Global Chronic Spontaneous Urticaria Market Size and Forecast – 2026-2033
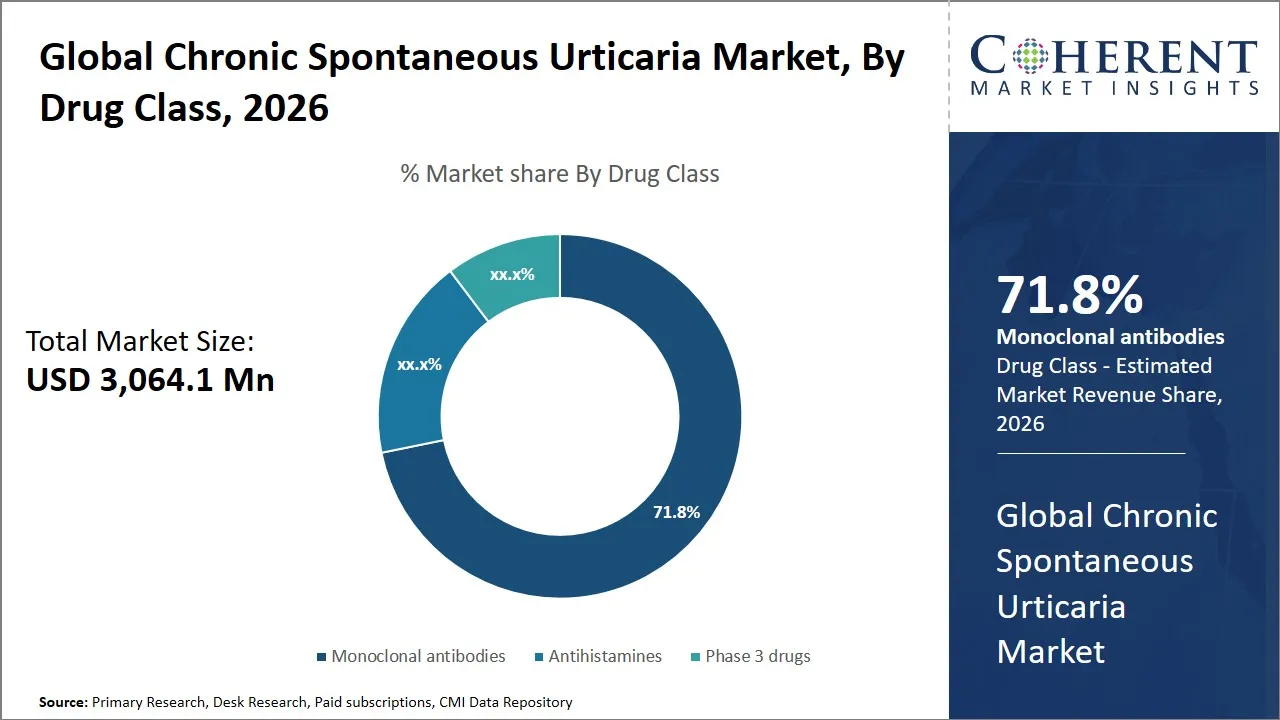
The global chronic spontaneous urticaria market is estimated to be valued at USD 3,064.1 Mn in 2026 and is expected to reach USD 4,657.1 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 9% from 2026 to 2033. This steady growth reflects increasing awareness of chronic spontaneous urticaria, advancements in treatment options, and rising prevalence rates globally, driven by environmental and lifestyle factors.
Key Takeaways of the Global Chronic Spontaneous Urticaria Market
- Monoclonal antibodies segment is expected to lead the global chronic spontaneous urticaria market, capturing 71.8% share in 2026.
- Branded segment is estimated to represent 81.9% of the global chronic spontaneous urticaria market share in 2026.
- Injectable segment is projected to dominate with 80.4% of the global chronic spontaneous urticaria market share in 2026.
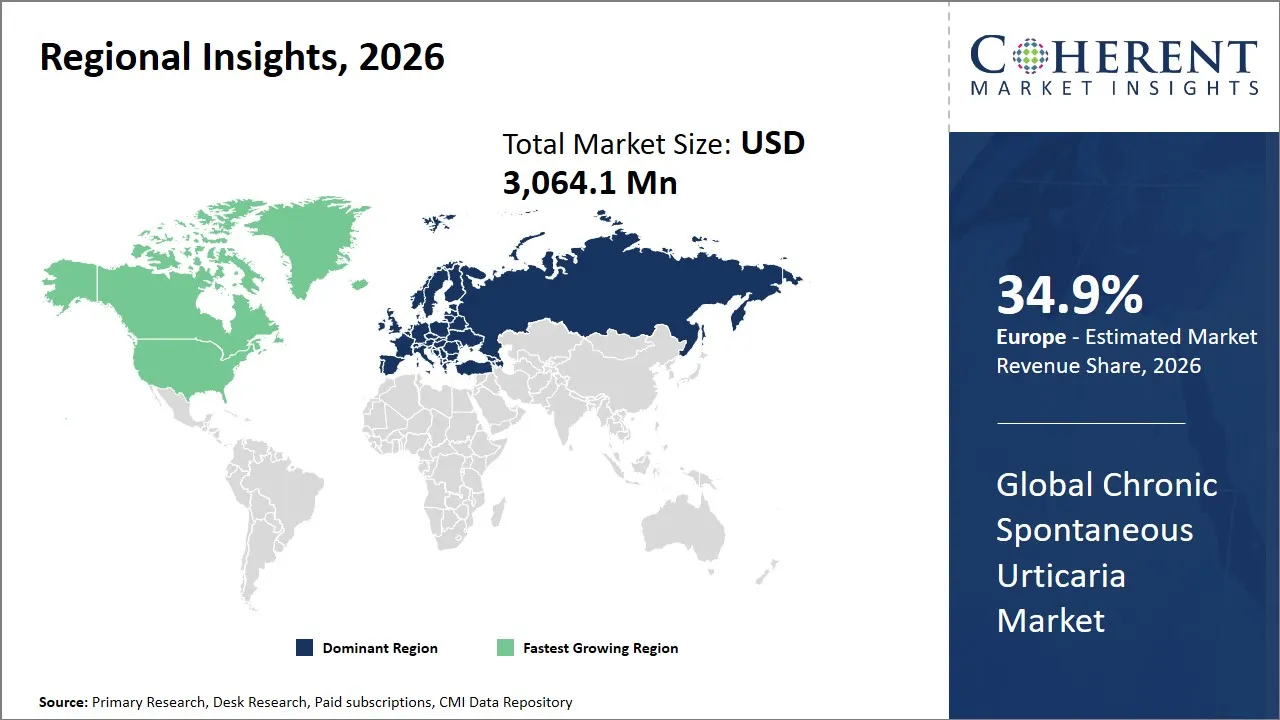
- Europe is expected to lead the market, holding a share of 34.9 in 2026. North America is anticipated to be the fastest-growing region, with 34.3% share in 2026.
Market Overview
- Chronic Spontaneous Urticaria (CSU) is a chronic autoimmune dermatological condition characterized by spontaneous hives and angioedema, affecting quality of life and daily functioning.
- The market is primarily driven by increasing prevalence of Chronic Spontaneous Urticaria (CSU), rising patient awareness, and growing adoption of biologic therapies.
- The growing adoption of mail-order and e-pharmacy services has positioned central fill facilities as critical nodes in the prescription distribution network.
- The market is witnessing strong innovation, including oral small molecules, anti-IgE therapies, and cytokine-targeting agents.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Approval of Novel Therapies |
|
|
Strategic Acquisitions |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Monoclonal antibodies Segment Dominate the Global Chronic Spontaneous Urticaria Market in 2026?
The monoclonal antibodies segment is expected to hold the largest market share of 71.8% in 2026, driven by their targeted mechanism of action and strong clinical efficacy. These biologic medicines, including Xolair (omalizumab) and Dupixent (dupilumab), represent an important advancement to the conventional antihistamines and provide relief to patients with moderate to severe CSU who fail to respond to first-line therapy. The specificity at which monoclonal antibodies can interact with certain immune pathways implicated in the pathology of CSU: primarily IgE-mediated allergic reactions, is beneficial in the effectiveness of their therapeutic applications.
For instance, in September 2025, Mabgeek Biotech’s innovative MG‑K10 humanized monoclonal antibody, designed to block IL‑4 and IL‑13 signaling with an extended dosing interval, received clinical trial approval from China’s National Medical Products Administration to begin Phase III studies for chronic spontaneous urticaria in adult patients.
(Source: Mabgeek Biotech)
Why Does the Branded Segment Dominate the Global Chronic Spontaneous Urticaria Market in 2026?
The branded segment is expected to hold 81.9% of the market share in 2026, driven predominantly by the perceived reliability, efficacy, and innovation associated with proprietary medications. Branded drugs, manufactured and marketed by original pharmaceutical companies, often come with rigorous clinical validation, robust post-market surveillance, and comprehensive patient support programs. These are some of the reasons why healthcare providers will prefer branded products to generics, particularly the treatment of a complex and chronic condition like CSU when treatment response may be very personalized.
Patient confidence is achieved through brand recognition, which fosters adherence and frequent follow-up, which are essential in the management of a condition where the symptoms are unpredictable. Additionally, branded drugs are often the result of high-investment research, with a combination of new mechanism of action and improved safety compared to previous therapy. The innovation provides differentiation and rationalizes their premium positioning in the market.
For instance, in March 2025, Celltrion, Inc announced that the U.S. Food and Drug Administration (FDA) has approved OMLYCLO (omalizumab‑igec) as the first interchangeable biosimilar referencing Xolair (omalizumab), a leading branded anti‑IgE biologic used to treat chronic spontaneous urticaria (CSU) alongside other allergic and immunologic conditions. This interchangeability designation means OMLYCLO can be substituted for Xolair at the pharmacy level where permitted, broadening access to a key biologic therapy and potentially helping reduce treatment costs while maintaining equivalent therapeutic effectiveness.
(Source: Celltrion, Inc)
The Injectable Segment Dominates the Global Chronic Spontaneous Urticaria Market
The injectable segment is expected to hold the largest market share of 80.4% in 2026, largely because of their direct and efficient mode of drug delivery. This is the formulation that combines the subcutaneous and intravenous injection, which allow bioaugmenting the biologics to overcome the digestive degradation that would otherwise lead to the loss of bioavailability and rapid therapeutic effect, namely monoclonal antibodies. Injectable therapies are also beneficial to patients managing severe and persistent symptoms, which may respond more rapidly to control a symptom than oral formulations.
The favorable aspect of injectables are also due to the nature of the drugs themselves with many monoclonal antibodies and more recent phase 3 pipeline molecules being designed to be parenterally administered in order to preserve molecular stability and potency. Injectable formulations ensure that the dosage is administered at controlled dosing schedules (which in many cases involve the utilization of injection every few weeks); this increases dose compliance and decreases treatment burden in patients.
Pipeline Developments and Emerging Therapies in the Global Chronic Spontaneous Urticaria Market
- The market of global chronic spontaneous urticaria is also experiencing high growth in its pipeline with more than 25 therapeutic candidates in the clinical and preclinical phases. They are oral small molecules and next-generation biologics that are aimed to address the major immune pathways implicated in CSU like mast cell activation and inflammatory signaling. Such high-profile candidates as remibrutinib, a Bruton tyrosine kinase inhibitor, and barzolvolimab, a monoclonal antibody, are under development to offer an alternative to patients who failed with standard antihistamines, with faster response and prolonged symptom management.
- Also anticipated in favor of innovation is the growing number of approved biologics indications such as dupilumab, which indicates a wider shift towards precision immunotherapy in the care of CSU. New anti-IgE-based therapy, cytokine-based therapy, and new receptor therapies are anticipated to broaden therapeutic choice, enhance clinical performance and generate competitive distinction among the biopharmaceutical firms in this niche chronic spontaneous urticaria market.
Regional Insights
To learn more about this report, Request Free Sample
Europe Chronic Spontaneous Urticaria Market Analysis and Trends
The Europe region is projected to lead the market with a 34.9% share in 2026, fueled by expanding healthcare access, rising medical awareness, and increasing incidence of autoimmune and allergic disorders. The market is changing with a growing use of precision biologic and sophisticated treatment of patients whose symptoms cannot be restrained with regular antihistamines. The increasing number of treatment options, regulatory acceptance, and physician awareness are leading to the increased access of patients in the most important EU nations, and the gaps in the actual treatment indicate the necessity to continue the innovation and adopt branded therapies.
For instance, on February 27, 2026, Novartis International AG announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion recommending marketing authorization for remibrutinib as a targeted oral treatment for chronic spontaneous urticaria (CSU) in adults whose symptoms persist despite H1‑antihistamine therapy. The CHMP recommendation represents a major step toward formal approval and commercialization of what could be the EU’s first BTK‑inhibitor therapy for CSU.
(Source: Novartis International AG)
North America Chronic Spontaneous Urticaria Market Analysis and Trends
The North America region is expected to exhibit the fastest growth in the chronic spontaneous urticaria market, contributing 34.3% share in 2026, primarily driven by a highly developed healthcare infrastructure, widespread awareness about dermatological conditions, and significant investments in pharmaceutical research and development. As the biggest contributor in the region, the U.S. enjoys good government programs that promote treatment of rare and chronic diseases, in addition to having a well-developed chain of specialty clinics and allergists.
Also, the existence of the major pharmaceutical industries including Novartis, Pfizer and Sanofi, which are also investing heavily in CSU therapies, strengthens the dominant position of the region. The market dominance is further enhanced by strong patent legislation in the region and reimbursement policies, which promote innovative treatments and guarantee the access of patients to highly advanced therapies.
For instance, on February 26, 2026, Celldex Therapeutics launched Barzolvolimab, a branded monoclonal antibody for chronic spontaneous urticaria (CSU) in antihistamine‑refractory patients, expanding advanced therapy options and increasing competition in the North America chronic spontaneous urticaria market.
(Source- Celldex Therapeutics)
Global Chronic Spontaneous Urticaria Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Chronic Spontaneous Urticaria Market?
The market of chronic spontaneous urticaria in the U.S. is the most developed and competitive in the world, which is facilitated by the high healthcare expenditure and the high level of the development of healthcare delivery. Large competitors like Novartis with their biologic therapies and Pfizer with antihistamine choices have large market shares. The U.S has the advantage of facilitated regulatory approvals through the FDA and comprehensive clinical research efforts that facilitate the development of new treatment modalities. Also, the advocacy and awareness among patients in CSU helps to achieve the timely uptake of treatment.
How is Germany Helping in the Growth of the Chronic Spontaneous Urticaria Market?
Germany is a major chronic spontaneous urticaria market within Europe, which is supported by a powerful pharmaceutical manufacturing sector, as well as by a widespread healthcare cover. The attention of the government to rare and chronic illnesses makes CSU treatment to be a common occurrence with statutory health insurance. Research and therapeutic developments in the field are being done by pharmaceutical giants such as Bayer and Boehringer Ingelheim that operate in the dermatology segment. In addition, trade dynamics, such as high export potential, within Germany contribute to the spread of innovations in the markets of the rest of Europe.
Key Drivers for the Growth of the China Chronic Spontaneous Urticaria Market
The chronic spontaneous urticaria market in China is fast developing following the growth of healthcare facilities and rise in patient count with allergic diseases. Multinational firms like GlaxoSmithKline and AstraZeneca are actively involved in the country as they are collaborating with domestic companies to develop and distribute drugs. The government can help in accelerating the introduction of new CSU therapies by supporting the reforms in regulations and innovation incentives. Also, the increasing disposable income and increasing awareness in patients encourages them to adopt treatment, albeit in areas like rural health care accessibility.
India Chronic Spontaneous Urticaria Market Trends
The chronic spontaneous urticaria market in India is in a rise because of availability of better healthcare and increasing prevalence of chronic skin conditions. The growing role of the private sector and the focus of the government on the universal health coverage promote and hasten the market potential. Such firms as Sun Pharmaceutical and Cipla play an important role since they specialize in low-cost antihistamines and new treatments that consider the specific demands of the region. The digital health industry and telemedicine are allowing more and more patients and doctors to interact with each other, which has resulted in higher diagnosis rates and adherence to treatment.
Stepwise Treatment Pathway and Therapeutic Strategies in the Global Chronic Spontaneous Urticaria Market
- The management of Chronic Spontaneous Urticaria (CSU) is based on the first-line therapy with second-generation H1-antihistamines that are recommended to all patients and help to manage itch and hives. In case of failure of proper symptom relief, clinicians tend to increase doses or introduce adjunctive measures, like H2-antihistamines, leukotriene receptor antagonists or brief courses of corticosteroids to treat acute flares. The response of the patient is routinely checked and adjustment of the intensity of therapy is done to ensure that the symptoms are kept in check without affecting the side effects.
- For symptomatic patients not responding to these treatments, there should be second- or third-line therapies with targeted biologic therapies such as omalizumab (anti-IgE) and dupilumab (anti-IL-4/anti-IL-13). Simultaneously, new treatments that are in clinical trials, like remibrutinib and barzolvolimab, will increase the number of new treatment alternatives in refractory cases. In general, the treatment pathway represents an individual, progressive course of treatment, focusing on the suppression of symptoms, quality of life, and the implementation of new immune modulating therapies as they become accessible.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On January 20, 2026, GlaxoSmithKline announced it will acquire RAPT Therapeutics for USD 2.2 billion, gaining ozureprubart, a Phase IIb anti-IgE antibody. The deal strengthens GlaxoSmithKline’s immunology pipeline with therapies relevant to allergy and immune modulation, including potential applications in CSU. The acquisition is expected to close in Q1 2026.
- In September 2025, the U.S. FDA approved Novartis’ Rhapsido, the first oral Bruton's tyrosine kinase inhibitor, for adult CSU patients unresponsive to antihistamines. Clinical trials showed rapid and significant reduction in itch and hives. The oral therapy offers a convenient, non-injectable option with no routine lab monitoring required.
- In April 2025, Sanofi announced that the U.S. Food and Drug Administration (FDA) has approved Dupixent (dupilumab) for the treatment of chronic spontaneous urticaria (CSU) in adults and adolescents aged 12 years and older who remain symptomatic despite H1 antihistamines. Approval follows Phase 3 LIBERTY‑CUPID trials demonstrating significant reductions in itch and hives, expanding Sanofi’s targeted biologic options for CSU patients with limited treatment choices.
Top Strategies Followed by Global Chronic Spontaneous Urticaria Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established companies dominate the chronic spontaneous urticaria market through heavy R&D investments in advanced biologics and targeted therapies, strategic partnerships, and global expansion into emerging markets. |
Roche, via Genentech, strengthens its chronic spontaneous urticaria market position with Xolair, co-developed with Novartis. Its strategy combines biologics R&D, strategic partnerships, and global expansion to sustain market dominance. |
|
Mid-Level Players |
Mid-level chronic spontaneous urticaria market players focus on cost-effective therapies for price-sensitive patients, often collaborating with CMOs and regional distributors to expand reach and adopt new technologies. |
Harrington Pharmaceuticals developed AllerBloc, an affordable oral therapy for antihistamine-refractory chronic spontaneous urticaria, targeting broader access in North America and emerging markets. |
|
Small-Scale Players |
Small-scale chronic spontaneous urticaria players focus on niche, innovative therapies for underserved patient subgroups, often using personalized medicine or digital health tools to differentiate. They form local partnerships with manufacturers, startups, or academia to accelerate development, expand distribution, and build brand recognition, staying agile and serving as incubators for ideas later scaled by larger firms. |
ImmunoTech Labs partnered with a regional biotech to develop a personalized chronic spontaneous urticaria diagnostic and treatment tool for patients unresponsive to standard therapies, targeting unmet needs in select European markets and advancing precision medicine in the segment. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Chronic Spontaneous Urticaria Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3,064.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9% | 2033 Value Projection: | USD 4,657.1 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Novartis AG, Sanofi S.A, Regeneron Pharmaceuticals Inc, GlaxoSmithKline plc, Pfizer Inc, Genentech Inc (Roche Group), Merck & Co Inc, Johnson & Johnson Services Inc, AstraZeneca plc, Bayer AG, Teva Pharmaceutical Industries Ltd, AbbVie Inc, Amgen Inc, Sun Pharmaceutical Industries Ltd, and UCB S.A |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Chronic Spontaneous Urticaria Market Dynamics
To learn more about this report, Request Free Sample
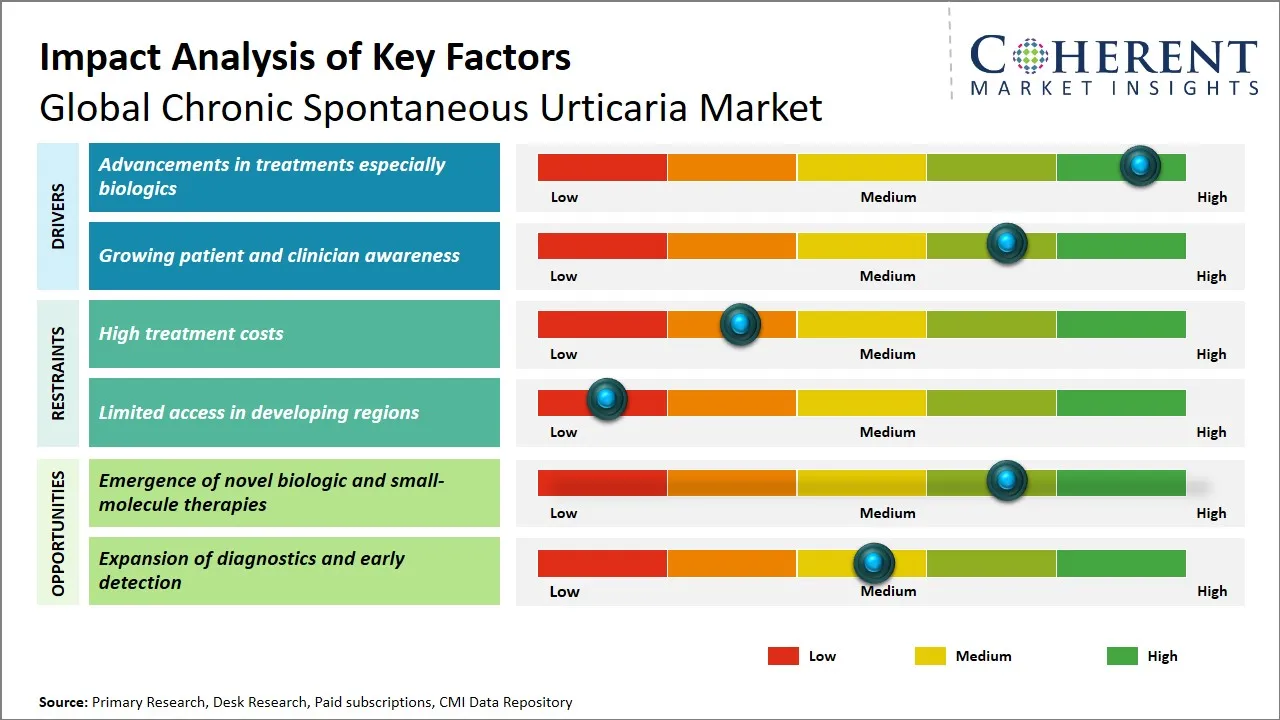
Global Chronic Spontaneous Urticaria Market Driver - Advancements in Treatments Especially Biologics
The chronic spontaneous urticaria market is driving immensely due to impressive developments in treatments especially the introduction of biologic treatments, which have transformed patient management. Conventional therapy has been concentrated on providing symptomatic management using antihistamines, however with the emergence of specific biologics, including monoclonal antibodies, the target of therapy has changed wherein much attention is being given to the underlying pathophysiology of the disease. These biologics are more effective in the reduction of the hives, angioedema symptoms in patients who do not respond to traditional therapies and thus enhances the quality of life and compliance to regimens.
Moreover, continued research and clinical trials are expanding the pipeline of biologic agents of improved safety profiles and long-term outcomes with the aim of increased acceptance by the healthcare community. Market momentum is further being created by the growing awareness and acceptance of these sophisticated treatments as doctors and patients pursue more effective ways of treating chronic spontaneous urticaria, which ultimately leads to innovation and the expansion of the treatment paradigm in the dermatology and immunology field.
For instance, in October 2023, UCB announced the U.S. launch and regulatory approval of Bimzelx for the treatment of moderate-to-severe plaque psoriasis, marking the first biologic designed to selectively inhibit both IL-17A and IL-17F cytokines. The approval was supported by multiple Phase 3 clinical trials demonstrating strong and sustained skin clearance outcomes. Subsequent label expansions in 2024 further extended its use across additional inflammatory conditions, reinforcing its clinical value and commercial rollout.
(Source: UCB)
Global Chronic Spontaneous Urticaria Market Opportunity - Emergence of Novel Biologic and Small-Molecule Therapies
Global chronic spontaneous urticaria market is a major growth point with the rapid creation and launch of new biologic and small-molecule treatment. Conventional interventions on CSU including antihistamines and corticosteroids are likely to offer small degrees of relief and are linked with side effects which render unmet medical needs among patients. In their turn, pharmaceutical organizations are directing their research into innovative biologics, including monoclonal antibodies that target certain pathways in the pathophysiology of CSU, including IgE and interleukin receptors. Such specific treatments have a better efficacy and safety profile, which may change the way of managing the disease among patients with moderate to severe symptoms.
Moreover, small-molecule drugs that control intracellular signaling pathways are also on the increase because of the convenience and oral bioavailability since it can improve patient compliance and quality of life. The CSU therapeutic pipeline is strong, and several of the candidates have reached their advanced clinical development stages, and can be granted regulatory approvals in the near future.
For instance, in October 2023, Amgen acquired Horizon Therapeutics to strengthen its rare disease and immunology portfolio, including biologic therapies targeting inflammatory conditions. This acquisition expands Amgen’s pipeline of advanced biologics and reinforces the broader industry trend toward developing novel targeted therapies for diseases with high unmet need, supporting continued innovation beyond traditional treatments.
(Source: Amgen)
Analyst Opinion (Expert Opinion)
- The global chronic spontaneous urticaria market is being influenced by an increase in patient awareness, prevalence, and new target therapy. Omalizumab and dupilumab are becoming increasingly adopted as biologics, and new oral agents like remibrutinib and barzolvolimab are increasing the number of patients who cannot respond to antihistamine. Earlier intervention is happening through regulatory approvals and broadened indications and an increased investment in research and development is speeding up the innovation. There are still difficulties in the management of refractory cases and access in emerging countries, yet there are promising opportunities of individualized therapies, digital monitoring of patients and the creation of real-world evidence to inform treatment plans.
- The conferences in the industry, such as the annual meeting of the American Academy of Allergy, Asthma and Immunology and the congress of the European Academy of Allergy and Clinical Immunology have been instrumental in sharing the knowledge and presenting new therapies as well as assisting in the development of clinical guidelines. Strategic acquisitions, biopharma to biotech collaborations and hospital programs that deploy centralized biologic therapy protocols are all market initiatives. These endeavors are enhancing patient results, reinforcing pipelines, and establishing the groundwork to keep on increasing chronic spontaneous urticaria market in the world.
Market Segmentation
- Drug Class Insights (Revenue, USD Mn, 2021 - 2033)
- Monoclonal antibodies
- Xolair
- Dupixent
- Antihistamines
- Phase 3 drugs
- Ligelizumab
- Remibrutinib
- Monoclonal antibodies
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Branded
- Generic
- Form Insights (Revenue, USD Mn, 2021 - 2033)
- Injectable
- Tablets
- Capsules
- Syrup
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Oral
- Subcutaneous
- Age Insights (Revenue, USD Mn, 2021 - 2033)
- Pediatric
- Adult
- Geriatric
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Novartis AG
- Sanofi S.A
- Regeneron Pharmaceuticals Inc
- GlaxoSmithKline plc
- Pfizer Inc
- Genentech Inc (Roche Group)
- Merck & Co Inc
- Johnson & Johnson Services Inc
- AstraZeneca plc
- Bayer AG
- Teva Pharmaceutical Industries Ltd
- AbbVie Inc
- Amgen Inc
- Sun Pharmaceutical Industries Ltd
- UCB S.A
Sources
Primary Research Interviews
Industry Stakeholders
- Dermatologists in major hospitals
- Biopharmaceutical R&D heads
- Clinical trial investigators
- Immunology specialists
- Hospital pharmacy directors
- Specialty care clinic managers
End Users List
- CSU patients enrolled in treatment programs
- Patient advocacy groups
- Chronic disease support groups
- Hospital outpatient pharmacy staff
- Primary care physicians treating CSU
- Specialty clinic nurses
Government and International Databases
- U.S. Food and Drug Administration (FDA)
- Centers for Disease Control and Prevention (CDC)
- European Medicines Agency (EMA)
- National Institutes of Health (NIH)
- World Health Organization (WHO)
- Health Canada
Trade Publications
- Journal of the American Academy of Dermatology
- Allergy and Clinical Immunology News
- Dermatology Times
- Immunology Today
- Clinical Dermatology Review
- Pharmaceutical Executive
Academic Journals
- Journal of Allergy and Clinical Immunology
- Clinical and Experimental Allergy
- International Archives of Allergy and Immunology
- The British Journal of Dermatology
- Allergy
- Annals of Allergy, Asthma & Immunology
Reputable Newspapers
- The New York Times – Health Section
- The Washington Post – Health
- The Guardian – Health
Industry Associations
- American Academy of Allergy, Asthma & Immunology (AAAAI)
- American Academy of Dermatology (AAD)
- European Academy of Allergy and Clinical Immunology (EAACI)
- International Dermatology Society (IDS)
- Global Allergy and Asthma Patient Platform (GAAPP)
- National Eczema Association
Public Domain Resources
- ClinicalTrials.gov
- WHO Global Health Observatory
- CDC National Health Statistics Reports
- FDA Drug Approval Database
- National Health ServiceTop of Form
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients