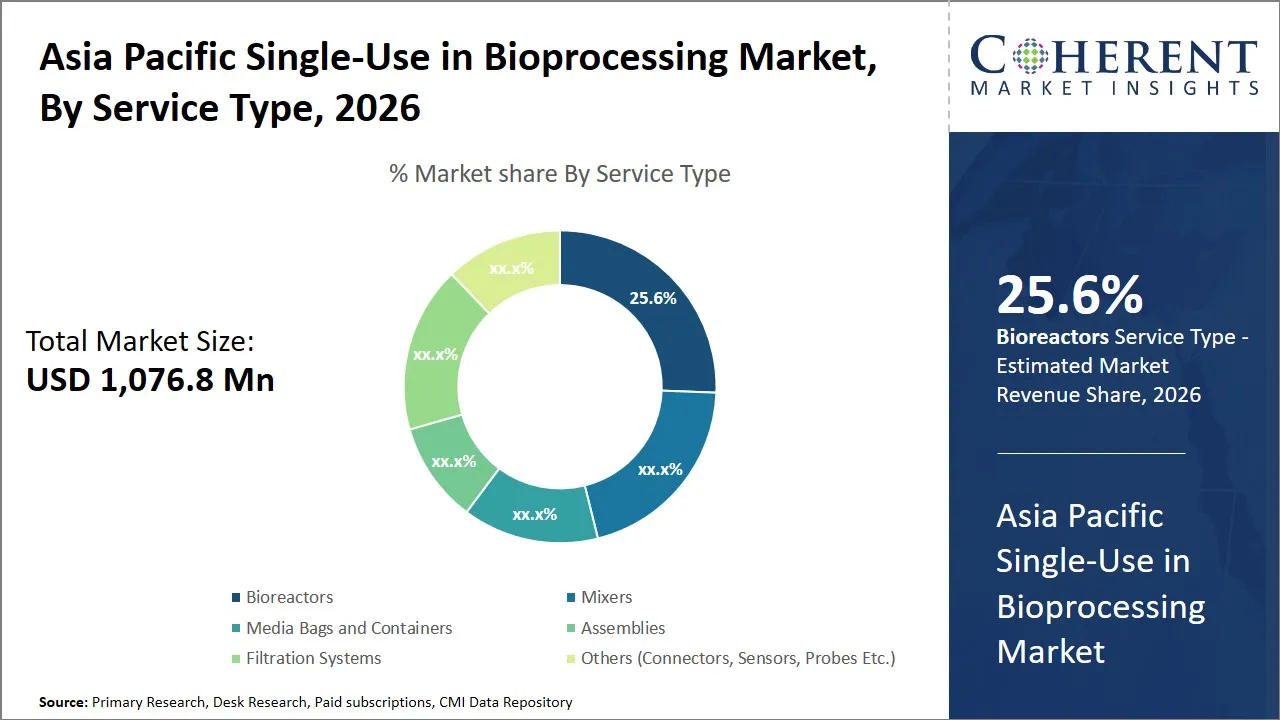
Asia Pacific Single-Use in Bioprocessing Market Size and Forecast – 2026-2033
The Asia Pacific single-use in bioprocessing market is estimated to be valued at USD 1,076.8 Mn in 2026 and is expected to reach USD 1,749.2 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 10% from 2026 to 2033. This steady growth signals sustained demand for single-use technologies in bioprocessing across Asia Pacific, driven by expanding biopharmaceutical production and advancements in biologics manufacturing.
Key Takeaways of the Asia Pacific Single-Use in Bioprocessing Market
- Bioreactors segment is expected to lead the Asia Pacific single-use in bioprocessing market, capturing 25.6% share in 2026.
- Cell culture segment is estimated to represent 29.1% of the Asia Pacific single-use in bioprocessing market share in 2026.
- Biopharmaceutical companies segment is projected to dominate with 44.2% of the Asia Pacific single-use in bioprocessing market share in 2026.
Market Overview
- The Asia Pacific single-use in bioprocessing market is growing rapidly due to rising biologics, biosimilars, vaccine, and advanced therapy manufacturing across China, India, South Korea, Singapore, and Southeast Asia.
- Single-use technologies are gaining strong adoption because they reduce cleaning requirements, lower contamination risk, shorten turnaround time, and improve production flexibility compared with conventional stainless-steel systems.
- Demand is increasing among biopharmaceutical companies, CDMOs, research organizations, and emerging biotech firms that require cost-effective and scalable manufacturing solutions.
- Key product demand is centered on single-use bioreactors, bags, mixers, tubing assemblies, filtration systems, and connectors, as these are essential for both upstream and downstream bioprocessing operations.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Expansion of regional biologics manufacturing capacity |
|
|
Rising investment in India as a biomanufacturing hub |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Bioreactors Segment Dominate the Asia Pacific Single-Use in Bioprocessing Market in 2026?
The bioreactors segment is expected to hold the largest market share of 25.6% in 2026, due to their critical role in cell cultivation and biopharmaceutical manufacturing. The trend of single-use bioreactors (SUBs) development in the area is supported by the flexibility and effectiveness of these devices as opposed to the traditional stainless-steel bioreactors. SUBs do not need to be pre-cleaned and sterilized, which saves a lot of time between production batches as well as can speed up time-to-market of biopharmaceutical products. This is particularly relevant since the bioprocessing industry in Asia Pacific is growing at a swift rate due to an augmentation in the demand of biologics and tailored medicines.
For instance, in March 2025, Emergent BioSolutions Inc. announced an agreement to sell its Baltimore-Bayview drug substance manufacturing facility to Syngene International, a global Contract Research, Development, and Manufacturing Organization (CRDMO), for USD 36.5 million. The divestment aligns with Emergent’s strategy to streamline operations and optimize its overall manufacturing network.
(Source- Emergent BioSolutions Inc.)
Why Does the Cell Culture Segment Dominate the Asia Pacific Single-Use in Bioprocessing Market in 2026?
The cell culture segment is expected to hold 29.1% of the market share in 2026, driven by the escalating demand for biologic drugs, vaccines, and regenerative medicine products. Cell culture is the basic procedure in the generation of complex biologics and the effectiveness, as well as scalability, of single-use systems, has transformed cell culture. The change in the traditional stainless steel bioprocessing platforms to disposable ones enables more simplified production workflow particularly in cell culture operations which require high sterility and process flexibility.
The fast pace of cell therapy and vaccine production in the region, especially in such countries as China, India, and South Korea, has resulted in the emergence of the increased demand in such single-use media bags, mixing systems, and assemblies, boosting cell growth and viability. Single-use systems minimize cross-contamination and facilitate rapid product transitions, which are important in an environment where it is becoming more and more widespread to have multiple cell lines and customized treatment regimens. Furthermore, these systems enable researchers and manufacturers to exercise a high level of control over culture conditions so that the quality of products will be consistent and this is the most important aspect that must be ensured to be approved by the regulators.
For instance, in April 2024, Merck expanded its Shanghai M Lab Collaboration Center by adding a new upstream application lab and process development training center, supporting cell culture optimization and single-use bioprocessing adoption in Asia Pacific.
(Sources- Merck)
The Biopharmaceutical Companies Segment Dominates the Asia Pacific Single-Use in Bioprocessing Market
The biopharmaceutical companies segment is expected to hold the largest market share of 44.2% in 2026, owing to the continuous efforts to innovate biologic drugs and vaccines tailored to meet regional and global healthcare demands. Emergence of biopharmaceutical companies in the area, both multinational corporations and domestic start-ups have highly contributed to the uptake of single-use technologies because of their ability to foster manufacturing agility and lower costs.
The development of biologics is a competitive environment that requires fast development cycles of the process and the single-use systems provide the best solution to the situation by enhancing equipment set-ups and lowering capital expenditures. In the case of biopharmaceutical firms, the reduction of production downtimes and adherence to regulatory requirements are of paramount importance that can be effectively met with the help of single-use bioprocessing equipment. Disposables give a competitive advantage to these businesses to handle multiple pipeline products at once since single use platforms allow them to flexibly produce products without having to extensively validate them through cleaning.
Bioprocessing Infrastructure Overview in the Asia Pacific Single-Use in Bioprocessing Market
|
Bioprocessing Infrastructure Parameter |
Asia Pacific Single-Use in Bioprocessing Market |
|
Number of biomanufacturing facilities designed with single-use systems |
Asia Pacific is seeing a steady increase in biologics and CDMO facilities that are either fully designed around single-use platforms or built as hybrid plants using both disposable and stainless-steel systems. This is especially visible in India, South Korea, Singapore, and China, where new investments are favoring flexible manufacturing layouts for biologics, vaccines, and advanced therapies. |
|
Pilot-scale bioprocessing capacity |
Pilot-scale infrastructure is expanding quickly across the region as suppliers and manufacturers invest in process development, scale-up, and training facilities. Thermo Fisher’s Singapore center is positioned for bench-to-pilot scale bioprocessing support, while its Hyderabad center includes upstream, downstream, and analytical labs designed to move projects from development toward manufacturing readiness. |
|
Commercial-scale plant capacity |
Commercial-scale single-use capacity in Asia Pacific is moving beyond small batches toward high-volume biologics manufacturing. Industry tracking points to large single-use bioreactor facilities in the region, including examples such as WuXi Biologics reported 54,000 L single-use bioreactor facility, showing that commercial-scale deployment is no longer limited to pilot or niche production. |
|
Process development and validation infrastructure |
The regional ecosystem is strengthening through dedicated process development and validation centers that directly support single-use adoption. Cytiva’s expanded Bengaluru Fast Trak center provides upstream and downstream process development, optimization, scale-up, validation services, and technical training under one roof, improving infrastructure readiness for single-use deployment in South Asia. |
|
Regional technical support and design centers |
Infrastructure growth is no longer limited to production plants. Asia Pacific is also adding bioprocess design centers and customer collaboration hubs that help companies test workflows, optimize process design, and de-risk implementation of disposable systems. Thermo Fisher’s expanded network across Hyderabad, Singapore, and Incheon is a clear indicator of this support-layer buildout. |
|
Upstream processing infrastructure |
Upstream infrastructure is becoming more single-use oriented, with stronger deployment of disposable bioreactors, mixing systems, and media preparation setups. The Hyderabad bioprocess design center specifically includes cell culture and microbial culture rooms, reflecting rising regional demand for flexible upstream systems suited to biologics and biosimilars production. |
|
Downstream processing infrastructure |
Downstream capabilities are also improving, though this remains more selective than upstream adoption because some purification steps still rely on hybrid configurations. Even so, new APAC facilities increasingly integrate single-use filtration, fluid management, and closed transfer assemblies to reduce contamination risk and improve turnaround time. Cytiva’s Bengaluru expansion explicitly combines upstream and downstream process support. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In December 2025, Thermo Fisher Scientific announced the expansion of its bioprocess design centers across Asia, including facilities in Hyderabad, Incheon, and Singapore. The initiative provides regional biopharma companies with access to advanced single-use bioprocessing technologies and process development support, helping accelerate biologics and vaccine manufacturing in the Asia Pacific region.
- In May 2025, Thermo Fisher Scientific expanded its DynaDrive single-use bioreactor portfolio with the launch of a 5-liter model, designed to support process development and small-scale biologics production. The system enables faster scale-up and flexible biomanufacturing workflows for biopharmaceutical companies in Asia Pacific, where demand for modular single-use technologies is rising alongside growth in biologics and vaccine production.
- In April 2025, Sartorius Stedim Biotech announced a strategic collaboration with Tulip Interfaces to introduce Biobrain Operate, a digital manufacturing platform integrated with single-use bioprocessing equipment. The partnership aims to digitize operations, reduce process variability, and support paperless manufacturing for biopharma facilities, strengthening efficiency and regulatory compliance in single-use bioprocessing environments.
Clinical and Biologic Pipeline Indicators in the Asia Pacific Single-Use in Bioprocessing Market
| Clinical & Biologic Pipeline Indicator |
Asia Pacific Single-Use in Bioprocessing Market |
| Number of biologic drug candidates in clinical trials in Asia Pacific |
Asia Pacific has a large and expanding biologics pipeline, led by China, Japan, South Korea, India, Singapore, and Australia. The region is seeing more monoclonal antibodies, bispecific antibodies, antibody-drug conjugates, biosimilars, and other advanced biologics move into mid- to late-stage development, with China in particular pushing more indigenous biologics into Phase III and toward approval. This rising clinical volume directly supports demand for single-use bioprocessing systems because developers need flexible pilot-scale and clinical-scale manufacturing capacity for multiple programs running in parallel. |
| Pipeline of cell and gene therapy programs in APAC |
APAC has become one of the most important global regions for cell and gene therapy development. By 2025, the region accounted for roughly one-third of active global cell and gene therapy trials, and ARM’s Q3 2025 sector data noted that Asia Pacific surpassed North America in the number of clinical trials. This is a major demand driver for single-use bioprocessing because CGT manufacturing relies heavily on closed, sterile, small-batch, contamination-sensitive production systems. |
| Country concentration of biologics pipeline | China and Japan are the strongest contributors to APAC’s advanced therapy and biologics trial activity, while South Korea is also building a strong innovation pipeline in monoclonal antibodies, bispecifics, ADCs, and cell therapies. China has designated cell and gene therapies as priority areas and has multiple CAR-T therapies in late-stage development, while Japan continues to advance regenerative medicine and iPSC-based trial programs. This concentration of pipeline activity creates high-value regional demand for disposable bioreactors, bags, assemblies, and filtration systems. |
| Late-stage biologics and commercialization readiness |
The APAC pipeline is no longer limited to discovery and early development. China is moving more biologics programs into Phase III and approval stages, and South Korean companies are contributing dozens of first-in-classes and best-in-class candidates. As more programs enter late-stage clinical development and commercial planning, the need arises for scalable single-use manufacturing infrastructure that can support tech transfer, process validation, and launch readiness without the long buildout times of stainless-steel facilities. |
| Cell therapy and CAR-T program momentum |
APAC’s CGT pipeline is especially active in cell therapy, including CAR-T for hematologic cancers and regenerative medicine programs. South Korea has advanced CAR-T trials, Japan continues regenerative medicine expansion, and China remains aggressive in late-stage CGT development through supportive policy and priority review pathways. This is highly relevant to the single-use market because cell therapy manufacturing is typically low-volume, patient-specific or indication-specific, and heavily dependent on sterile disposable components and closed processing systems. |
| Clinical trial ecosystem support in APAC |
The broader APAC trial ecosystem is improving alongside the pipeline. India, for example, is emerging as an alternative hub for early-stage trials, and Parexel said in February 2025 that it operates 100–150 trial sites in India and plans to expand headcount to support growth. A broader and more distributed trial network increases demand for development-scale and pilot-scale bioprocessing infrastructure, where single-use technologies are often preferred because they allow faster setup, lower contamination risk, and lower capital commitment. |
|
Implication for single-use in bioprocessing |
The clinical and biologic pipeline indicators in APAC clearly support strong long-term adoption of single-use bioprocessing. A larger number of biologics, biosimilars, and CGT candidates in clinical development means more need for flexible upstream and downstream systems during process development, clinical supply manufacturing, and scale-up. In practice, the region’s growing pipeline is one of the clearest structural demand indicators for single-use bioreactors, mixers, bags, tubing, filtration units, and closed fluid-management assemblies. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Top Strategies Followed by Asia Pacific Single-Use in Bioprocessing Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established players maintain leadership by investing heavily in R&D to develop more reliable, scalable, and high-performance single-use bioprocessing products. They also strengthen their position through partnerships with biopharma companies and OEMs, while expanding distribution and technical support networks across high-growth APAC markets such as China, India, and Southeast Asia. |
Thermo Fisher Scientific expanded its bioprocess design center network across Hyderabad, Singapore, and Incheon in 2025, reinforcing its regional presence and improving local process development and scale-up support for biopharma manufacturers. |
|
Mid-Level Players |
Mid-level players compete by offering cost-effective single-use bioprocessing solutions that balance quality, compliance, and affordability. Their strategy usually focuses on improving manufacturing efficiency, tightening supply chains, and building partnerships to expand product range and market reach, especially among price-sensitive biopharma companies and CMOs in Asia Pacific. |
Repligen strengthened its position in the region by expanding partnerships and broadening its bioprocessing portfolio, helping mid-tier customers access reliable single-use technologies without moving to the highest-cost premium systems. |
|
Small-Scale Players |
Small-scale players compete by focusing on niche single-use products such as sensors, connectors, and modular components that solve specific bioprocessing challenges. They usually differentiate through innovation, faster customization, and local partnerships with startups, research centers, and domestic manufacturers to build credibility and reach targeted customers in Asia Pacific. |
In Hyderabad, the launch of 1 Bio, described as India’s first single-use bioprocess design and scale-up hub, highlights how smaller and emerging ecosystem players are using localized innovation platforms to strengthen biologics development and scale-up capabilities in the region. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Asia Pacific Single-Use in Bioprocessing Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,076.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10% | 2033 Value Projection: | USD 1,749.2 Mn |
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific, Merck KGaA, Sartorius AG, Cytiva, Danaher Corporation, Avantor, Repligen Corporation, Corning Incorporated, Eppendorf SE, Meissner Corporation, Pall Corporation, Getinge AB, FUJIFILM Irvine Scientific, Saint Gobain Life Sciences, and PBS Biotech |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Asia Pacific Single-Use in Bioprocessing Market Dynamics
To learn more about this report, Request Free Sample
Asia Pacific Single-Use in Bioprocessing Market Driver - Rising Biologics and Biosimilars Production Across Asia Pacific
The Asia Pacific region has seen a tremendous growth of biologics and biosimilars through the growing healthcare needs and the favorable governmental policies that have been enacted to promote the establishment of biopharmaceuticals. Most of the nations in the region like China, India and South Korea are investing in state-of-the-art bioprocessing plants to serve both their domestic market and the global markets. Such growth is driven by such factors as increasing rates of chronic diseases, aging of the population, and increasing knowledge regarding sophisticated treatments. The manufacturers are therefore moving towards flexible and efficient bioprocessing solutions with single-use technologies being favored because they can make production easier, minimize risks of contamination and minimize the turnaround time.
For instance, in December 2025, Thermo Fisher Scientific announced the expansion of its bioprocess design centers in Hyderabad (India), Incheon (South Korea), and Singapore to strengthen the regional biopharma ecosystem. These facilities provide local expertise, advanced technologies, and support for process development using single-use and hybrid bioprocessing systems, helping biopharma companies accelerate biologics manufacturing and scale-up across Asia Pacific.
(Source- Thermo Fisher Scientific)
Asia Pacific Single-Use in Bioprocessing Market Opportunity - Robust Expansion in Single-Use Systems for ADC and Advanced Therapy Manufacturing
Asia Pacific has a great potential of developing Single-Use Systems (SUS) in the bioprocessing market, specifically so due to the increased pace and rising production needs of Antibody-Drug Conjugates (ADCs) and advanced therapies including cell and gene therapies. These innovative therapies demand contamination-free and flexible and cost-effective manufacturing schemes, which single-use technologies are alone in a position to offer. The use of ADCs and advanced therapies is so complex and sensitive that it requires sterile environments and high turnover rates so that single-use systems are highly appealing because of their capacity to reduce the risk of cross-contamination and lower the time required to validate their cleaning.
Additionally, biologics and personalized medicine manufacturing by biopharmaceutical firms and Contract Development and Manufacturing Organizations (CDMOs) in significant amounts is driving the need of modular, scalable and integrated single-use platforms.
For instance, in March 2025, ABEC launched the Advanced Therapy Bioreactor, a single-use platform for cell therapy manufacturing, highlighting the robust expansion of advanced single-use systems for next-generation therapy production relevant to Asia Pacific bioprocessing markets.
(Source- ABEC)
Analyst Opinion (Expert Opinion)
- The Asia Pacific single-use in bioprocessing market is highly dynamic and is rapidly developing based on the continued growth in biologics, biosimilars, vaccines, and advanced therapies in China, India, South Korea, Singapore and Japan. The greatest growth contributors are increased pace of facility establishment, decreased contamination threat, lesser validation of cleaning necessities, and enhance multiproduct manufacturing flexibility. The recent growth of Cytiva in Bengaluru, Thermo Fisher in Hyderabad, Singapore and Incheon and Merck in South Korea demonstrates that suppliers are adding more local capacity and technical assistance to win this demand. The greatest prospects are in CDMO growth, pilot to commercial scale transfer, and recurring consumable demand, whereas the primary issues are the reliability of supply, management of waste, and standardization of processes.
- As well as reinforcing the direction of the market, recent industry gatherings, including BioProcess International Asia and ISPE Singapore, have taken manufacturers, technology suppliers, and regulatory stakeholders to deliberate on single use systems, facility design, automation, and scale up strategies. They are important events as they lead to more rapid knowledge sharing and increase the level of adoption confidence and move the region into further levels of biomanufacturing ecosystems that are more localized. All in all, APAC is moving away to a cost based production base and towards a more strategic biologics manufacturing hub and single use technologies are reaping the rewards of that change.
Market Segmentation
- Service Type Insights (Revenue, USD Mn, 2021 - 2033)
- Bioreactors
- Mixers
- Media Bags and Containers
- Assemblies
- Filtration Systems
- Others (Connectors, Sensors, Probes Etc.)
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Cell Culture
- Filtration
- Storage
- Mixing
- Purification
- Others (Media, Buffer Preparation etc.)
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Biopharmaceutical Companies
- CROs and CMOs
- Academic and Research Institutes
- Others (Government Organizations etc.)
- Key Players Insights
- Thermo Fisher Scientific
- Merck KGaA
- Sartorius AG
- Cytiva
- Danaher Corporation
- Avantor
- Repligen Corporation
- Corning Incorporated
- Eppendorf SE
- Meissner Corporation
- Pall Corporation
- Getinge AB
- FUJIFILM Irvine Scientific
- Saint Gobain Life Sciences
- PBS Biotech
Sources
Primary Research Interviews
Industry Stakeholders list
- Bioprocessing plant directors
- Biomanufacturing operations heads
- Process development scientists
- Regulatory affairs managers
- CDMO business development heads
- Biologics manufacturing consultants
End-users list
- Biopharmaceutical company manufacturing teams
- Vaccine production managers
- Cell and gene therapy developers
- Biosimilar developers
- Clinical manufacturing teams
- Contract manufacturing users from emerging biotech firms
Government and International Databases
- WHO International Clinical Trials Registry Platform (ICTRP)
- ClinicalTrials.gov
- Pharmaceuticals and Medical Devices Agency (PMDA), Japan
- National Medical Products Administration (NMPA) / Center for Drug Evaluation, China
- Central Drugs Standard Control Organisation (CDSCO), India
- Health Sciences Authority (HSA), Singapore
Trade Publications
- BioProcess International
- Pharmaceutical Engineering
- BioPharma APAC
- BioPharma Asia
- GEN – Genetic Engineering & Biotechnology News
- The Medicine Maker
Academic Journals
- Nature Biotechnology
- Biotechnology and Bioengineering
- Bioprocess and Biosystems Engineering
- Journal of Biotechnology
- Cytotherapy
- Frontiers in Bioengineering and Biotechnology
Reputable Newspapers
- Financial Times
- The Straits Times
- Nikkei Asia
- The Hindu BusinessLine
- South China Morning Post
Industry Associations
- Bio-Process Systems Alliance (BPSA)
- International Society for Pharmaceutical Engineering (ISPE)
- Alliance for Regenerative Medicine (ARM)
- Asia Pacific Biosafety Association (A-PBA)
- Biotechnology Innovation Organization (BIO)
- International Federation of Pharmaceutical Manufacturers & Associations (IFPMA)
Public Domain Resources
- Google Scholar
- WHO Global Observatory on Health R&D
- WIPO PATENTSCOPE
- OECD Data
- data.gov.sg
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients