Global Cell and Gene Therapy for Oncology Market Size and Forecast: 2026-2033
The global cell and gene therapy for oncology market is expected to grow from USD 8,641.9 Mn in 2026 to USD 138,425.0 Mn by 2033, registering a compound annual growth rate (CAGR) of 44.7%. The market for cell and gene therapy for oncology is poised for significant expansion, fueled by soaring global burden of cancer, creating a need for better and personalized therapeutic approaches like cell and gene therapies.
According to the World Health Organization, in 2022, there were an estimated 20 million new cancer cases and 9.7 million deaths. The estimated number of people who were alive within 5 years following a cancer diagnosis was 53.5 million. About 1 in 5 people develop cancer in their lifetime, approximately 1 in 9 men and 1 in 12 women die from the disease. Further, over 35 million new cancer cases are predicted in 2050, a 77% increase from the estimated 20 million cases in 2022.
(Source: World Health Organization)
Key Takeaways of the Global Cell and Gene Therapy for Oncology Market
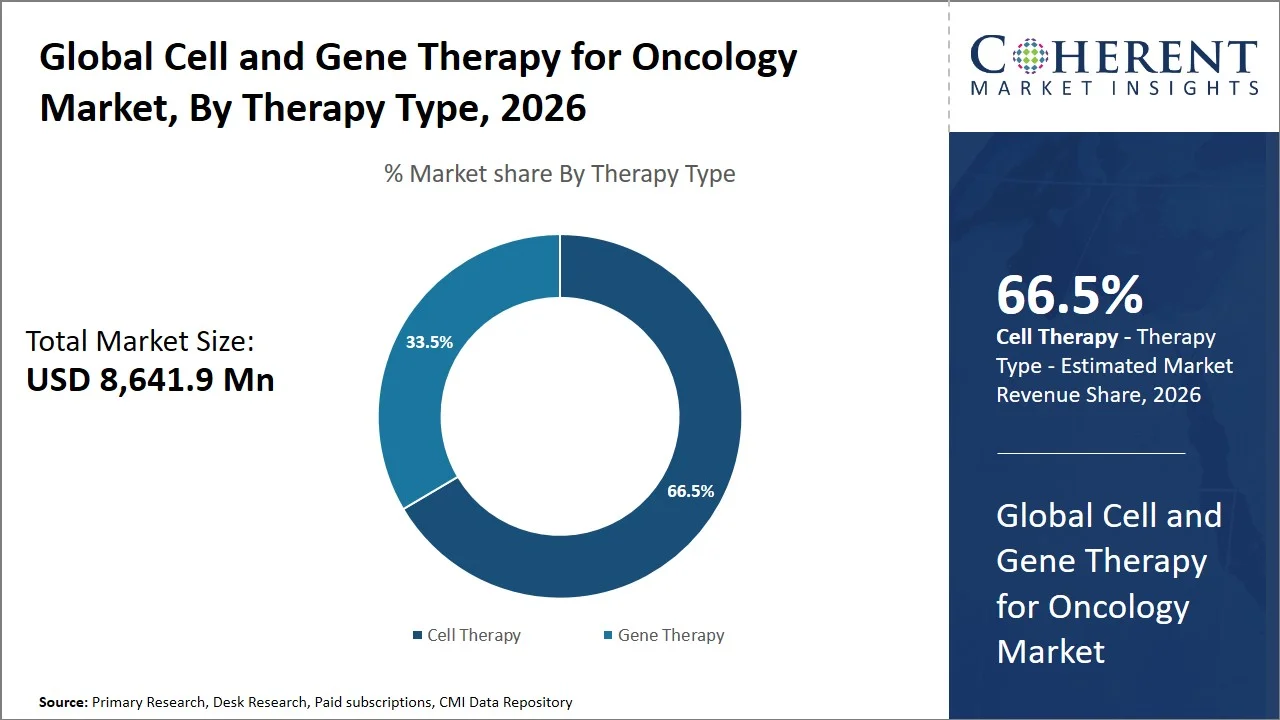
- Cell therapy is projected to hold 66.5% of the global cell and gene therapy for oncology market share in 2026, making it the dominant therapy type segment, led by North America, especially the U.S., owing to its advanced commercialization and adoption of CAR-T cell therapy (FDA approved therapies such as Kymriah and Yescarta) along with the existence of certified treatment centers. These therapies are preferred due to its high success rate in treating hematologic malignancies through patient-specific immune cell modification. For instance, CAR-T therapy in the U.S. was traditionally restricted to fewer than 160 authorized treatment centers under the U.S. FDA’s Risk Evaluation and Mitigation Strategy (REMS) program, emphasizing the unique clinical environment required for their administration. (Source: WCG Clinical)
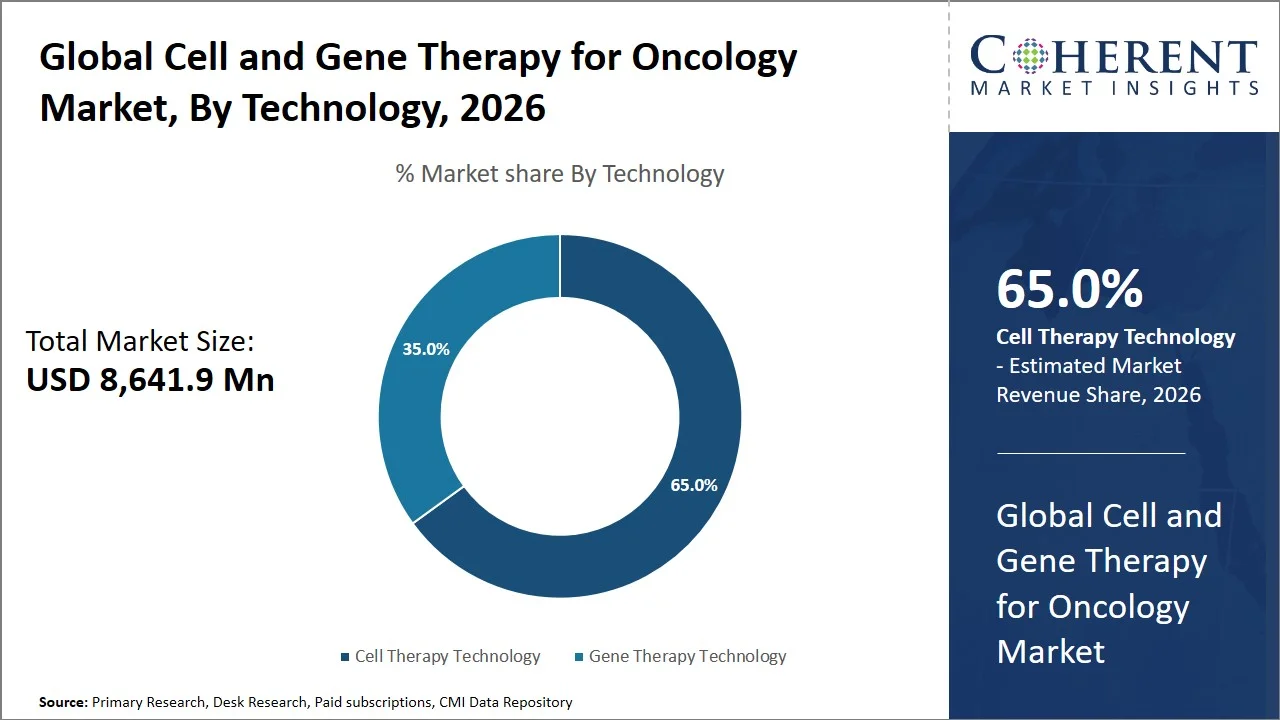
- Cell therapy technology is projected to hold 65.0% of the global cell and gene therapy for oncology market share in 2026, making it the dominant technology segment, with Asia Pacific rising as a prominent growth and manufacturing region attributed to cost advantages and the increasing availability of cell and gene therapy (CGT) manufacturing facilities. Countries like China and South Korea are quickly ramping up production for viral vectors and cell therapies, supported by government initiatives and private investments. For instance, the Ministry of Health and Welfare of South Korea in its “K-Bio/Pharma Strategy” kept on supporting the growth of next-generation biopharmaceutical manufacturing including the manufacture of cell and gene therapies by focusing on building bio clusters and GMP facilities in order to position itself as a cell and gene therapy (CGT) manufacturing hub. (Source: Invest Korea)
- Leukaemia and lymphoma is projected to hold 26.1% of the global cell and gene therapy for oncology market share in 2026, making it the dominant indication segment, with Europe and North America being at the forefront owing to robust evidence of clinical efficacy and reimbursement for CAR-T treatment in hematological cancers (such as CMS coverage under the National Coverage Determination (NCD 110.24) for CAR-T therapies in the U.S., Germany’s statutory health insurance (GKV) reimbursement under the AMNOG framework, and England’s NHS funding for CAR-T therapies through the Cancer Drugs Fund (CDF)). They prevail in the context where blood cancers are easier for the manipulated immune cells to reach than solid tumors, hence greater response rates. For instance, in November 2025, the U.S. FDA approved epcoritamab-bysp (Epkinly) when used in combination with rituximab and lenalidomide for relapsed or refractory follicular lymphoma after its fast-track approval in 2024 based on clinical results of the EPCORE FL-1 study, showing significant improvements in PFS and overall response rates in relapsed or refractory follicular lymphoma patients, reinforcing the strong therapeutic success in hematologic malignancies. (Source: Food and Drug Administration)
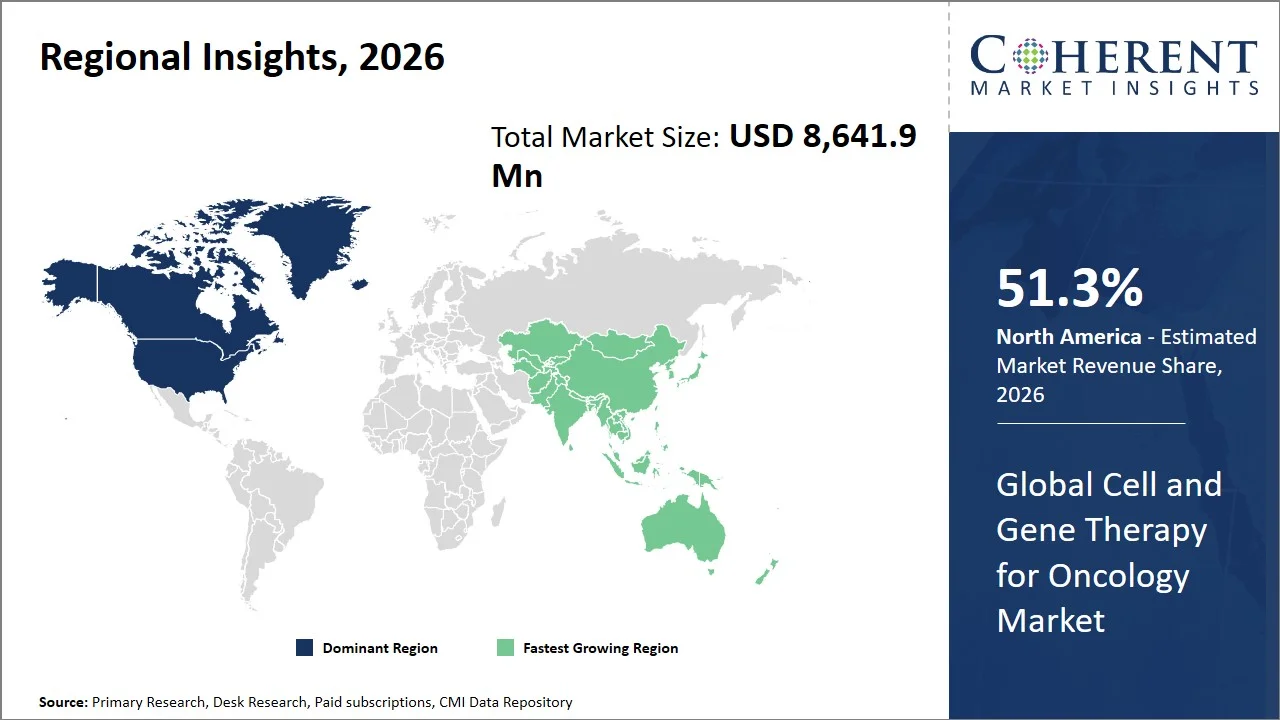
- North America maintains dominance with an expected share of 51.3%in 2026, led by the U.S., attributed to the early commercialization of the next-generation cell and gene therapy (CGT) products and progress in pipeline development under accelerated regulatory approval (e.g., RMAT designation). The region also gains from rapid transition of therapies from clinical to commercial stages. For instance, in February 2024, the U.S. Food and Drug Administration (FDA) approved lifileucel (Amtagvi) as the first tumor-infiltrating lymphocyte (TIL) therapy for unresectable or metastatic melanoma, marking a major advancement in personalized cell therapy beyond CAR-T. (Source: Food and Drug Administration)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 18.2% share in 2026, due to rapid regulatory evolution and growing innovation within the cell and gene therapy (CGT) industry in countries such as China and Japan. Governments are actively working towards accelerating clinical translation and approvals of new advanced therapies through adaptive regulatory frameworks. For instance, in June 2025, the National Medical Products Administration of China provided conditional approval to amimestrocel injection through its priority review program for the indication of steroid-refractory acute graft-versus-host disease, underscoring China’s fast-growing commitment towards supporting advanced cellular treatments, thus cementing its role in the Asia Pacific region in cellular therapy commercialization. (Source: National Medical Products Administration)
- Expansion of In Vivo Gene Editing Platforms in Solid Tumors: The development of in-vivo gene editing technology is offering a new growth opportunity apart from the already known ex vivo CAR-T products. Gene editing platforms incorporating techniques of targeted delivery, including lipid nanoparticles and virus-based vector approaches, are making it possible to directly alter the genes that cause cancer within solid tumors, which was a largely ignored market for cell and gene therapies for cancers in the past.
- Growing Integration of AI-Driven Target Discovery in Cell and Gene Therapy (CGT) Development: Application of AI to detect tumor-specific antigens are bringing a new wave of precision medicine to the market. Utilization of AI technology is contributing to accelerated discovery of gene targets and engineering of cell therapy treatments that are both efficient and less toxic to reduce toxicity. Cooperation between companies such as Ginkgo Bioworks and Recursion Pharmaceuticals indicates the potential of computational biology in enhancing the efficiency of the cell and gene therapy (CGT) pipeline. This trend is most apparent in rare and heterogeneous cancers due to inefficient detection methods in conventional approaches.
Why Does Cell Therapy Segment Dominate the Global Cell and Gene Therapy for Oncology Market?
Cell therapy is projected to hold the market share of 66.5% in 2026, attributed to robust clinical validation, regulatory approval success, and growing indications in various types of blood-related disorders. The application of cell therapies, especially CAR-T, has been proven to bring about complete and sustained remission rates. For instance, in September 2024, meta-analysis of CAR-T therapies in mantle cell lymphoma demonstrated the overall response rate stood at 89% while the complete remission rate was 74%, showcasing effective and enduring clinical results, thus strengthening the dominance of cell therapy in cancer treatment. (Source: Frontiers)
Moreover, the use of CAR-T treatments by companies like Gilead Sciences and Novartis has been successful in setting significant clinical standards and commercial footprints for leukemia and lymphoma patients, underscoring the supremacy of cell-based treatment approaches. The consistent development of next-generation technology platforms, such as those involving allogeneic and gene-edited cells, is ensuring that cell therapy continues to lead in the global cell and gene therapy for oncology market.
Why Does Cell Therapy Technology Represent the Largest Technology Segment in the Cell and Gene Therapy for Oncology Market?
To learn more about this report, Request Free Sample
Cell therapy technology is projected to hold a market share of 65.0% in 2026, owing to their proven efficacy and reliability, scalable nature of the platforms, and solid regulatory progress made in both autologous and promising allogeneic cell therapy methods. Autologous cell therapies have been adopted widely because they have low risk of rejection and offer high efficacy, whereas improvements are being made to the scalability and speed of manufacturing allogeneic therapies.
The dominance of the segment is further supported by the innovations in cell harvesting, automation, and expansion technologies that enables more efficient production and broader accessibility. For instance, in February 2025, Allogene Therapeutics announced the findings from its Phase 1 ALPHA/ALPHA2 studies involving ALLO-501A (cemacabtagene ansegedleucel). The overall response rate stood at 67%, while the complete response rate reached 58% for patients with relapsed/refractory large B-cell lymphoma, with remissions lasting more than 23 months, confirming the feasibility and effectiveness of allogeneic cell therapy. (Source: Allogene Therapeutics)
Leukaemia and Lymphoma Segment Dominates the Global Cell and Gene Therapy for Oncology Market
The leukaemia and lymphoma segment is projected to hold a market share of 26.1% in 2026, since they possess an increased level of clinical sensitivity towards immunotherapy treatments, whereby the cancer cells that develop in the body can be easily recognized by the immune system. These conditions have experienced quick clinical translation and have become extensively applied within CAR-T treatments, courtesy of consistent approval and introduction into various treatments.
For instance, in May 2024, the U.S. FDA granted an accelerated approval to lisocabtagene maraleucel (Breyanzi) for patients affected by relapsed or refractory follicular lymphoma, with an overall response rate of 95.7%. It indicates that cell therapy techniques can be considered extremely efficient in treating blood-based cancers. (Source: Food and Drug Administration) Therefore, the ongoing application of CAR-T treatment techniques in lymphoma and leukaemia, along with positive results, will ensure that the segment maintains its dominance in the global cell and gene therapy for oncology market.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Expansion of CGT-Specific Guidance & Real-World Evidence Requirements (2025–2026) |
|
|
EU Pharmaceutical Legislation Reform & ATMP Regulatory Acceleration (2025 Agreement) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Food and Drug Administration, European Commission)
Global Cell and Gene Therapy for Oncology Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
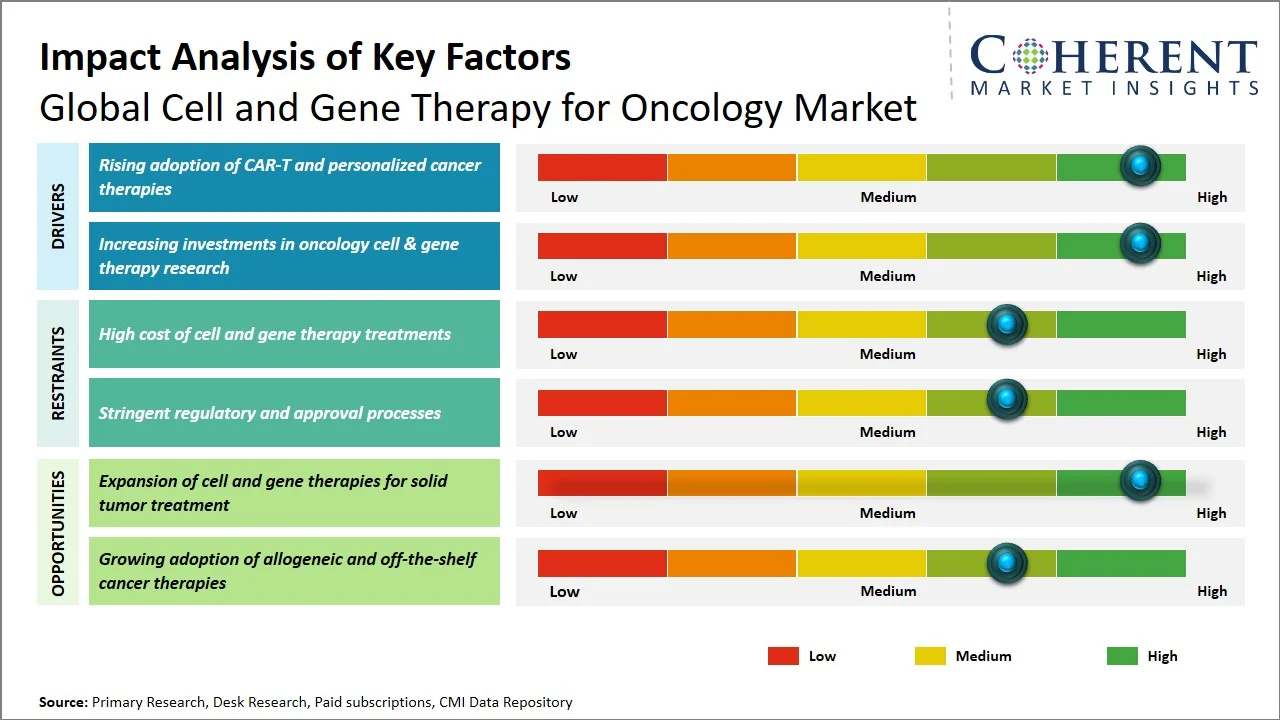
- Rising adoption of CAR-T and personalized cancer therapies: The growing use of CAR-T and personalized cancer drugs represents a major factor driving the global market for cell and gene therapy for oncology, given their customized nature that results in sustainable effects in hematological cancers. Moreover, technological advancements in regulations have increased the uptake of such drugs owing to ease of access and less treatment barriers. For instance, in June 2025, the U.S. Food and Drug Administration discontinued the Requirement for Risk Evaluation and Mitigation Strategies for CAR-T treatments, eliminating the requirement of specialized facilities and reducing logistical burden, hence facilitating greater accessibility and rapid adoption across healthcare settings. (Source: Food and Drug Administration)
- Increasing investments in oncology cell & gene therapy research: Investments in research and development efforts related to oncology-specific cell and gene therapies have been identified as being major contributors to the growth witnessed by the market. This is primarily because there have been continued investments in developing immunotherapy solutions, gene editing tools, and manufacturing technology for new generation therapies. These investments are accelerating clinical trials, upgrading success rates, and enabling faster commercialization of advanced therapies. For instance, in May 2026, Latus Bio raised USD 97 million through Series A financing for developing its pipeline for gene therapy products and providing these therapies to a larger number of patients, reflecting high investor interest in the cell and gene therapy for oncology market. (Source: Latus Bio)
Emerging Trends
- Shift Toward Allogeneic (“Off-the-Shelf”) Cell Therapies: There is a shift from autologous to allogeneic cell therapy for addressing scalability, cost, and turnaround time issues. Firms such as Allogene Therapeutics have developed CAR-T pipelines derived from donors that seek to facilitate large-scale manufacturing and rapid access for patients. It is projected that the shift will increase acceptance, especially in oncology applications.
- Convergence of Gene Therapy with Next-Generation Immuno-Oncology: New strategies have begun merging gene editing with immunotherapy to increase their specificity for tumor destruction and to bypass the development of resistance mechanisms. Companies like CRISPR Therapeutics are engineering immune cells that are more persistent and accurate in their destruction of solid tumors. This convergence is driving innovation toward more durable and effective cancer treatments.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Cell and Gene Therapy for Oncology?
North America leads the global cell and gene therapy for oncology market, accounting for an estimated 51.3% share in 2026, attributed to highly supportive regulatory environment, promising clinical pipeline, and fast adoption of innovation. The U.S. Food and Drug Administration play a vital role through initiatives such as RMAT designation (Regenerative Medicine Advanced Therapies), which is especially aimed at accelerating research and approval for the treatment of critical diseases including cancer. (Source: Food and Drug Administration)
This has directly fueled the development of cell and gene therapy for oncology, such that the U.S. FDA gave RMAT designation in March 2026 to a CAR-T therapy (CB-011), allogenic, for the treatment of multiple myeloma owing to positive results from clinical trials, enabling faster regulatory review and potential accelerated approval. (Source: Caribou Biosciences, Inc.) Combined with the availability of renowned cancer research institutions (such as MD Anderson Cancer Center, Memorial Sloan Kettering Cancer Center, and Dana-Farber Cancer Institute), extensive clinical trials, and supportive reimbursement mechanisms, these constant regulatory support and innovations have strongly established North America as the dominant center for cell and gene therapies within oncology applications.
Why Does Asia Pacific Cell and Gene Therapy for Oncology Market Exhibit High Growth?
The Asia Pacific cell and gene therapy for oncology market is expected to exhibit the fastest growth with an estimated contribution of 18.2% share to the market in 2026, owing to the high prevalence of cancer in the region, accessibility of health care, and positive changes in regulations. For example, regulatory agencies such as National Medical Products Administration (NMPA) greatly speed up the approval of new medical products, which results in a rapid increase in the number of clinical trials of cell and gene therapy for oncology in China. In a similar way, Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) provides conditional and time-limited approvals for regenerative medicines, which help to facilitate commercialization. The Asia Pacific region also boasts active research communities such as those at Peking University Cancer Hospital and National Cancer Centre Japan that conduct cell and gene therapy for oncology trials.
Moreover, increased government spending (health expenditure in East Asia & Pacific reached ~6.8% of GDP in 2023, reflecting steady public investment growth) and the growth of local biopharma companies have enhanced the capability of manufacturing locally. Together with growing collaborations between multinational firms and regional firms, these trends are helping the Asia Pacific region become the fastest-growing in the global cell and gene therapy for oncology market.
Global Cell and Gene Therapy for Oncology Market Outlook for Key Countries
Why is the U.S. Leading Innovation and Adoption in the Cell and Gene Therapy for Oncology Market?
The U.S. has the potential for leadership in innovation and adoption in the field of cell and gene therapy for oncology due to its strong research ecosystem consisting of research institutions, biotechnology firms, and a robust healthcare delivery system (Such as the National Cancer Institute (NCI), MD Anderson Cancer Center, Memorial Sloan Kettering Cancer Center, and Johns Hopkins Medicine). These aspects facilitate quick technological advancements in the diagnosis and treatment processes. For instance, the Cancer Moonshot initiative helps accelerate the process of conducting research for personalized medicine for cancer patients to ensure that it is widely adopted in practice (Source: National Cancer Institute). Moreover, the increasing participation of genomic testing firms and the rising use of next-generation sequencing (NGS) are contributing to the market growth.
Is U.K. a Favorable Market for Cell and Gene Therapy for Oncology?
The U.K. represents an ideal environment for the cell and gene therapy for oncology market because of state-led programs that have contributed to the successful implementation of genetic testing in the country’s health care system. For example, NHS Genomics Medicine Service has been instrumental in ensuring the provision of genetic testing throughout the country. In addition, the emergence of genomic laboratory hubs is contributing to widespread acceptance of genomic tests across clinical settings. Programs such as Genome U.K. (aiming to sequence up to 5 million genomes) are accelerating precision medicine adoption, strengthening the U.K.’s position in cell and gene therapy for oncology.
Is China Emerging as a Key Growth Hub for the Cell and Gene Therapy for Oncology Market?
China stands out as one of the most prominent countries with respect to the future growth of the cell and gene therapy for oncology market owing to country's huge patient base, and government’s emphasis on precision medicine. This is mainly due to investments being made in the area of genomics in various projects like the China Precision Medicine Initiative (with planned funding of CNY 60 billion, equivalent to USD 9–9.2 billion over 15 years, making it one of the largest precision medicine programs globally). Moreover, the growth of sequencing firms is contributing to increased availability of genomic cancer tests for patients. Further adoption of next-generation sequencing (NGS) in early cancer detection and diagnosis is boosting the demand for cell and gene therapies.
Why Does Germany Top the Europe Cell and Gene Therapy for Oncology Market?
Germany is the leader in the Europe market for cell and gene therapy for oncology due to its favorable reimbursement environment, efficient healthcare setup, and early introduction of personalized oncology treatments compared to other countries in Europe. The country has created highly structured molecular diagnostic networks through reimbursement approaches, including "Neue Untersuchungs- und Behandlungsmethoden (NUB)", and projects such as the National Decade Against Cancer have made significant contributions toward the implementation of advanced diagnostic tests such as genomics. In addition, Germany has several research centers and institutions specialized in cancer treatment (such as German Cancer Research Center – DKFZ, Charité – Universitätsmedizin Berlin, University Hospital Heidelberg, and the National Center for Tumor Diseases – NCT), which apply the latest technology like next-generation sequencing.
Is Cell and Gene Therapy for Oncology Market Developing in Japan?
The market for cell and gene therapy for oncology is rapidly growing in Japan with the aid of favorable initiatives by the government (such as the Cancer Genomic Medicine Initiative and the National Cancer Center’s nationwide genomic profiling network), as well as integration into its healthcare system. The Japan government had started covering the cost of cancer gene panel testing under the national health insurance since 2019; hence, wider adoption within hospitals has become possible. In addition, there is a presence of solid infrastructure for the development of genomics in Japan due to the establishment of the Center for Cancer Genomics and Advanced Therapeutics (C-CAT). This center has gathered vast information on genomics within a huge number of patients for use in research and clinical decisions.
Regulatory Landscape Governing the Global Cell and Gene Therapy for Oncology Market
|
Region
|
Key Regulatory Bodies |
Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration |
Biologics License Application (BLA); RMAT (Regenerative Medicine Advanced Therapy); Fast Track & Breakthrough Therapy |
Highly structured and innovation-friendly environment with expedited approvals accelerating oncology CGT commercialization |
|
Europe |
European Medicines Agency |
Advanced Therapy Medicinal Products (ATMP) regulation; PRIME (Priority Medicines) scheme |
Harmonized but stringent framework ensuring safety; supports innovation via accelerated access programs |
|
Asia Pacific |
Pharmaceuticals and Medical Devices Agency, National Medical Products Administration |
Sakigake designation (Japan); dual-track regulation & IND pathway (China); accelerated approvals for unmet needs |
Fastest evolving regulatory ecosystem with growing approvals and supportive policies boosting oncology CGT pipeline |
|
Latin America |
National agencies (e.g., ANVISA, COFEPRIS) |
Case-by-case approvals; adaptive regulatory pathways |
Gradual regulatory strengthening; dependency on global clinical data and external approvals for oncology CGT |
|
Middle East & Africa |
National regulatory bodies (e.g., SFDA, SAHPRA) |
Emerging frameworks; reliance on international approvals (FDA/EMA) |
Developing market with slower approvals but increasing adoption via regulatory reliance pathways |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is expansion of cell and gene therapies for solid tumor treatment creating new growth opportunities in the global cell and gene therapy for oncology market?
The expansion of cell and gene therapies products into the solid tumor space is generating considerable growth opportunities due to the creation of a far wider customer base and fostering the development of next-generation innovations outside the realm of hematologic cancers. Solid tumors represent the majority of cancers, and new innovations are showing the viability of cell and gene therapies (CGT) applications in this field. For instance, in February 2024, the U.S. Food and Drug Administration granted accelerated approval to lifileucel (Amtagvi), a tumor-infiltrating lymphocyte (TIL) therapy developed by Iovance Biotherapeutics, for patients with unresectable or metastatic melanoma who had progressed after prior treatments. (Source: National Cancer Institute)
Furthermore, clinical study published by Alliance for Cancer Gene Therapy, Inc., demonstrated significant anti-tumor effects, with reported response rates exceeding 30% among patients with advanced-stage melanomas, thus proving its efficacy as a treatment modality. (Source: Alliance for Cancer Gene Therapy, Inc.) Moreover, the approval of engineered T-cell therapies to treat synovial sarcomas, along with numerous CAR-T clinical trials involving the treatment of lung and breast cancers, is a strong indication of the rapid pipeline expansion within the solid tumor space.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In March 2026, Merck entered into a Memorandum of Understanding (MoU) with Cyto-Facto to enhance its cell and gene therapy manufacturing expertise in the Asia Pacific (APAC) region. The MoU is aimed at developing its lentiviral vector manufacturing process and optimizing its manufacturing processes for the next generation of therapies that will be used for oncology.
- In January 2026, Siren Biotechnology made an announcement regarding the U.S. FDA approval for their first IND application. This event marks the company’s shift towards the clinical development stage. The drug is meant to enhance the use of AAV immuno-gene therapy to treat recurrent high-grade glioma, which is one of the most aggressive types of brain cancer. The developments emphasize an increase in innovation in oncology-based gene therapies, as well as more regulatory backing for next generation cancer treatments.
Competitive Landscape
The global cell and gene therapy for oncology market is highly competitive owing to continuous innovation in CAR-T, gene editing, and next-generation therapies. Major biopharmaceutical companies, as well as up-and-coming biotechnology players, are concentrating their efforts on increasing their pipeline of clinical candidates as well as bringing advanced therapies to the market. Market dynamics are being shaped through strategic collaborations and licensing partnerships. Companies demonstrating successful clinical outcomes, scalability in production, and efficient logistics costs are gaining an advantage. Key focus areas include:
- Advancement of allogeneic (“off-the-shelf”) cell therapy platforms
- Expansion of CGT applications into solid tumors
- Strengthening manufacturing capabilities and global supply chain networks
- Strategic collaborations with CDMOs, research institutes, and healthcare providers
- Integration of gene editing and next-generation vector technologies
- Focus on cost optimization and improving therapy accessibility
Market Report Scope
Cell and Gene Therapy for Oncology Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8,641.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 44.7% | 2033 Value Projection: | USD 138,425.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Gilead Sciences, Novartis AG, Bristol Myers Squibb, Johnson & Johnson, Legend Biotech, bluebird bio, CRISPR Therapeutics, Allogene Therapeutics, Iovance Biotherapeutics, and Adaptimmune Therapeutics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the cell and gene therapy for oncology market will undergo transformation from limited and expensive individualized therapy to scalable next-gen CGT platforms, including off-the-shelf allogeneic cellular therapies and gene-edited immune therapies. While the current trend in terms of market expansion is the use of CAR-T in hematologic cancers, the future lies in the development of these CGTs for solid tumors, further development of gene editing technology, and more efficient manufacturing process.
- The biggest opportunity would come in the allogeneic cell therapy market in the U.S. and China due to the presence of robust clinical pipelines, favorable regulatory policies, and manufacturing abilities. Apart from that, there are also some emerging markets such as Asia Pacific, which have shown immense potential for growth due to the government support towards biomanufacturing operations and approvals for advanced therapies.
- For gaining any competitive advantage in the market, companies in the market should look beyond the initial success in the clinical stage and work towards industrializing the process of CGT delivery. This will involve creating a modular manufacturing network that is able to cope with autologous and allogeneic manufacturing processes and decrease the vein-to-vein period along with minimizing batch differences. The utilization of automation, closed systems, and tracking (chain of identity/ chain of custody) should also be considered.
Market Segmentation
- Therapy Type Insights (Revenue, USD Mn, 2021 - 2033)
- Cell Therapy
- Stem Cell Therapy
- T-Cell Therapy
- Mesenchymal Stem Cell (MSC) Therapy
- Others
- Gene Therapy
- Gene Induced Immunotherapy
- Oncolytic Virotherapy
- Therapy Gene Transfer
- Cell Therapy
- Technology Insights (Revenue, USD Mn, 2021 - 2033)
- Cell Therapy Technology
- Autologous
- Allogeneic
- Xenogeneic
- Gene Therapy Technology
- Viral Vector-Based Gene Therapy
- Non-Viral Vector-Based Gene Therapy
- Cell Therapy Technology
- Indication Insights (Revenue, USD Mn, 2021 - 2033)
- Breast Cancer
- Lung Cancer
- Prostate Cancer
- Leukaemia and Lymphoma
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals and Cancer Treatment Centers
- Research and Academic Institutes
- Biotechnology and Pharmaceutical Companies
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Gilead Sciences
- Novartis AG
- Bristol Myers Squibb
- Johnson & Johnson
- Legend Biotech
- bluebird bio
- CRISPR Therapeutics
- Allogene Therapeutics
- Iovance Biotherapeutics
- Adaptimmune Therapeutics
Sources
Primary Research Interviews
- Senior executives and R&D heads from cell and gene therapy companies (e.g., CAR-T developers, viral vector manufacturers, CGT-focused biotech firms)
- Clinical oncologists, hematologists, and transplant specialists from leading cancer treatment centers administering CAR-T and other cell therapies
- Regulatory affairs experts and consultants specializing in advanced therapy medicinal products (ATMPs) and CGT approvals
- CRO executives and manufacturing specialists involved in CGT clinical trials, viral vector production, and cell therapy scale-up
Stakeholders
- Cell and Gene Therapy for Oncology Solution Providers: (e.g., CAR-T developers, viral vector manufacturers, contract development and manufacturing organizations (CDMOs), CGT platform technology providers)
- End-use Sectors:
- Biotechnology & Pharmaceutical Companies
- Hospitals and Cancer Treatment Centers
- Academic and Research Institutes
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – CGT approvals and RMAT designations, European Medicines Agency (EMA) – ATMP evaluation, National Medical Products Administration (NMPA, China) – CGT approvals, Pharmaceuticals and Medical Devices Agency (PMDA, Japan) – regenerative medicine approvals, World Health Organization (WHO) – global cancer burden data
Databases
- ClinicalTrials.gov – global CGT oncology clinical trials database
- WHO Global Cancer Observatory (GCO) – cancer incidence and prevalence data
- FDA Cellular & Gene Therapy Products Database – approved CGT therapies
- EMA ATMP Database – advanced therapy approvals in Europe
- OECD Health Statistics – healthcare expenditure and oncology treatment data
Magazines
- Nature Reviews Drug Discovery – CGT innovations and oncology therapeutics
- Nature Biotechnology – gene therapy and cell therapy advancements
- BioPharma Dive – industry developments in CGT commercialization
- Genetic Engineering & Biotechnology News (GEN) – CGT research and industry trends
Journals
- The Lancet Oncology
- Journal of Clinical Oncology
- Nature Medicine
- Molecular Therapy
- Blood (American Society of Hematology)
Newspapers
- Financial Times – biotech investments and CGT deals
- The Wall Street Journal – pharmaceutical and CGT industry developments
- The New York Times (Health) – oncology breakthroughs and advanced therapies
- The Guardian (Science & Health) – global healthcare and innovation coverage
Associations
- American Society of Clinical Oncology (ASCO)
- European Society for Medical Oncology (ESMO)
- Alliance for Regenerative Medicine (ARM)
- International Society for Cell & Gene Therapy (ISCT)
- American Association for Cancer Research (AACR)
Public Domain Sources
- National Institutes of Health (NIH) – CGT research funding and clinical studies
- National Cancer Institute (NCI) – CAR-T and immunotherapy research initiatives
- U.S. FDA – CGT approvals and regulatory frameworks
- European Commission – Horizon Europe funding for advanced therapies
- World Health Organization (WHO) – global cancer statistics and policy frameworks
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients