source: U.S. Food and Drug Administration) Similarly, combination products like Epiduo (adapalene and benzoyl peroxide) have broad regulatory approval within the EU based on European Medicines Agency (EMA) guidelines, which further strengthens the high acceptance of fixed-dose products in dermatology practices. (Source: European Medicines Agency)
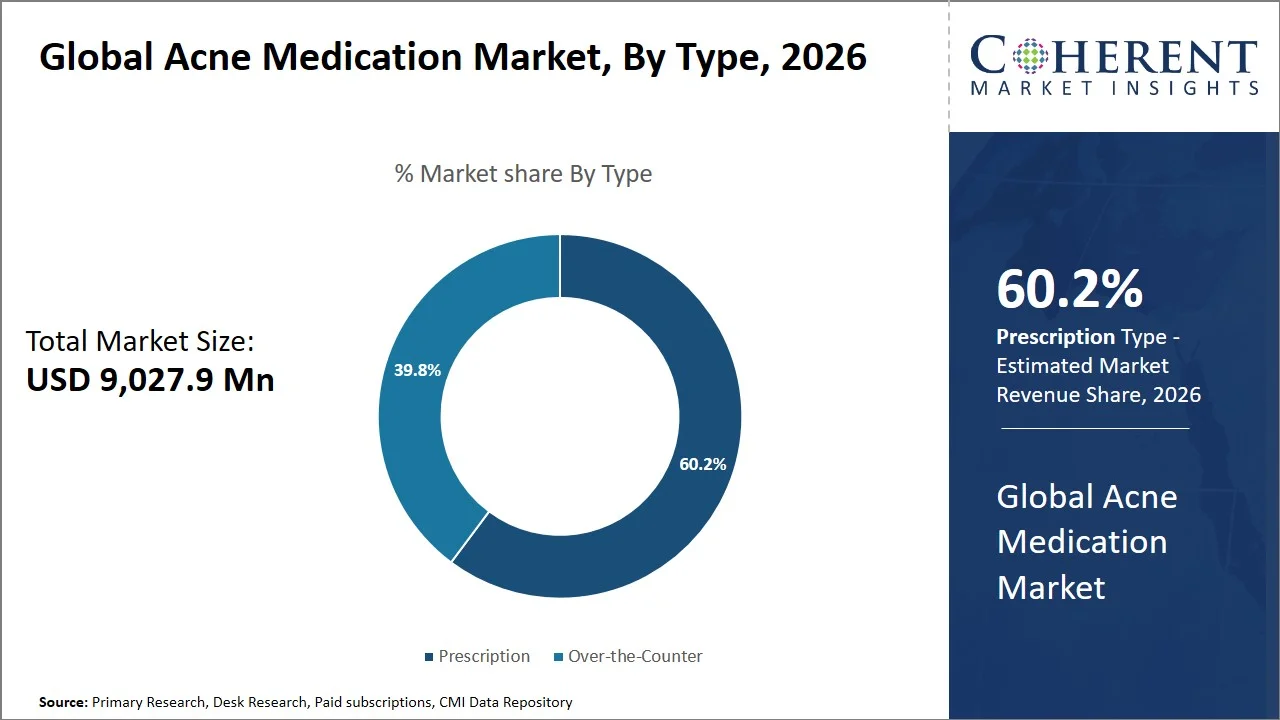
- Prescription is projected to hold 60.2% of the global acne medication market share in 2026, making it the dominant type segment, in conjunction with North America, especially the U.S., leading because of rigorous regulation and high adherence to dermatological treatment protocols. For instance, Isotretinoin is distributed through the iPLEDGE program, which is a required risk evaluation and mitigation strategy (REMS) in the U.S. The iPLEDGE program entails enrollment, certification for the prescribing physician and the pharmacist, and constant monitoring. Monitoring may involve the use of pregnancy testing and controlled distribution periods. As such, the stringent and well-regulated access route to isotretinoin makes it less likely to be sold over-the-counter. (Source: iPLEDGE)
- Inflammatory acne is projected to hold 62.6% of the global acne medication market share in 2026, making it the dominant indication segment, within the Asia Pacific region, particularly China and South Korea, takes precedence owing to a greater incidence of severe acne with the need for anti-inflammatory treatment. Treatment protocols in these countries emphasize a combined treatment approach that addresses inflammation and bacterial growth, resulting in increased medication use. For instance, recommendations from the South-East Asia dermatology guidelines state that the combination of topical retinoids with benzoyl peroxide and/or antibiotics are used to treat moderate inflammatory acne, reinforcing strong clinical alignment toward combination-based therapy in APAC acne medication market. (Source: ResearchGate)
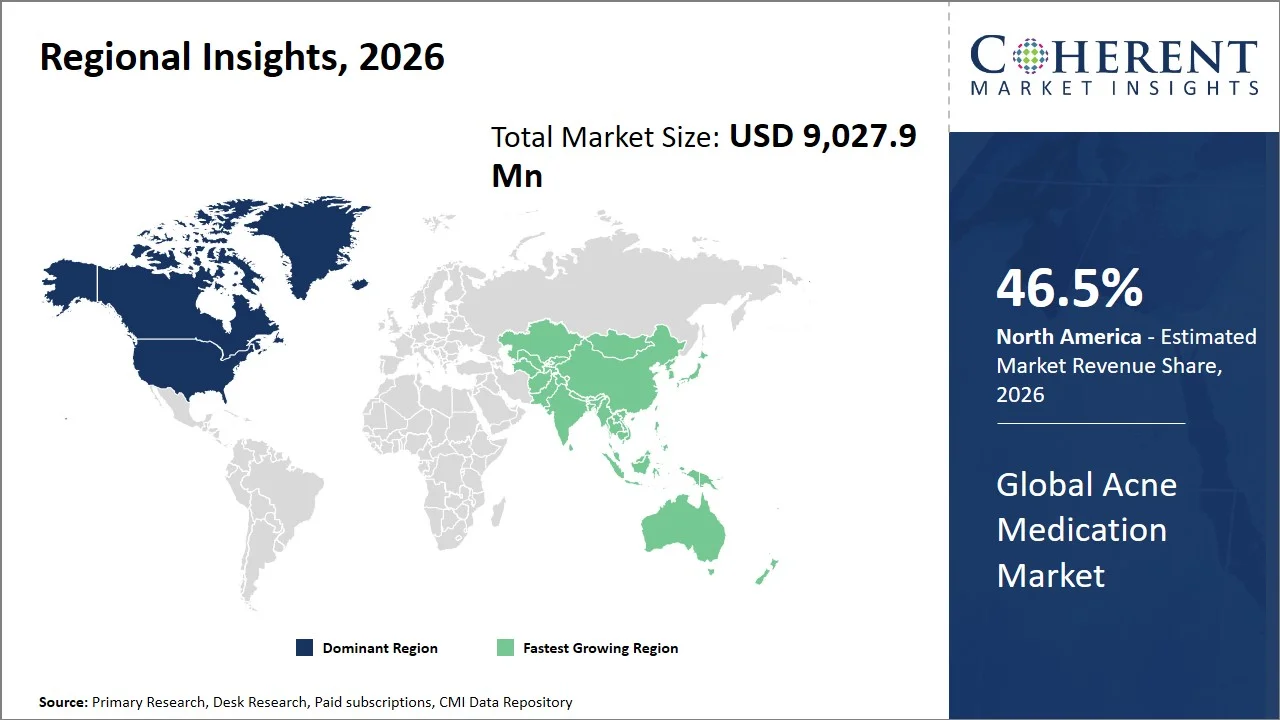
- North America maintains dominance with an expected share of 46.5%in 2026, propelled by rising instances of personalized dermatology and integrated care models where digital health plays an instrumental role in ensuring higher patient compliance and participation. There is an increasing trend of AI-powered teledermatology platforms being used in the region to diagnose and make prescriptions more accessible. For instance, development of technologies like DermTech have led to the availability of non-invasive testing for skin genomics, aiding in acne diagnosis and management, enabling more targeted treatment decisions and strengthening dermatology care delivery across the U.S. (Source: DermTech 2.0.)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 21.0% share in 2026, driven by growth in dermatological drug manufacturing and the rising number of regulatory approvals for acne therapies in emerging markets. Nations like China and India are seeing a surge in the local production of generic and branded dermatological drugs, making them more affordable and accessible. For instance, Galderma has announced its plan to expand their manufacturing and innovation presence in Asia Pacific by assessing the possibility of setting up a new plant in Singapore. This was geared toward boosting their manufacturing capacity and minimizing dependence on their supply chain, enabling faster market access across the Asia Pacific region. (Source: Galderma)
- Expansion of Hormonal Acne Therapies in Adult Female Segment: A large number of adult females are experiencing persistent or late-onset acne linked to hormonal imbalances. Such a condition results in a need for specific treatments to cater to the market demand. Oral contraceptives and anti-androgenic drugs are becoming more prevalent in markets such as the U.S. and Japan. This trend encourages pharmaceutical firms to invest in developing gender-specific products that can treat acne. There is tremendous room for growth due to high treatment adherence and recurring therapy needs.
- Rising Demand for Microbiome-Friendly and Non-Antibiotic Treatments: The threat of antibiotic resistance has sparked the development of alternative approaches to treat acne that protect the skin’s microbiome. Bacteriophage-based therapies, probiotics, and actives which target sebum production are among the innovations receiving attention, especially in Europe. The increasing regulatory push to use fewer antibiotics has contributed to the emergence of alternatives. There is significant business opportunities for firms focused on the microbiome into a high-value, differentiation-driven niche.
Why Does the Fixed Dose Combinations Segment Dominate the Global Acne Medication Market?
Fixed dose combinations are projected to hold a market share of 31.6% in 2026, due to their ability to achieve synergy without adding more complexity to the drug regime, and therefore enhancing effectiveness and compliance, which are key characteristics of effective acne treatment management. By combining agents with complementary mechanisms of action (antibacterial + keratolytic), such products have been able to reduce the number of drugs required and prevent issues related to antibiotic resistance with the inclusion of benzoyl peroxide. The use of fixed-dose combinations for treating acne is clear from the ongoing product development and lifecycle management activities conducted by leading companies in the market.
For instance, Epiduo Forte by Galderma, is an advanced fixed-dose combination aimed at addressing cases of moderate to severe acne, available across the U.S. and Europe, emphasizing sustained demand for optimized combination therapies. (Source: Galderma) Further, continuous clinical development projects, including IDP-126 (before commercialization), showed statistically better results of lesion reduction compared to dual therapy in Phase III studies, which emphasizes the transition within the industry to more powerful combinations of multiple agents. (Source: ClinicalTrials.gov) Collectively, these developments emphasize why fixed-dose combinations continue to exceed monotherapy in both prescribing preference and market share.
Why does Prescription Represent the Largest Type Segment in the Acne Medication Market?
To learn more about this report, Request Free Sample
Prescription is projected to hold a market share of 60.2% in 2026, since they are indispensable in the treatment of moderate to severe and chronic cases of acne as the over-the-counter medications fail to be effective enough in such cases. The prescription drugs feature well-proven clinical effectiveness as they target several paths of acne development, such as the regulation of sebum production, antibacterial, and anti-inflammatory effect. For instance, in October 2024, a study published by ResearchGate, investigating topical retinoids found that 83% of the participants suffering from acne exhibited clinical improvement. This further emphasizes the ongoing importance of prescription medications, particularly for patients requiring sustained and clinically supervised treatment. (Source: ResearchGate)
The segment’s dominance is further buttressed by the constant development of innovative, prescription-based medications having distinct mechanisms of action. For instance, Winlevi, approved by U.S. Food and Drug Administration, is the first of its kind to be a topical androgen receptor inhibitor used in the treatment of acne. It presents itself as an alternative method to the currently used conventional retinoids and antibiotics and, as an Rx-only drug, has seen great adoption in clinical use, strengthening the reliance on prescription treatments in the acne medication market. (Source: Food and Drug Administration)
Inflammatory Acne Segment Dominates the Global Acne Medication Market
The inflammatory acne segment is projected to hold a market share of 62.6% in 2026, since this category signifies the most important type of the ailment in terms of its clinical significance and treatment requirements. In contrast to the comedonal type of acne, inflammatory lesions are characterized by the presence of bacteria and immune-mediated reactions, necessitating pharmaceutical interventions to avoid scarring, propelling surging demand for prescription and combination therapies. For instance, a clinical study published by Dovepress, revealed that combination therapy (retinoids, benzoyl peroxide, and antibiotics) effectively decreased the number of inflammatory lesions and the severity of acne in 12 weeks, outperforming monotherapies. (Source: Dovepress)
Similarly, clinical reviews published by CosmoDerma, highlighted that combination therapy can result in 60-65% reduction in inflammatory lesions during the course of treatment, emphasizing their significance in the management of moderate to severe inflammatory acne. (Source: CosmoDerma) These results clearly indicate that the high clinical burden and high treatment intensity involved in inflammatory acne contribute significantly to increased drug demand, making it the leading indication segment in the global acne medication market.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Industry-wide Alert on Benzene Contamination Risk in Drug Products (July 2025) |
|
|
U.S. FDA Benzoyl Peroxide Safety Review & Acne Product Recalls (March 2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Food and Drug Administration, Food and Drug Administration,)
Global Acne Medication Market Dynamics
To learn more about this report, Request Free Sample
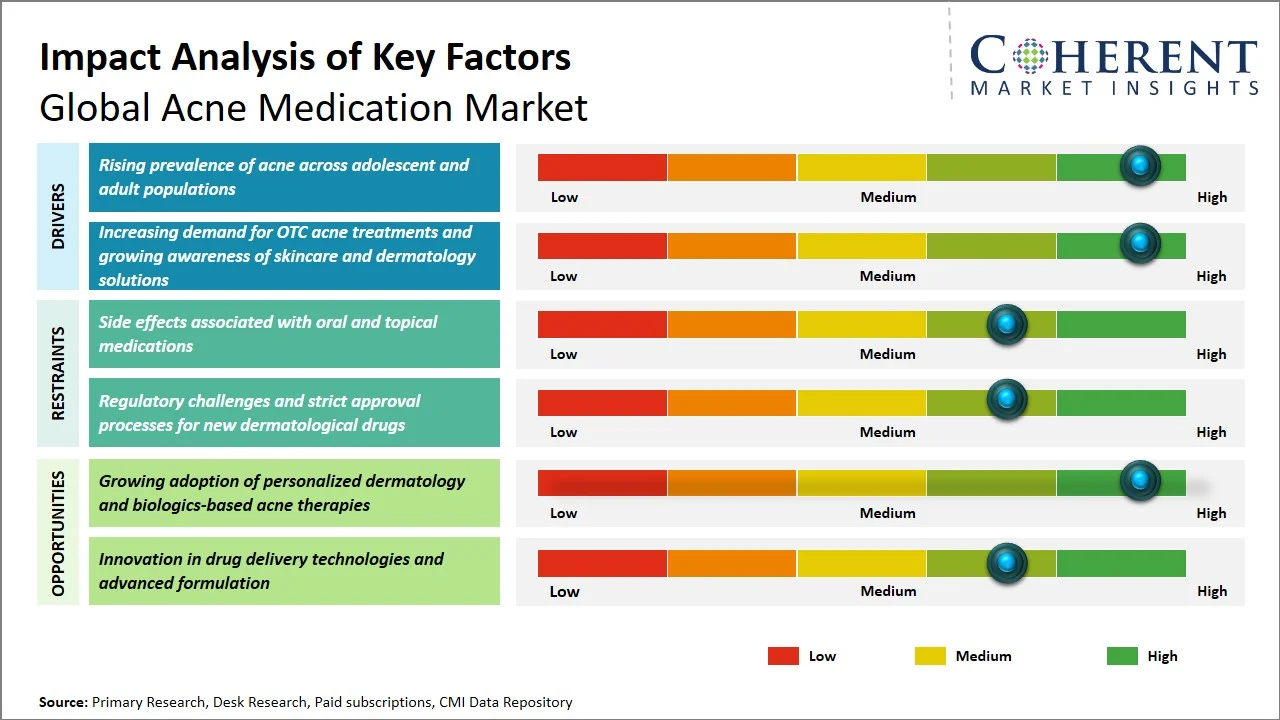
Market Drivers
- Rising prevalence of acne across adolescent and adult populations: The increasing prevalence of acne among adolescents and adults alike is one of the major factors fueling the growth of the global acne medication market since the condition is no longer limited to teenage years but now affects adults as well due to hormonal disorders, environmental pollution, and other external factors. As a result, the number of patients suffering from acne is constantly growing, thus creating a continuous demand for topical and systemic treatments across regions. For instance, according to American Academy of Dermatology Association, acne is the most common skin condition in the U.S., affecting up to 50 million Americans annually. It further states that, acne usually begins in puberty and affects many adolescents and young adults however, it can occur at any stage of life and may continue into one’s 30s and 40s. Adult acne continues to increase and affects up to 15% of women. Additionally, approximately 85% of people between the ages of 12 and 24 experience at least minor acne. (Source: American Academy of Dermatology Association) These trends highlight a widening and aging pool of patients that continuously drives up demand for treatment and stimulates market growth.
- Increasing demand for OTC acne treatments and growing awareness of skincare and dermatology solutions: Nowadays, consumers are opting for cost-effective products that can be easily purchased from pharmacies and require minimal professional assistance, such as those used in treating mild to moderate cases of acne. Over-the-counter products include cleansers containing benzoyl peroxide, salicylic acid products, and retinoids gels, which are increasingly being made available due to the rapid expansion of pharmacy chains and the rise of direct-to-consumer skincare brand. Moreover, consumer interest in skin care, efficacy of ingredients in skin care products, and early intervention, amplified through digital platforms and dermatology-backed education initiatives. For instance, U.S. Food and Drug Administration approval of adapalene (Differin Gel 0.1%), an over-the-counter drug, was a critical step in the evolution that led to formerly prescription-only retinoids becoming available to consumers, substantially expanding the OTC treatment base. (Source: Food and Drug Administration) Moreover, organizations such as the American Academy of Dermatology have continued their efforts towards educating the general public regarding proper treatment and usage of products to effectively manage their acne issues, thus further promoting the use of OTC products by consumers at an earlier stage.
Emerging Trends
- Growth of Combination Drug-Device Therapies in Acne Management: There has been increased synergy between topical/ingestible treatments and energy-based devices such as lasers, light, and radiofrequency treatments in order to increase treatment outcomes, particularly in the case of moderate-to-severe acne. This hybrid approach enhances treatment effectiveness, lowers recurrence rates, and lessens drug dependency, becoming increasingly popular in dermatological practices throughout North America and Europe.
- Rising Focus on Scar Prevention and Post-Acne Treatment Solutions: There is a growing trend towards transitioning from active treatment of acne to early intervention to avoid any scarring and prevent post-inflammatory hyperpigmentation. The companies are developing their product portfolio by introducing complementary products like retinoids and chemicals that address future skin problems, particularly in the Asia Pacific region.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Acne Medication?
North America leads the global acne medication market, accounting for an estimated 46.5% share in 2026, due to the existence of a sophisticated dermatology industry, and continuous emergence of novel therapeutic approvals, resulting in rapid adoption of advanced treatments. Moreover, the region has strong clinical research infrastructure and early adoption of novel treatments that accelerate its entry into the market as opposed to other regions.
For instance, Seysara, an oral antibacterial that has a narrow spectrum of activity and is formulated particularly for acne, was approved by the U.S. Food and Drug Administration and has been extensively used in the U.S. because of its selective activity and enhanced safety. (Source: Food and Drug Administration) Furthermore, high availability of specialized dermatological clinics (such as U.S. Dermatology Partners, Forefront Dermatology, Schweiger Dermatology Group, Epiphany Dermatology, and Advanced Dermatology and Cosmetic Surgery) coupled with the provision of insured services to patients makes this region a major leader in the acne medication market.
Why Does the Asia Pacific Acne Medication Market Exhibit High Growth?
The Asia Pacific acne medication market is expected to exhibit the fastest growth with an estimated contribution of 21.0% share to the market in 2026, attributed to the fast development of local pharmaceutical manufacturing and regulatory approvals, as well as improved dermatology services in emerging economies. The countries like China and India are developing their own systems of drug manufacturing and speeding up their approvals for dermatology drugs, thus providing wider availability of effective and affordable acne medicines.
In addition, there have been increased investments in health care facilities and dermatology services in emerging economies, contributing to better diagnostics and treatment. For instance, the Ministry of Health and Family Welfare has been scaling up health service provision through programs such as the Pradhan Mantri Ayushman Bharat Health Infrastructure Mission (PM-ABHIM), which is aimed at building infrastructure for primary healthcare services, diagnostics, and specialized services, thus increasing availability of dermatology services. (Source: Ministry of Health and Family Welfare) These trends illustrate that local manufacturing combined with increased treatment accessibility are jointly contributing to significant growth momentum in the Asia Pacific acne medication market.
Global Acne Medication Market Outlook for Key Countries
Why is the U.S. Leading Innovation and Adoption in the Acne Medication Market?
The U.S. has the potential for leadership in innovation and adoption in the field of acne medication due to its well-established research and development ecosystem coupled with robust support from regulatory framework headed by the U.S. Food and Drug Administration. Additionally, the presence of a large number of global pharmaceutical and biotechnology firms (such as Johnson & Johnson, Pfizer, AbbVie, and Bausch Health Companies) within the country accelerates product development and clinical advancements.
Furthermore, the presence of established dermatology infrastructure and the timely availability of both prescribed and over-the-counter products contribute to the quick adoption process. Besides, high consumers awareness as well as their willingness to invest in skincare products reinforce the U.S. as a key innovation hub in acne treatment.
Is Japan a Favorable Market for Acne Medication?
Japan emerges as a favorable market for acne medication owing to the country’s sophisticated healthcare system and high priority on dermatological services. The existence of a well-defined regulatory framework under the Pharmaceuticals and Medical Devices Agency guarantees high-quality and safe product approvals, which fosters market stability. Furthermore, the heightened consumer consciousness regarding skincare and the culture’s strong personal grooming habits ensures the constant consumption of both prescription-based and OTC treatments for acne. Moreover, the existence of prominent local companies, such as Shiseido and Rohto Pharmaceutical, strengthens innovation and product availability.
Is China Emerging as a Key Growth Hub for the Acne Medication Market?
The acne medication market in China is currently experiencing significant development attributed to the substantial number of adolescent and young adult who suffer acne. The recent modifications in policy by the National Medical Products Administration (such as priority review pathways, acceptance of overseas clinical trial data, streamlined IND/NDA approval timelines, and the Marketing Authorization Holder (MAH) system) are facilitating faster approval processes and speeding up the availability of new dermatological medications. Moreover, the emergence of numerous virtual healthcare platforms and internet pharmacies (such as Ping An Good Doctor, JD Health, AliHealth, and internet pharmacies like JD Pharmacy and 1 Pharmacy) is changing the face of the China acne medication market emerging as a key growth hub.
Why Does Germany Top the Europe Acne Medication Market?
Germany is the leader in the Europe market for acne medication due to its solid and efficient clinical dermatology infrastructure and consultations, hence creating a greater need for prescription drug treatments. There is a well-designed reimbursement framework (such as Statutory Health Insurance under Gesetzliche Krankenversicherung (GKV)), which guarantees the access to prescribed medications that have been clinically certified, not just over-the-counter medicines.
Moreover, there are high-quality standards imposed on medications by the Federal Institute for Drugs and Medical Devices, resulting in high consumer confidence in such drugs. Moreover, the increasing health consciousness among an aging population leads to growing interest in sustainable skin management solutions.
Is the Acne Medication Market Developing in India?
The market for acne medication is rapidly growing in India owing to its transition towards treatment-based skincare from cosmetic skin care, particularly among the youth suffering from skin conditions due to pollution and lifestyle issues. In terms of availability and affordability, acne medication in country is more accessible compared to many developed economies. The unique aspect of the India's acne medication market is that there is considerable similarity between drugs used for the treatment of acne and derma cosmetics, hence patients use both in combination. Quality is ensured through oversight from the Central Drugs Standard Control Organization, although the increase in online consultation and rapid commerce pharmacies quietly transforms the way people obtain treatments for acne.
|
Region
|
Key Regulatory Bodies |
Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (FDA); Health Canada |
U.S.: FD&C Act; OTC Acne Monograph (21 CFR Part 333); REMS (iPLEDGE) Canada: Food and Drugs Act; Natural Health Products Regulations (for OTC acne products); Drug Identification Number (DIN) system |
In the U.S., stringent OTC monographs and REMS programs ensure controlled access and high safety compliance for acne drugs. In Canada, products must obtain a DIN or NPN (Natural Product Number) before commercialization, with Health Canada enforcing strict labeling, safety, and efficacy standards. This results in high regulatory credibility but longer approval timelines across the region. |
|
Europe |
European Medicines Agency (EMA) |
EU Directive 2001/83/EC; EudraLex; centralized & decentralized drug approval procedures |
Harmonized regulatory system across EU ensures consistent product standards, but requires extensive clinical data and pharmacovigilance, increasing time-to-market |
|
Asia Pacific |
National Medical Products Administration (China), Pharmaceuticals and Medical Devices Agency (Japan), CDSCO (India) |
Country-specific drug approval frameworks; classification differences between drugs and cosmetics |
Regulatory fragmentation leads to varied approval timelines and product classification challenges, especially for OTC vs prescription acne treatments |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico) |
National drug approval frameworks; cosmetic vs drug classification differences |
Regulatory processes are evolving, with increasing alignment to global standards, but still involve long approval cycles and documentation requirements |
|
Middle East & Africa |
Saudi FDA, South African Health Products Regulatory Authority (SAHPRA) |
Import licensing systems; reliance on international approvals (FDA/EMA) |
Heavy reliance on external approvals results in delayed product entry but lower local R&D burden |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is growing adoption of personalized dermatology and biologics-based acne therapies creating new growth opportunities in the global acne medication market?
Rising personalization of dermatology and use of biological products for the treatment of acne is opening up new growth prospects in the acne medication market by shifting the approach from "one-size-fits-all" to precision medicines. With the advent of AI-based diagnostics of skin conditions, biomarkers discovery, and genome profiling, personalized treatment protocols are being adopted, which will improve efficacy and compliance. (Source: Biores Scientia)
In addition to this, emerging biological drugs and treatments such as cytokines inhibitors and bimekizumab (under development for severe acne) are targeting therapy-resistant cases based on action on selective inflammatory pathways. (Source: Dermatology Times) Moreover, the companies are investing on developing the next generation of biological treatments, such as those based on microbiome (live skin microbiome therapeutics such as SBM-01 by S-Biomedic, currently in clinical evaluation) and Sanofi's pipeline development in dermatology through clinical research studies. (Source: S-BIOMEDIC, Sanofi) The fusion of precision diagnostics and targeted therapeutics are generating opportunities for premium treatment categories that can offer highly valuable acne treatments.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On April 27, 2026, Sagimet Biosciences Inc. revealed plans for commencing a Phase 3 clinical trial of denifanstat in moderate to severe cases of acne in the U.S. during the second half of 2026. The company is also progressing its FASN inhibitor pipeline (TVB-3567) with ongoing early-stage trials.
- In November 2025, Cosmo Pharmaceuticals revealed that its proprietary product, Winlevi (clascoterone cream), was approved for use in Brazil, indicating an important milestone in the expansion of its market share within its novel class of anti-acne therapy. This represents a major breakthrough for Cosmo Pharmaceuticals in Latin America and is a positive move towards worldwide commercialization of this drug.
Competitive Landscape
The global acne medication market will likely stay highly competitive due to the significant presence of international pharmaceutical firms and the rising number of specialized dermatological brands. The competing companies are using diverse product lines, including prescription medications, over-the-counter drugs, and combined medications. In addition, the competition is being heightened by the need for effective and low-irritating products and moving away from antibiotic-based drugs. Key focus areas include:
- Development of advanced fixed-dose combination therapies
- Expansion of non-antibiotic and microbiome-friendly treatment pipelines
- Strengthening partnerships with dermatology clinics and e-pharmacy platforms
- Focus on faster product approvals and differentiated formulations
- Expansion into high-growth markets
Market Report Scope
Acne Medication Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 9,027.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.9% | 2033 Value Projection: | USD 12,658.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Galderma S.A., Johnson & Johnson, Bausch Health Companies Inc., AbbVie Inc., GlaxoSmithKline plc, Pfizer Inc., Sun Pharmaceutical Industries Ltd., Bayer AG, Viatris Inc., and Teva Pharmaceutical Industries Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the acne medication market will witness the emergence of personalized and sustained treatment approaches for acne as more cases of adult and recurring acne develop in the future. This includes non-antibiotic treatment modalities, products that favor skin microflora, as well as dermatologic cosmeceuticals due to the trend among regulatory agencies and clinicians of avoiding sustained antibiotics treatments. Moreover, there is also a potential for AI-based diagnostics and tele-dermatology platforms such as Miiskin.
- The greatest opportunity exists in the fixed-dose combinations market segment within the U.S., as the clinical trend in the country prefers combination treatments for patients with moderate acne. Furthermore, the prescription hormone therapy segment within Japan is also considered an opportunity segment as more women are suffering from acne and frequently consulting dermatologists for the same.
- For gaining any competitive advantage in the market, companies need to make investments in combination drugs with improved tolerance levels and reduced irritation and develop their non-antibiotic pipeline in line with regulatory requirements. Collaboration with dermatology clinics and technology-based systems will help in enhancing patient compliance and engagement. In addition, the organization should take advantage of its brand strength in combination over-the-counter prescription medicines and explore new markets by developing products locally.
Market Segmentation
- Drug Class Insights (Revenue, USD Mn, 2021 - 2033)
- Retinoid
- Antibiotics
- Fixed Dose Combinations
- Salicylic Acid
- Azelaic Acid
- Benzoyl Peroxide
- Others
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Prescription
- Over-the-Counter
- Indication Insights (Revenue, USD Mn, 2021 - 2033)
- Inflammatory Acne
- Non-Inflammatory Acne
- Formulation Insights (Revenue, USD Mn, 2021 - 2033)
- Topical Medication
- Oral Medication
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Galderma S.A.
- Johnson & Johnson
- Bausch Health Companies Inc.
- AbbVie Inc.
- GlaxoSmithKline plc
- Pfizer Inc.
- Sun Pharmaceutical Industries Ltd.
- Bayer AG
- Viatris Inc.
- Teva Pharmaceutical Industries Ltd.
Sources
Primary Research Interviews
- Dermatologists and clinical specialists from leading hospitals and skin clinics (e.g., acne treatment centers, aesthetic dermatology chains)
- Pharmaceutical companies developing topical and systemic acne therapies (retinoids, antibiotics, hormonal treatments)
- Contract research organizations (CROs) involved in dermatology clinical trials and formulation development
- Skincare product developers and cosmeceutical experts focusing on acne-prone skin solutions
Stakeholders
- Acne Medication Solution Providers: (e.g., pharmaceutical companies, dermatology-focused biotech firms, cosmeceutical brands, OTC skincare manufacturers)
- End-use Sectors:
- Hospitals and Dermatology Clinics
- Retail Pharmacies and Drug Stores
- E-commerce Platforms
- Homecare / Self-treatment users
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – drug approvals and safety regulations for acne treatments, European Medicines Agency (EMA) – evaluation of dermatology drugs and retinoid safety frameworks, World Health Organization (WHO) – global skin disease burden and antimicrobial resistance guidance
Databases
- ClinicalTrials.gov – clinical trials on acne drugs, biologics, and dermatology treatments
- WHO Global Health Observatory – dermatological disease burden data
- PubMed / NCBI – research publications on acne pathophysiology and treatment outcomes
- OECD Health Statistics – healthcare spending and dermatology treatment trends
Magazines
- Nature (Dermatology / Translational Medicine sections) – advancements in acne therapeutics
- Science Magazine – innovations in biologics, microbiome-based skincare, and drug delivery
- NIH Research Matters – updates on skin disease research and treatment innovations
Journals
- Journal of the American Academy of Dermatology (JAAD)
- JAMA Dermatology
- British Journal of Dermatology
- Dermatology and Therapy
Newspapers
- The New York Times (Health Section) – coverage on acne trends, skincare awareness, and treatment innovations
- The Guardian (Health & Science) – reporting on antibiotic resistance and dermatology research
- Financial Times – pharmaceutical developments and dermatology drug market trends
- The Economic Times – coverage on dermatology market growth and OTC skincare expansion in Asia Pacific
Associations
- American Academy of Dermatology (AAD)
- European Academy of Dermatology and Venereology (EADV)
- International League of Dermatological Societies (ILDS)
- British Association of Dermatologists (BAD)
Public Domain Sources
- National Institutes of Health (NIH) – dermatology research and acne-related studies
- Centers for Disease Control and Prevention (CDC) – skin health awareness and epidemiology data
- European Commission – healthcare and pharmaceutical regulatory frameworks
- National health ministries (e.g., India, China, Japan) – dermatology programs and public health initiatives
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients