Asia Pacific Pharmaceutical CDMO Market Size and Forecast – 2026-2033
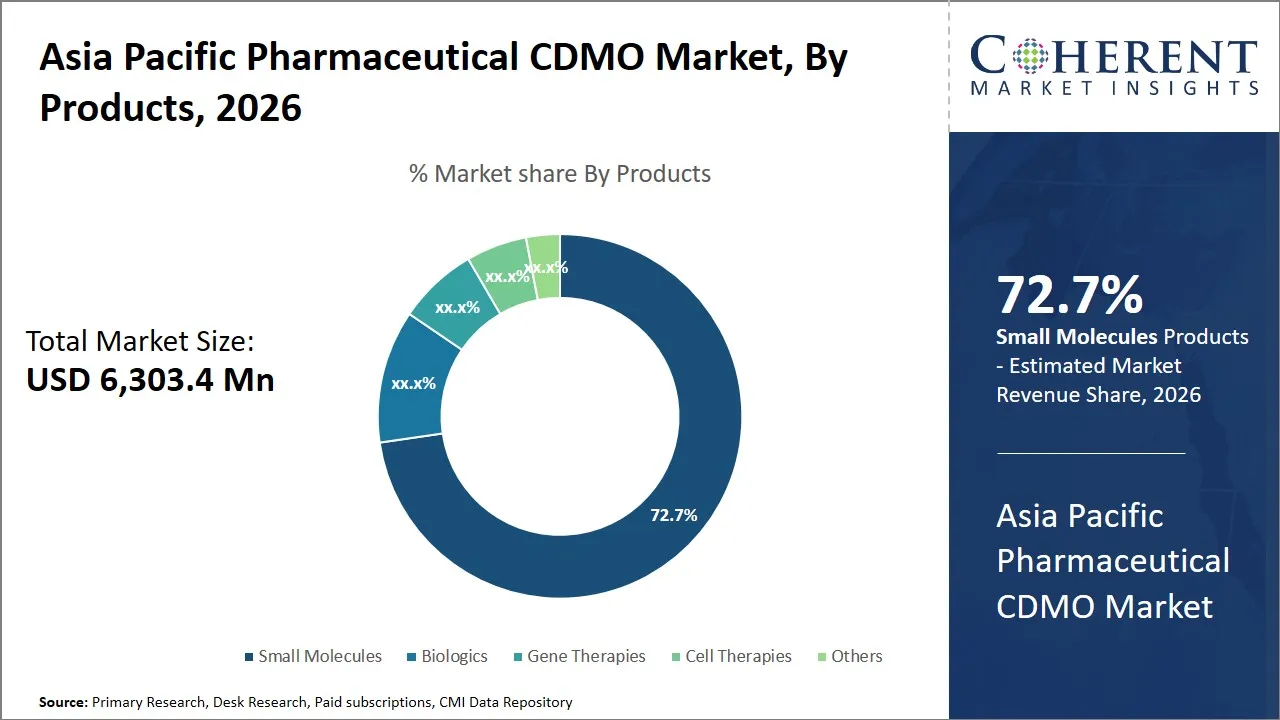
The Asia Pacific pharmaceutical CDMO market is estimated to be valued at USD 6,303.4 Mn in 2026 and is expected to reach USD 9,860.2 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.4% from 2026 to 2033. This substantial growth reflects increasing demand for outsourced pharmaceutical manufacturing and development services driven by rising drug pipelines, cost-efficiency needs, and advancements in biopharmaceuticals across the region.
Key Takeaways of the Asia Pacific Pharmaceutical CDMO Market
- Small molecules segment is expected to lead the Asia Pacific pharmaceutical CDMO market, capturing 72.7% share in 2026.
- Drug manufacturing services segment is estimated to represent 43.9% of the Asia Pacific pharmaceutical CDMO market share in 2026.
- Oncology segment is projected to dominate with 42.8% of the Asia Pacific pharmaceutical CDMO market share in 2026.
Market Overview
- The Asia Pacific pharmaceutical CDMO market is on a strong growth path, supported by rising outsourcing from global pharma and biotech companies that want lower development costs, faster scale-up, and more flexible manufacturing networks across India, China, South Korea, Japan, Singapore, and Taiwan.
- The market is shifting beyond traditional small-molecule manufacturing toward higher-value segments such as biologics, biosimilars, sterile injectables, ADCs, and cell and gene therapy, which is pushing CDMOs to invest in specialized facilities and advanced production platforms.
- A second major driver is the rapid shift toward biologics, biosimilars, sterile injectables, and other complex modalities, which require specialized development and manufacturing infrastructure.
- Growing opportunities in biologics advanced therapies and China plus one outsourcing are creating strong expansion potential for Asia Pacific pharmaceutical CDMOs.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
India’s CRDMO policy push amid China-plus-one diversification |
|
|
Samsung Biologics expanding advanced manufacturing capabilities in South Korea and Japan |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Small Molecules Segment Dominate the Asia Pacific Pharmaceutical CDMO Market in 2026?
The small molecules segment is expected to hold the largest market share of 72.7% in 2026, owing to their established manufacturing processes and broad therapeutic applicability. It is their small molecules that are still the pillars of the Asia Pacific pharmaceutical CDMO market because of a number of reasons that propel their full domination in the market. To begin with, small molecules can take advantage of established technologies of chemical synthesis that are the result of decades of development and offer a solid basis to build effective and affordable contract manufacturing. This level of maturity in the production processes guarantees increased yield and scale which are very important elements to pharmaceutical firms trying to outsource manufacturing to CDMOs.
For instance, in January 2025, Australia-based BioCina and NovaCina announced a strategic merger to create an integrated CDMO platform spanning biopharmaceutical and small-molecule manufacturing. The combined company said the deal would strengthen end-to-end manufacturing capabilities and expand its global reach from an Asia Pacific base, making it a relevant consolidation move for the regional pharmaceutical CDMO market.
(Source- BioCina)
Why Does the Drug Manufacturing Services Segment Dominate the Asia Pacific Pharmaceutical CDMO Market in 2026?
The drug manufacturing services segment is expected to hold 43.9% of the market share in 2026, driven by demand for scalable production and specialized manufacturing technologies. The biggest sector in the Asia Pacific pharmaceutical CDMO market is drug manufacturing services due to the very important level of involvement in the supply chain of batches in clinical trials to commercial-scale production.
The main driver of growth is the growing level of complexity that comes with the production of modern drugs and that demand specialized machinery and technical skills and meeting high-quality standards. These requirements are frequently met by pharmaceutical firms that do not need to incur the costly procedure of constructing and renovating a facility to meet the requirements of the local market.
For instance, in February 2024, Suven Pharmaceuticals announced a strategic merger with Cohance Lifesciences to create a stronger CDMO platform. The merger expands Suven’s research and manufacturing capabilities, enhances its customer base, and strengthens its position in the global contract development and manufacturing market, particularly across Asia Pacific and Europe.
(Source- Suven Pharmaceuticals)
The Oncology Segment Dominates the Asia Pacific Pharmaceutical CDMO Market
The oncology segment is expected to hold the largest market share of 42.8% in 2026, due to increasing cancer prevalence and demand for innovative targeted therapies. The oncology market is the most dominant clinical therapy division sector in the Asia Pacific pharmaceutical CDMO market due to the trend of increasing cancer burden and high demand for sophisticated therapeutic solutions. The increasing incidence and mortality rates of various cancers across countries such as China, India, Japan, and South Korea are driving pharmaceutical companies to make substantial investments in the development of cancer therapeutics. CDMOs play a crucial role in this ecosystem, as they provide specialized services to meet the specific needs of the oncology drug manufacturing.
Clinical Trial Expansion and CRO CDMO Collaboration in Asia Pacific
- Asia Pacific has emerged with a robust clinical trial base that has been backed by huge patient resources, accelerated enrollment, strengthening investigator networks and cheaper execution costs as compared to most of the Western markets. According to BioSpectrum Asia, APAC registered 14,346 clinical trials in 2023 with Western Pacific recording 27,172 trials in Western Pacific alone, as per WHO data, a clear indication that the region has got the capacity to meet the increasing demand of manufacture of clinical supplies, formulations, packaging and tech-transfer services in the Asia Pacific pharmaceutical CDMO market.
- On the CRO side, there exists no known, annual, APAC-wide number of the number of CROs that partner with CDMOs and it would be a guesswork to use a hard number. What is evident is that the partnership model is more becoming integrated: firms like Aurigene explicitly state that they are an integrated CRO-CDMO system, and in June 2025, Axcelead and A2 Healthcare in Japan declared that they were going to have a strategic relationship to construct an integrated support system across drug discovery, non-clinical and clinical development. This is important since closer CRO-CDMO cooperation would reduce time spans and make the Asia Pacific region more appealing to end-to-end outsourcing.
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- On March 9, 2026, Swiss CDMO company, Lonza announced plans to establish a Global Capability Centre in Hyderabad, India, reinforcing the region’s growing importance in the biopharmaceutical manufacturing ecosystem. The facility is expected to support global life sciences operations and strengthen India’s role as a key outsourcing destination for pharmaceutical manufacturing services.
- In October 2025, Japan-based Terumo Corporation completed the acquisition of a drug product manufacturing plant and quality control laboratory from WuXi Biologics in Leverkusen, Germany. The transaction strengthens Terumo’s global CDMO manufacturing capabilities and improves responsiveness to pharmaceutical clients seeking integrated biologics development and manufacturing services.
- In March 2023, Samsung Biologics announced plans to construct Plant 5 at its Songdo campus in South Korea as part of its Bio Campus II expansion. The facility will add 180,000 liters of biologics manufacturing capacity and support rising global demand for contract development and manufacturing services. The investment of over USD 1.27 billion also aims to strengthen capabilities in advanced biologics and next-generation therapies.
Biopharmaceutical Manufacturing Capacity Expansion in Asia Pacific
| Metric |
Asia Pacific Pharmaceutical CDMO Market Insights |
|
|
Capacity of high-potency API (HPAPI) manufacturing facilities |
A single standardized Asia Pacific-wide HPAPI capacity figure is not consistently disclosed in the public domain, but capacity expansion is clearly underway. A recent example is Hikal, which opened a new HPAPI laboratory in Pune, India on October 15, 2025 to strengthen CDMO capabilities for oncology and specialty molecules, indicating rising investment in potent small-molecule infrastructure across the region. |
|
|
Number of cell and gene therapy manufacturing facilities |
There is no single verified public count for all cell and gene therapy manufacturing facilities across Asia Pacific, but new site additions confirm steady expansion. For example, AGC Biologics announced on June 25, 2025 that it would begin cell therapy process development and clinical manufacturing services in Yokohama, Japan from July 1, 2025, showing that the regional advanced-therapy manufacturing base is increasing. |
|
|
Vaccine manufacturing capacity in Asia Pacific |
Asia Pacific has substantial vaccine production capacity and remains a major global manufacturing base. Bharat Biotech alone produces around 700 million vaccine doses annually from four manufacturing facilities, excluding COVID-19 vaccines, according to BioSpectrum Asia. This highlights the region’s strong large-scale biologics and vaccine manufacturing foundation. |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Top Strategies Followed by Asia Pacific Pharmaceutical CDMO Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading CDMOs in Asia Pacific focus on heavy R&D investment, advanced manufacturing technologies, and global partnerships to meet strict regulatory standards and serve multinational pharmaceutical clients. They expand through strategic alliances, capacity additions, and regional facilities to capture growing pharmaceutical outsourcing demand. |
Samsung Biologics, which has continuously expanded its Songdo biomanufacturing campus and launched advanced services such as ADC manufacturing while strengthening partnerships with global pharmaceutical companies. |
|
Mid-Level Players |
Mid-level CDMOs in Asia Pacific compete by offering a practical balance of cost, quality, and customization, making them attractive to generic drugmakers and regional pharma companies that need reliable manufacturing without paying premium pricing. They also strengthen their position through partnerships, technology upgrades, and flexible production models that help them serve niche and mid-volume outsourcing demand more efficiently. |
Suven Pharmaceuticals, which merged with Cohance Lifesciences in February 2024 to expand its CDMO capabilities across contract manufacturing, APIs, and specialty platforms, showing how mid-sized players use strategic deals to scale operations and improve competitiveness. |
|
Small-Scale Players |
Small-scale players in the Asia Pacific pharmaceutical CDMO market stay competitive by focusing on niche technologies, specialized therapeutic areas, and flexible service models that larger firms may not prioritize. They often build strength in areas such as HPAPIs, early-stage biologics, complex formulations, or localized development support, while partnering with regional pharma and biotech companies to secure targeted business opportunities. |
Hikal, which strengthened its niche CDMO position by opening a new HPAPI laboratory in Pune in October 2025, expanding its capabilities in high-potency small-molecule development for oncology and specialty therapies. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Asia Pacific Pharmaceutical CDMO Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6,303.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.4% | 2033 Value Projection: | USD 9,860.2 Mn |
| Segments covered: |
|
||
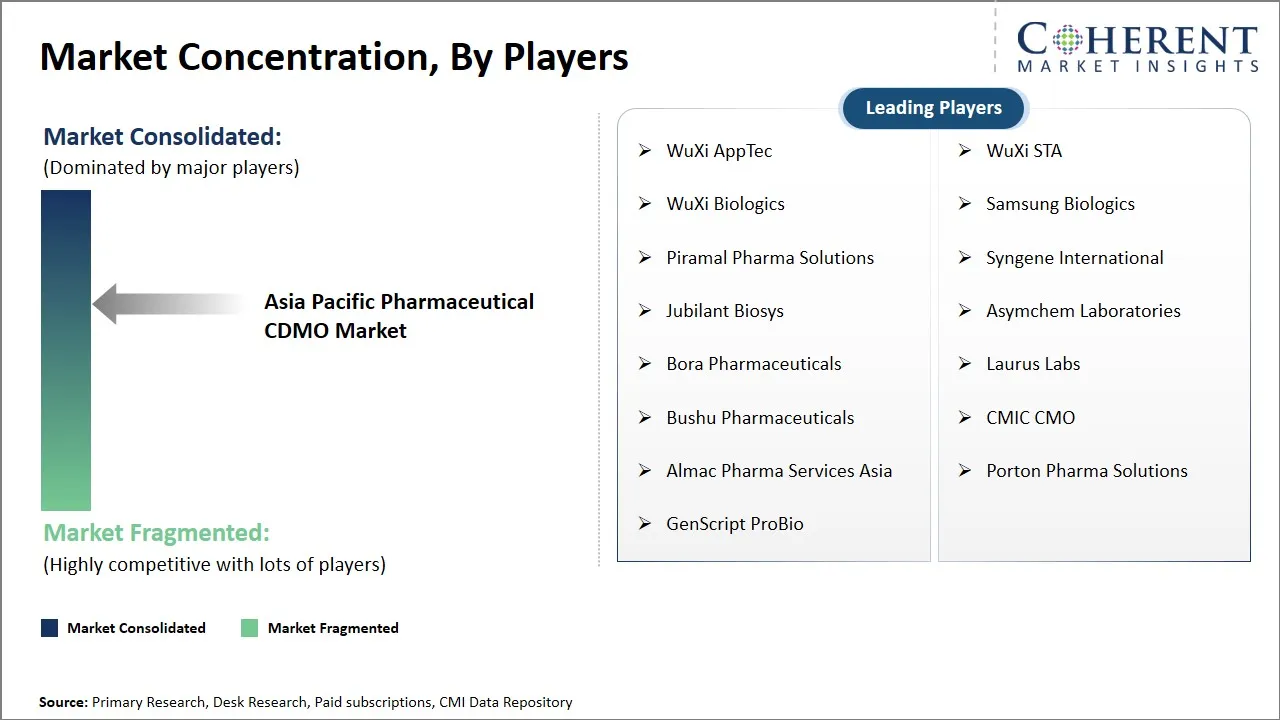
| Companies covered: |
WuXi AppTec, WuXi STA, WuXi Biologics, Samsung Biologics, Piramal Pharma Solutions, Syngene International, Jubilant Biosys, Asymchem Laboratories, Bora Pharmaceuticals, Laurus Labs, Bushu Pharmaceuticals, CMIC CMO, Almac Pharma Services Asia, Porton Pharma Solutions, and GenScript ProBio |
||
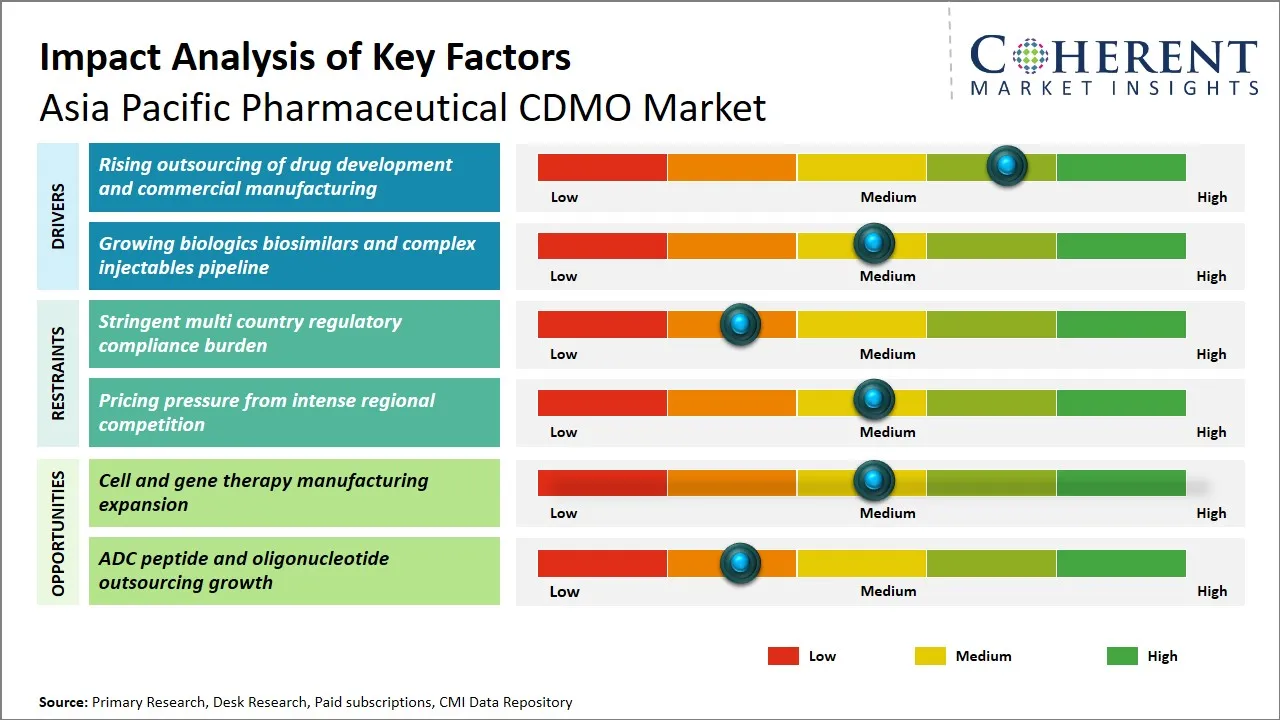
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Asia Pacific Pharmaceutical CDMO Market Dynamics
To learn more about this report, Request Free Sample
Asia Pacific Pharmaceutical CDMO Market Driver - Rising Outsourcing of Drug Development and Commercial Manufacturing
The region of Asia Pacific is also experiencing an unprecedented increase in outsourcing drug development and commercial manufacturing processes due to the increased interest of pharmaceutical companies in operational efficiency optimization and time-to-market minimization. Pharmaceutical companies are increasingly turning to Contract Development and Manufacturing Organizations (CDMOs) as a way to obtain specialized expertise, innovative technologies, and capacity to produce at will without the hefty investment and the risk of developing and continuing to operate an in-house facility. This is also driven by increased complexity of drug pipelines in the biologic and novel therapy where advanced development capabilities are required, with many pharmaceutical companies opting to outsource to reduce their development load.
For instance, in March 2025, Syngene International acquired its first U.S. biologics facility from Emergent, expanding its manufacturing network and strengthening its ability to serve global outsourcing clients. Although the acquired site is in the U.S., the deal is highly relevant to Asia Pacific because it shows an India-based CDMO scaling up to meet rising international outsourcing demand.
(Source- Syngene International)
Asia Pacific Pharmaceutical CDMO Market Opportunity - Expansion in Cell and Gene Therapy Manufacturing
The Asia Pacific pharmaceutical CDMO market is a great growth prospect due to the increase of the cell and gene therapy manufacturing capacities. As the rate of chronic and genetic diseases are rising, the need to have new-fangled treatments aimed at providing targeted and individualized treatment solutions is on the rise. Cell and gene therapies are a novel model in the medical field, which requires special production practices which involve high precision, technology as well as strict compliance with regulations.
Asia Pacific, with the support of countries like China, Japan, South Korea, and India, has been furiously investing in biopharmaceutical infrastructures to facilitate the establishment and commercial scale manufacturing of these complex solutions. In addition, positive government policies, more investment in biotechnology research, and partnerships between pharma firms and CDMOs are driving capacity growth in the region.
For instance, in June 2025, AGC Biologics announced that it would start cell therapy process development and clinical manufacturing services in Yokohama, Japan from July 1, 2025. The move directly reflects capacity expansion in advanced therapy manufacturing and strengthens Japan’s position in the Asia Pacific cell and gene therapy CDMO space.
(Source- AGC Biologics)
Analyst Opinion (Expert Opinion)
- Asia Pacific pharmaceutical CDMO market is expanding due to the increasing number of development and manufacturing assignments outsourced by global pharma companies to minimize their costs, shorten their production times, diversify their supply chains and to gain access to special capabilities in biologics, HPAPIs, sterile injectables, and advanced therapies. The best chance is in the CDMOs that will be able to provide end to end services and the primary challenge will be the regulatory compliance, consistency in quality and the large capital requirements associated with the complex manufacturing. The shift has been facilitated by industry platforms like BIO KOREA 2025 and CPHI Japan 2025 that enhance knowledge sharing, partnerships and visibility of technology throughout the regional outsourcing ecosystem.
- Things have changed in the recent past and this indicates that the market is growing to be more strategic and capability led. The acquisition in March 2025 of a U.S. bio facility by Syngene represents the growth of the region to global platforms in the CDMO business and the entrance of AGC Biologics into higher-value outsourcing business (June 2025 expansion into cell therapy development and clinical manufacturing based in Yokohama). Meanwhile, CRDMO industry in India is still lobbying to gain access to policy and infrastructure assistance in order to win over additional international demand, a move that would further enhance the role of Asia Pacific in the pharmaceutical CDMO market.
Market Segmentation
- Products Insights (Revenue, USD Mn, 2021 - 2033)
- Small Molecules
- Biologics
- Monoclonal Antibodies (mAbs)
- Recombinant Proteins
- Vaccines
- Others
- Gene Therapies
- Cell Therapies
- Autologous Cell Therapy Manufacturing
- Allogeneic Cell Therapy Manufacturing
- Others
- Services Insights (Revenue, USD Mn, 2021 - 2033)
- Drug Manufacturing Services
- Drug Development Services
- Packaging and Labelling Services
- Quality Assurance and Regulatory Services
- Others
- Therapeutic Area Insights (Revenue, USD Mn, 2021 - 2033)
- Oncology
- Cardiovascular Diseases
- Neurology
- Infectious Diseases
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Pharmaceutical and Biotechnology Companies
- Academic Research Organizations
- Others
- Key Players Insights
- WuXi AppTec
- WuXi STA
- WuXi Biologics
- Samsung Biologics
- Piramal Pharma Solutions
- Syngene International
- Jubilant Biosys
- Asymchem Laboratories
- Bora Pharmaceuticals
- Laurus Labs
- Bushu Pharmaceuticals
- CMIC CMO
- Almac Pharma Services Asia
- Porton Pharma Solutions
- GenScript ProBio
Sources
Primary Research Interviews
Industry Stakeholders list
- CDMO business development heads
- API manufacturing plant heads
- Biologics manufacturing directors
- HPAPI and sterile manufacturing specialists
- Regulatory affairs and quality assurance experts
- Clinical supply chain and tech transfer managers
End-users list
- Pharmaceutical company procurement heads
- Biotech company R&D leaders
- Generic drug company outsourcing managers
- Specialty pharma operations heads
- Virtual biotech founders and program managers
- Hospital research and clinical trial procurement teams
Government and International Databases
- ClinicalTrials.gov
- WHO International Clinical Trials Registry Platform
- CDSCO
- PMDA
- NMPA
- HSA
Trade Publications
- BioSpectrum Asia
- Outsourced Pharma
- Pharmaceutical Technology
- Pharmaceutical Commerce
- BioProcess International
- Pharmaceutical Engineering
Academic Journals
- International Journal of Pharmaceutics
- Journal of Pharmaceutical Sciences
- Molecular Therapy
- Nature Biotechnology
- Journal of Pharmaceutical Policy and Practice
- PDA Journal of Pharmaceutical Science and Technology
Reputable Newspapers
- Financial Times
- The Wall Street Journal
- Nikkei Asia
- The Economic Times
- Business Standard
Industry Associations
- Asia Partnership Conference of Pharmaceutical Associations
- Pharmaceutical Manufacturers Association of Japan
- KoreaBIO
Public Domain Resources
- Company annual reports
- Investor presentations
- Stock exchange filings
- WIPO PATENTSCOPE
- The Lens
- UN Comtrade
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients