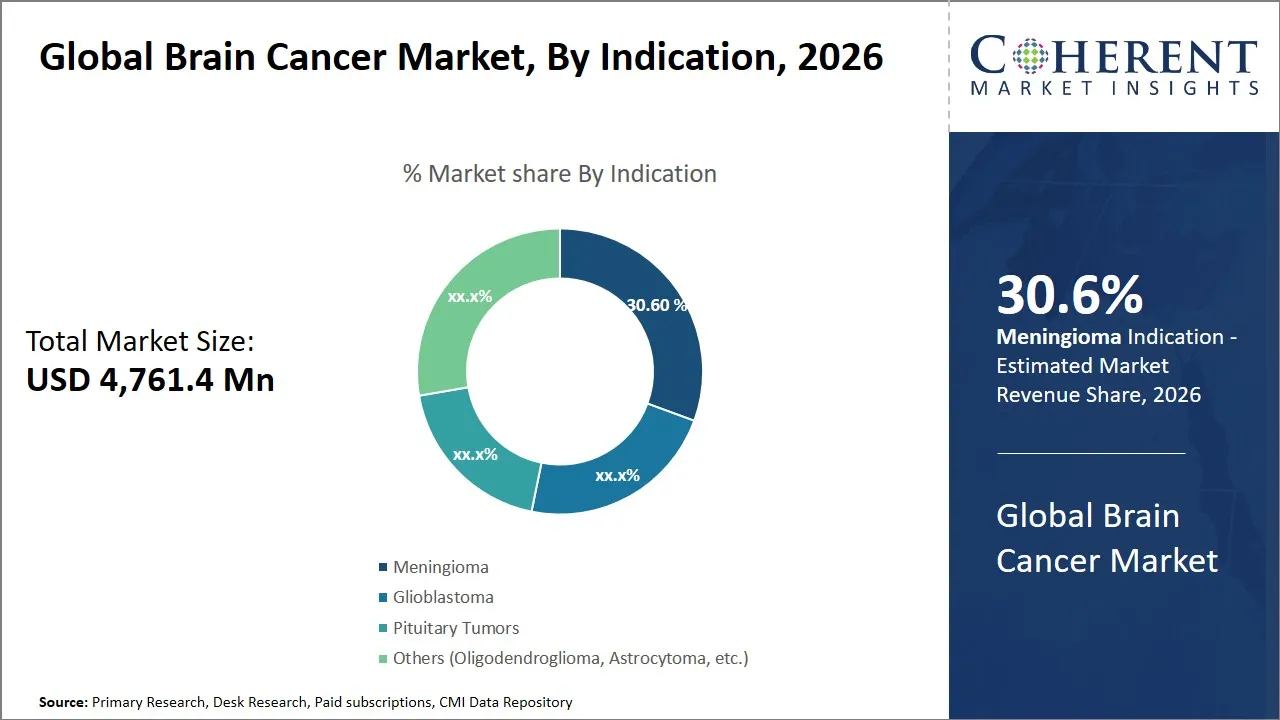
Global Brain Cancer Market Size and Forecast – 2026-2033
The global brain cancer market is estimated to be valued at USD 4,761.4 Mn in 2026 and is expected to reach USD 6,586.6 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 8.3% from 2026 to 2033. This growth reflects increasing investments in research, advancements in diagnostic technologies, and the rising prevalence of neurological disorders worldwide, driving the demand for innovative treatment options and supportive care solutions within the brain cancer domain.
Key Takeaways of the Global Brain Cancer Market
- Meningioma segment is expected to lead the brain cancer market, capturing 30.6% share in 2026.
- Chemotherapy segment is estimated to represent 51.7% of the brain cancer market share in 2026.
- Hospital pharmacies segment is projected to dominate with 53.9% of the global brain cancer market share in 2026.
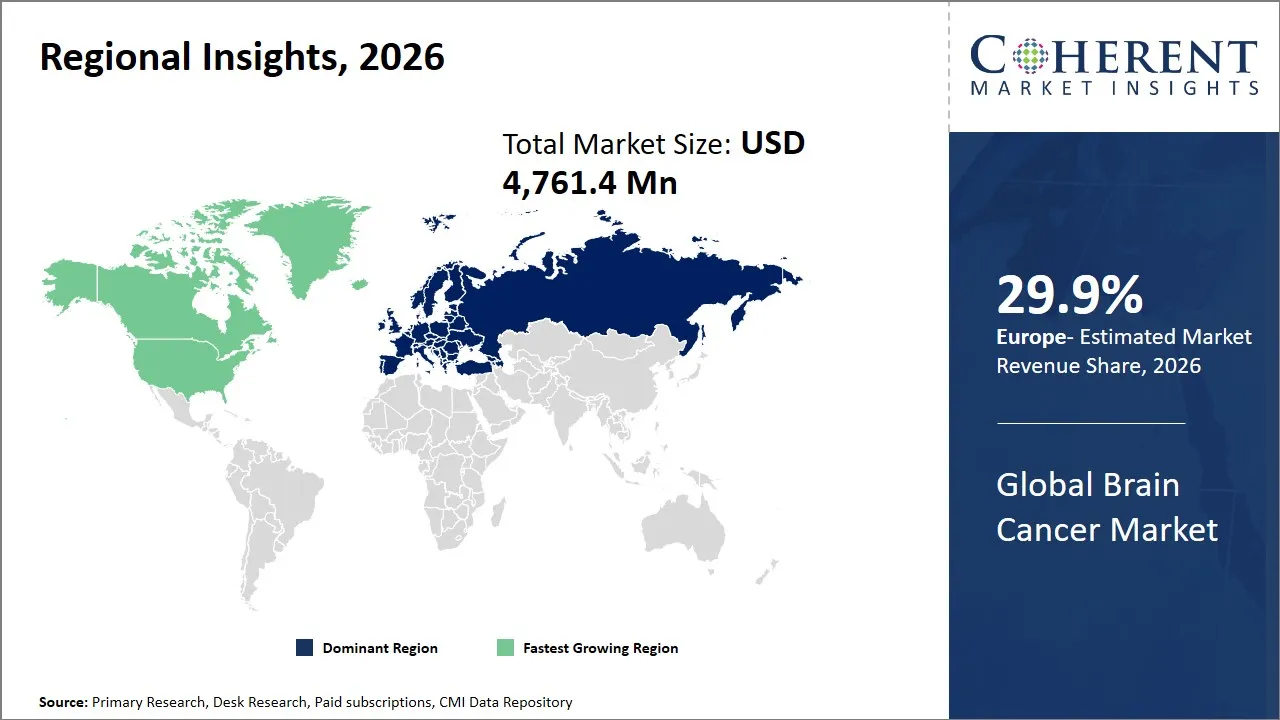
- Europe is expected to lead the market, holding a share of 29.9% in 2026. North America is anticipated to be the fastest-growing region, with 27.3% share in 2026.
Market Overview
- The global brain cancer market is expanding due to the increasing incidence of brain tumors, particularly glioblastoma and meningioma, with aging populations and lifestyle changes contributing to higher diagnosis rates across both developed and developing regions.
- Significant advancements in imaging technologies (like AI-enhanced MRI and PET scans) and targeted therapies (such as IDH inhibitors and BRAF-targeted drugs) are improving early detection, diagnosis accuracy, and treatment efficacy for brain cancer patients.
- As treatment regimens become more personalized, there is growing demand for biomarker-driven therapies, including genetic testing and immunotherapies, which are showing promise in treating previously difficult-to-target brain tumors like gliomas and glioblastomas.
- The development of novel treatments such as immune checkpoint inhibitors, tumor-treating fields (TTF), and radiopharmaceuticals is reshaping the treatment landscape, bringing new hope to patients and expanding market opportunities in the therapeutic segment.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Approval of Vorasidenib for IDH Mutant Gliomas |
|
|
Expansion of AI in Brain Cancer Diagnosis |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Meningioma Segment Dominate the Global Brain Cancer Market in 2026?
The meningioma segment is expected to hold the largest share of 30.6% in 2026, due to its higher prevalence compared to other brain tumors like glioblastoma, as meningiomas are the most common primary brain tumors, accounting for around 36% of all brain cancers. The improvements in treatments and growth in the market are created by the advanced levels of imaging and biomarker-based therapies, which means that the improvement of their early diagnosis is a trend. In addition to this, non-invasive therapies like radiation therapy and stereotactic radiosurgery are improving the outcome of patients; consequently, meningioma has become a primary target of clinical trials and treatment novelty, hence its market leadership.
For instance, on February 17, 2026, the RTOG Foundation announced a Phase II study of 177Lu-DOTATATE Radionuclide in progressive meningioma, while Baptist Health South Florida initiated a trial of cabozantinib for recurrent cases, reflecting growing pipeline activity for targeted treatment options.
(Source - clinicaltrials.gov)
Why Does the Chemotherapy Segment Dominate the Global Brain Cancer Market in 2026?
The chemotherapy segment is expected to hold 51.7% share in 2026, due to its established role as a foundational therapeutic approach and continuous advancements in drug formulations. Chemotherapy is the foundation of the treatment of glioblastoma and other brain malignancies and is commonly employed as monotherapy or in conjunction with surgery and radiotherapy, which increases its significant role in the treatment regimen of patients all over the world.
The pervasiveness of chemotherapy agents like Temozolomide and Carmustine is indicative of the massive clinical validation and their inherent presence in the standard of care guidelines. In particular, temozolomide is a preferable choice as it can cross the blood-brain barrier and it is relatively tolerable, which increases its usability in different groups of patients. The continuous clinical research on the optimization of dosage regimen and combination therapies maintains the continuous demand in this segment.
For instance, in December 2025, scientists from the University of Sydney discovered a mechanism that allows a subset of glioblastoma cells to survive standard chemotherapy and drive tumor regrowth, and identified ways to block this survival trick in preclinical models. When combined with chemotherapy, the new approach dramatically improved survival in mice, offering insights that could lead to more effective future therapies for chemo‑resistant brain cancer.
(Source- The University of Sydney)
The Hospital Pharmacies Segment Dominates the Global Brain Cancer Market
The hospital pharmacies segment is expected to hold the largest share of 53.9% in 2026, due to their pivotal role in ensuring accurate, timely, and safe delivery of complex medication regimens. The visibility of this segment is highly related to the specialized aspect of the treatment of brain cancer that frequently requires multidisciplinary care that hospitals alone are well placed to offer.
The complexity and toxicity of drugs used to treat brain cancer and immunotherapeutics, particularly chemotherapeutics, are factors that require administration under close medical care. Preventing the risks of errors and adverse reactions, hospital pharmacies promote accuracy in dosing, composition, and monitoring. Their internal knowledge on the management of cytotoxic drugs and biologics makes sure that all necessary safety standards are met and this is a critical aspect that makes them dominate the market.
Expansion of Advanced Imaging Infrastructure in the Brain Cancer Market
- Advanced imaging infrastructure in the brain cancer market is expanding at a very high rate with the adoption of technologies such as MRI, PET, and AI-enhanced imaging systems. The tools allow more accurate detection of tumors and tailor the treatment strategy, particularly in high-incidence regions such as North America and Europe. With increased incorporation of advanced imaging in clinical practice, there is a tremendous improvement in the diagnostic precision of gliomas and glioblastomas.
- But the penetration is still uneven, especially in the developing areas because it is too expensive and there is less access to the high-tech. To counter this, regional alliances are coming up with solutions such as mobile MRI services, increased access to quality diagnostics and enhanced early detection in underserved markets.
Regional Insights
To learn more about this report, Request Free Sample
Europe Brain Cancer Market Analysis and Trends
The Europe region is projected to lead the market with a 29.9% share in 2026, driven by advanced diagnostics, personalized treatment research, and collaborative regional initiatives. Investments in novel imaging technology and molecular diagnostics are increasing the number of diagnostic options, and the market is expected to become very vigorous until the end of the decade, as the desire to get better care options and the government policies facilitate the right way of care. The healthcare spending and increased R&D are facilitating this growth which is being contributed by the diagnostic infrastructure in Europe mostly in Germany and other Western countries.
For instance, in June 2025, Siemens Healthineers launched software and hardware enhancements for its Biograph mMR PET/MRI systems, optimized for advanced brain tumor imaging. The updates improve lesion detectability, incorporate AI‑assisted tools, and streamline workflows, enhancing tumor characterization and treatment assessment across European imaging centers. This underscores the growing role of integrated PET/MRI systems in brain cancer diagnostics in Europe.
(Source- Siemens Healthineers)
North America Brain Cancer Market Analysis and Trends
The North America region is expected to exhibit the fastest growth in the brain cancer market contributing 27.3% share in 2026, driven by a highly developed healthcare infrastructure, strong government support for oncology research, and the presence of major pharmaceutical and biotechnology companies. The U. S. ranks first with strong investments in brain cancer research projects, which are supported by the National Cancer Institute (NCI) and National Institutes of Health (NIH) agencies.
The regulatory systems in the region facilitate quick usage of novel treatments gaining momentum to the companies to introduce novel and refined treatment modalities such as immunotherapies and targeted modalities. A highly developed healthcare ecosystem, with the greatest hospitals and research facilities that promote clinical trial and personalized medicine strategies, is also a strong advantage for North America.
Global Brain Cancer Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Brain Cancer Market?
The market of brain cancer in the U.S. can be defined by the innovative atmosphere of research and early use of new treatments. Several clinical trials in the country are undertaken on brain cancer indications, which is backed by good venture capital and government grants.
AbbVie and Bristol-Myers Squibb are key biopharma firms that are driving the field of immunotherapy and precision medicine. There are also the novel device makers like Novocure in the area of tumor treating fields technology which offer alternative treatment. The abundant insurance coverage in the U.S. brain cancer market provides extensive access to costly but beneficial treatment to patients, which serves to facilitate strong market activity.
How is Germany Helping in the Growth of the Brain Cancer Market?
The market of brain cancer in Europe is still dominated by Germany because of its developed healthcare industry and the focus of the government on the development of cancer research and treatment facilities. The ecosystem of specialized cancer centers is well established in the country, which makes the provision of high-quality patients care and clinical research possible.
Leading firms like Bayer and BioNTech are vital in the field of innovation especially in the development of new immunotherapies and oncolytic virus therapies. Academia, industry and healthcare providers collaborative networks facilitate the effective translation of research into practice. Moreover, the reimbursement policies of Germany provide access to new therapies to patients, which supports its existing strong position in the market.
Key Drivers for the Growth of the China Brain Cancer Market
The brain cancer market in China is growing fast due to the policies of the government to increase the capacity of diagnosing and treating cancer. The national health reforms are aimed to provide more access to the oncology facilities and to create awareness amongst the population regarding the importance of early diagnosis.
Local firms such as Hualan Bio and international corporations likeTakeda Pharmaceutical have taken the initiative to release new drugs and diagnostic devices in brain cancer. Demand is further triggered by increasing middle-class population and spending on healthcare. Notably, the regulatory changes have simplified the drug approval and as a result, increased the number of clinical trials and foreign investments, all of which enhance innovation and market growth.
Japan Brain Cancer Market Trends
The brain cancer market in Japan enjoys the advantage of a highly developed medical industry and high-level focus on precision medicine. There are domestic firms, including Daiichi Sankyo and Chugai Pharmaceutical, which play their role in the form of creating targeted therapeutics and more sophisticated diagnostic kits. There is also an emerging demand in the treatment of brain cancer and its support by the government is in line with the health care services required by the aging population.
The healthcare insurance system in Japan enables the access to these treatments with a wide range of people. Moreover, proactive partnership of the government with the industry promotes the development of brain tumor biology research that positively impacts the strength of the pipeline and efficacy of treatment of this illness in the country.
Country-Level Reimbursement Landscape for the Global Brain Cancer Market
| Country |
Reimbursement Scenario |
|
U.S. |
Reimbursement is payer-fragmented but commercially meaningful. Medicare has established coverage for Tumor Treating Fields in newly diagnosed glioblastoma under defined clinical criteria, which supports uptake of high-cost device-based therapy. At the same time, FDA approvals such as vorasidenib and dabrafenib plus trametinib improve the pathway to formulary review, but actual access still varies by commercial plan, prior authorization rules, and site of care. |
|
U.K. |
The U.K. remains a strong HTA-gated market. NICE has recommended dabrafenib plus trametinib for eligible BRAF V600E mutation-positive glioma in children and young people, enabling NHS funding for that subgroup. In contrast, vorasidenib has U.K. regulatory approval through MHRA, but NICE appraisal is still in development, so broad NHS reimbursement is not yet locked in. |
|
Germany |
Germany offers a structured statutory-insurance pathway, with reimbursement shaped by G-BA benefit assessment and subsequent price negotiation. Vorasidenib entered the formal G-BA assessment process in late 2025, which is the key step before routine SHI pricing and access. Germany also already has national reimbursement for Tumor Treating Fields for newly diagnosed glioblastoma, making it one of the more supportive markets for advanced brain-cancer technologies. |
|
France |
France is a selective but supportive reimbursement market where access depends on HAS clinical assessment and later pricing steps. In July 2024, HAS issued a favorable reimbursement opinion for dabrafenib plus trametinib in pediatric low-grade and high-grade glioma with BRAF V600E mutation, indicating support for biomarker-defined therapy in brain cancer. |
|
Canada |
Canada is public-plan driven and condition-based. CDA-AMC has recommended that vorasidenib be reimbursed for eligible patients with grade 2 IDH1/2-mutant astrocytoma or oligodendroglioma, subject to conditions. For dabrafenib plus trametinib in pediatric low-grade glioma, Canadian review activity shows access is still more uneven, with reimbursement depending heavily on jurisdictional follow-through and submission status. |
|
Australia |
Australia remains a PBS-led access market, where reimbursement follows PBAC recommendation and listing arrangements. For pediatric glioma, dabrafenib plus trametinib moved through the PBAC process and was PBS listed from April 2025 for eligible BRAF V600E-mutant low- and high-grade glioma, improving access in a biomarker-defined segment. |
|
Japan |
Japan’s reimbursement model is shaped by national health insurance and has shown willingness to fund selected high-value brain-cancer technologies. Optune received governmental reimbursement in Japan for newly diagnosed glioblastoma, and Japan has also covered BNCT under the National Health Insurance system, indicating openness to advanced device and radiation-based modalities in neuro-oncology. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
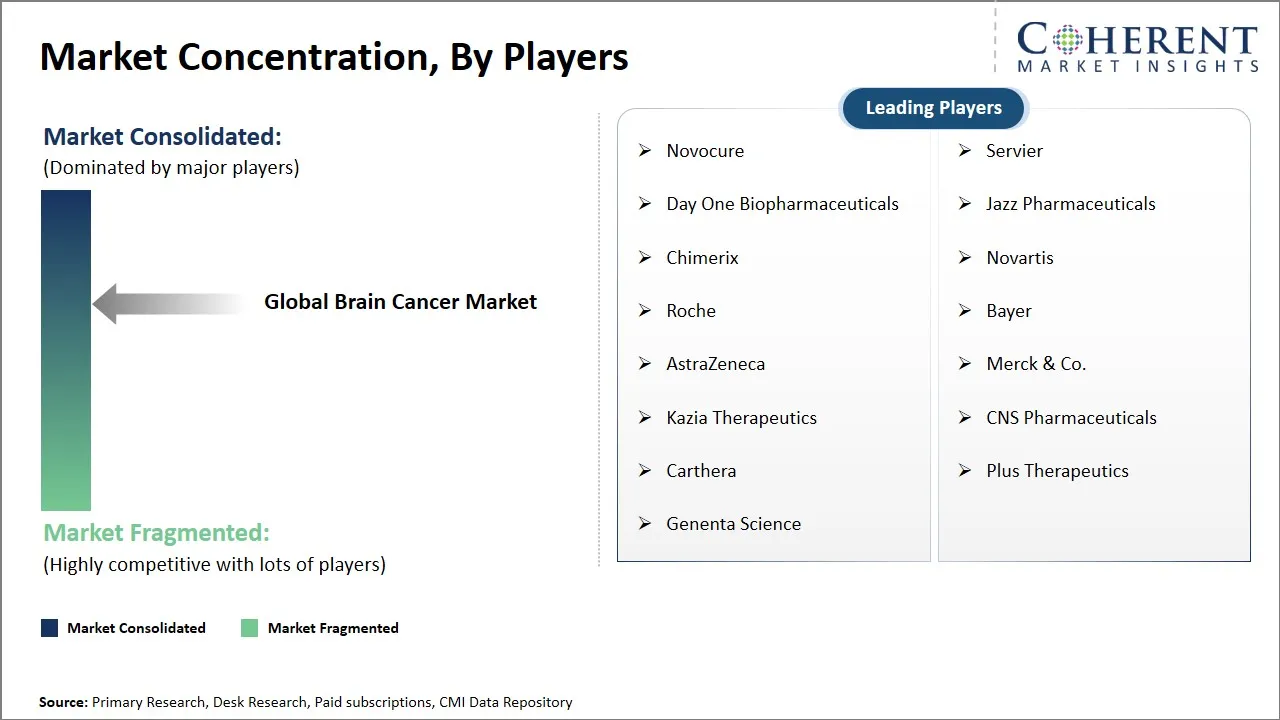
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- On February 20, 2026, FluoGuide announced that the U.S. FDA cleared its IND application for FG001, enabling the start of a U.S. clinical trial in high-grade glioma. The imaging candidate is designed to make malignant tissue light up during surgery, which may improve tumor resection precision in brain cancer treatment.
- On February 18, 2026, Telix announced that it filed a marketing authorization application in Europe for TLX101-Px (18F-FET), its glioma imaging candidate. The product is intended to help distinguish recurrent or progressive glioma from treatment-related changes and is also being developed to support patient selection and response assessment for Telix’s therapeutic brain cancer program.
- In April 2025, Jazz Pharmaceuticals announced the completion of its acquisition of Chimerix for approximately USD 935 million in cash, adding dordaviprone to its oncology portfolio. The deal strengthened Jazz’s position in brain cancer, particularly in rare pediatric and young-adult glioma segments.
Top Strategies Followed by Brain Cancer Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established players dominate the market by investing heavily in R&D, driving innovation in therapeutic products and diagnostic technologies to improve treatment efficacy and patient outcomes. Strategic acquisitions of smaller companies, research institutions, and technology innovators enhance their capabilities and broaden their product portfolios. These acquisitions not only accelerate innovation but also enable expanded market reach, particularly in emerging markets with growing patient demand, strengthening their competitive edge in the global market. |
Jazz Pharmaceuticals exemplifies this strategy with its acquisition of Chimerix, broadening its brain cancer treatment portfolio and strengthening its position in emerging markets for targeted therapies. |
|
Mid-Level Players |
Mid-level companies in the brain cancer market focus on delivering cost-effective solutions that balance affordability and quality to appeal to price-sensitive healthcare providers, especially in emerging markets. By forming strategic partnerships with larger manufacturers or technology providers, they enhance production efficiencies, improve product offerings, and expand market reach without bearing the full burden of high R&D costs. |
Actelion Pharmaceuticals, a mid-sized player, partnered with Johnson & Johnson to leverage their technology and distribution networks, enhancing its reach in the oncology space, particularly in emerging markets where cost containment is crucial. |
|
Small-Scale Players |
Small-scale players in the brain cancer market focus on specialized product features and innovative therapies to address unmet needs. Their agility enables quick adoption of emerging technologies like AI diagnostics and personalized medicine, allowing them to differentiate in a competitive market. By forming localized partnerships with startups and regional manufacturers, they gain access to specific markets with less competition and better regulatory support. |
CNS Pharmaceuticals, a small-scale player, collaborates with research labs and regional manufacturers to expand the availability of its BP-100 brain cancer drug, targeting glioblastoma with personalized treatment approaches. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Brain Cancer Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4,761.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.3% | 2033 Value Projection: | USD 6,586.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novocure, Servier, Day One Biopharmaceuticals, Jazz Pharmaceuticals, Chimerix, Novartis, Roche, Bayer, AstraZeneca, Merck & Co., Kazia Therapeutics, CNS Pharmaceuticals, Carthera, Plus Therapeutics, and Genenta Science |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Brain Cancer Market Dynamics
To learn more about this report, Request Free Sample
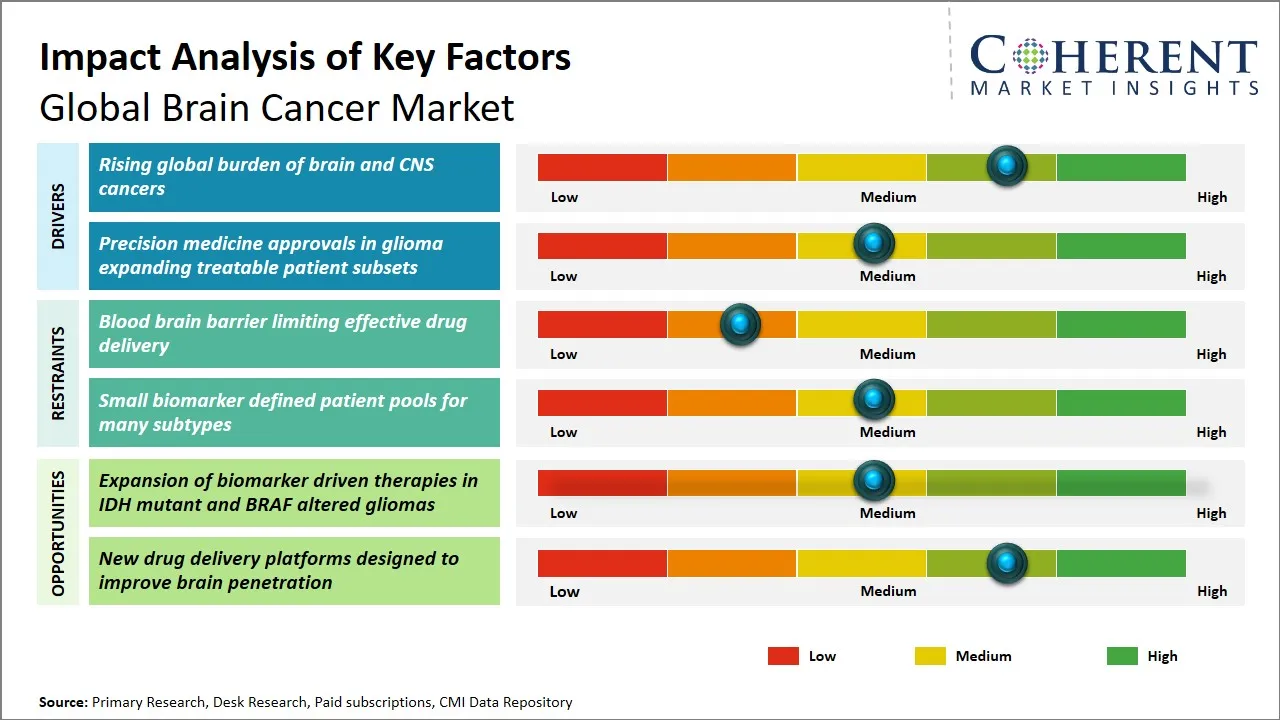
Global Brain Cancer Market Driver - Rising Global Burden of Brain and CNS Cancers
The growing level of prevalence of brain and central nervous system (CNS) cancers in the world is a major factor that contributes to the increase in the global brain cancer market. Improvement in life expectancy and better diagnostic measures has resulted in the detection of these malignancies at earlier stages and this has added to the number of patients who have to undergo more specific treatment modalities. The issue of environmental factors, exposure to radiations that cause harm and genetic inclinations are also important in the increased prevalence of brain tumors.
In addition, brain cancers are complex, and the traditional treatment approaches are not that effective, which drives the need towards innovative and focused treatment options. This growing disease burden drives the necessity of more sophisticated treatment options, and the investments in research and development in this field are driven.
For instance, a 2025 study from Northwestern University reported that nearly 32,000 Americans were diagnosed with primary brain or CNS cancer in 2021, underscoring the continued public health impact of these tumors even as mortality and disability rates show modest declines with improved care.
(Source- Northwestern University)
Global Brain Cancer Market Opportunity - Expansion of Biomarker-Driven Therapies in IDH Mutant and BRAF Altered Gliomas
The opportunity to grow in the global market of brain cancer lies in the potential of growth of biomarker-based therapies against IDH mutant and BRAF altered glioma. Molecular diagnostics have provided the opportunity to identify genetic mutations with great accuracy including isocitrate dehydrogenase (IDH) mutations and BRAF changes that have a significant impact in the pathogenesis and development of different forms of glioma. This model of precision medicine enables development and use of specific therapies that enhance the effectiveness and outcome of patients as opposed to traditional therapies. The presence of IDH mutant gliomas with their unique metabolism and an improved prognosis provides a perfect therapeutic target to be addressed with the help of new agents targeting mutant IDH enzymes, which interferes with the tumor metabolism.
Analyst Opinion (Expert Opinion)
- Technological advancements have created a global market in the treatment of brain cancer as it is becoming more popular in the market because of the support of new treatment procedures by the regulation authorities and the rising demand of tailor-made treatment. The development of biomarker-directed therapy, including IDH and BRAF mutations-targeted therapy, is a driving force. Nevertheless, there are still certain challenges, such as expensive treatment and diagnostic complications that postpone the care.
- Knowledge sharing and policy making has been stimulated by conferences, such as AACR and ESMO, identifying new therapy and technologies. Significant milestones in the market have been the U.S. FDA approval of Vorasidenib in the treatment of IDH-mutant gliomas and the approval of tovorafenib in the treatment of BRAF-altered gliomas which, among other advances in partnerships, such as the acquisition of Chimerix by Jazz Pharmaceuticals, are reinventing the targeted therapies and providing the way to treat brain cancer.
Market Segmentation
- Indication Insights (Revenue, USD Mn, 2021 - 2033)
- Meningioma
- Glioblastoma
- Pituitary Tumors
- Others (Oligodendroglioma, Astrocytoma, etc.)
- Treatment Type Insights (Revenue, USD Mn, 2021 - 2033)
- Chemotherapy
- Carmustine
- Temozolomide
- Combination Drugs (Lomustine, Procarbazine, and Vincristine)
- Targeted Therapy
- Bevacizumab
- Larotrectinib
- Entrectinib
- Others (Dinutuximab, Naxitamab-gqgk etc.)
- Immunotherapy
- Dostarlimab
- Granulocyte-macrophage colony-stimulating factor
- Pembrolizumab
- Chemotherapy
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Novocure
- Servier
- Day One Biopharmaceuticals
- Jazz Pharmaceuticals
- Chimerix
- Novartis
- Roche
- Bayer
- AstraZeneca
- Merck & Co.
- Kazia Therapeutics
- CNS Pharmaceuticals
- Carthera
- Plus Therapeutics
- Genenta Science
Sources
Primary Research Interviews
Industry Stakeholders list
- Neuro-oncology clinicians
- Radiologists specializing in brain cancer
- Brain cancer treatment center administrators
- Healthcare policymakers in oncology
- Medical device manufacturers for brain cancer diagnostics
- Pharmaceutical R&D heads in oncology
End-users list
- Neurosurgeons
- Oncology specialists
- Patients’ advocacy groups for brain cancer
- Research institutions focused on brain tumor treatment
- Cancer hospitals and treatment centers
- Diagnostic laboratories specialized in cancer genomics
Government and International Databases
- National Cancer Institute (NCI)
- World Health Organization (WHO)
- Centers for Disease Control and Prevention (CDC)
- European Medicines Agency (EMA)
- International Agency for Research on Cancer (IARC)
- Global Cancer Observatory (GCO)
Trade Publications
- Journal of Clinical Oncology
- Cancer Research Journal
- Neuro-Oncology Journal
- Oncology Times
- Journal of Neuro-Oncology
- The Lancet Oncology
Academic Journals
- Journal of Clinical Oncology
- Brain Tumor Research Journal
- Cancer Science Journal
- Cancer Letters
- Journal of Neuro-Oncology
- International Journal of Cancer
Reputable Newspapers
- The New York Times
- The Guardian
- The Washington Post
- The Wall Street Journal
Industry Associations
- American Association for Cancer Research (AACR)
- American Society of Clinical Oncology (ASCO)
- European Society for Medical Oncology (ESMO)
- Brain Tumor Society
- National Brain Tumor Society (NBTS)
- World Brain Tumor Alliance
Public Domain Resources
- ClinicalTrials.gov
- National Institutes of Health (NIH)
- Cancer Research UK
- U.S. Food and Drug Administration (FDA)
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients