Global Human Biopreservation Market Size and Forecast – 2026-2033
The global human biopreservation market is expected to grow from USD 5,783.2 Mn in 2026 to USD 11,698.0 Mn by 2033, registering a compound annual growth rate (CAGR) of 12.3%. The global human biopreservation market is poised for significant expansion, fueled by the surging volume of clinical trials and biomedical research activities.
As per the data from ClinicalTrials.gov, there are more than 579,621 clinical trials registered in 225 different countries and territories reflecting the rising number of biological studies conducted globally.
(Source: ClinicalTrials.gov)
Key Takeaways of the Global Human Biopreservation Market
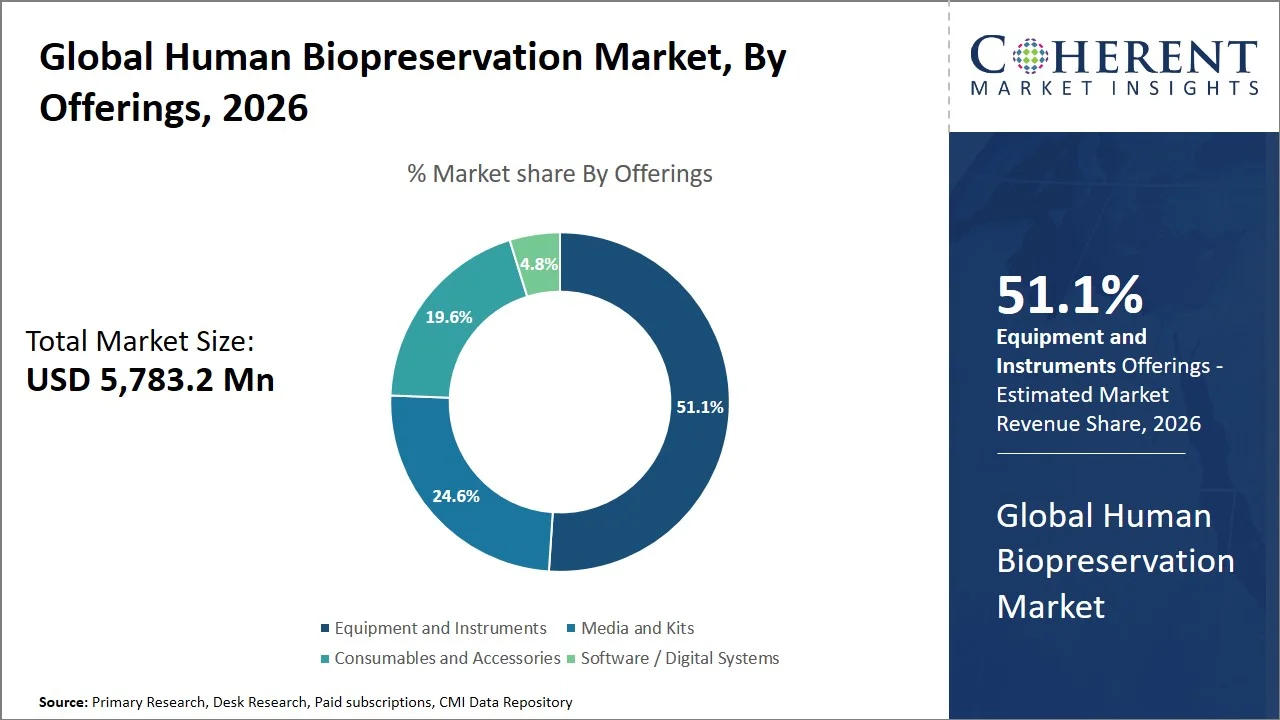
- Equipment and instruments are projected to hold 51.1% of the global human biopreservation market share in 2026, making it the dominant offerings segment. These offerings are used mostly in North America and Europe, as biobanks in these regions have advanced infrastructure and automation in place to maintain the integrity of samples. For example, major biobanks like the UK Biobank and All of Us Research Program use ultra-low temperature freezers as well as robots for storing samples in order to preserve quality over time.
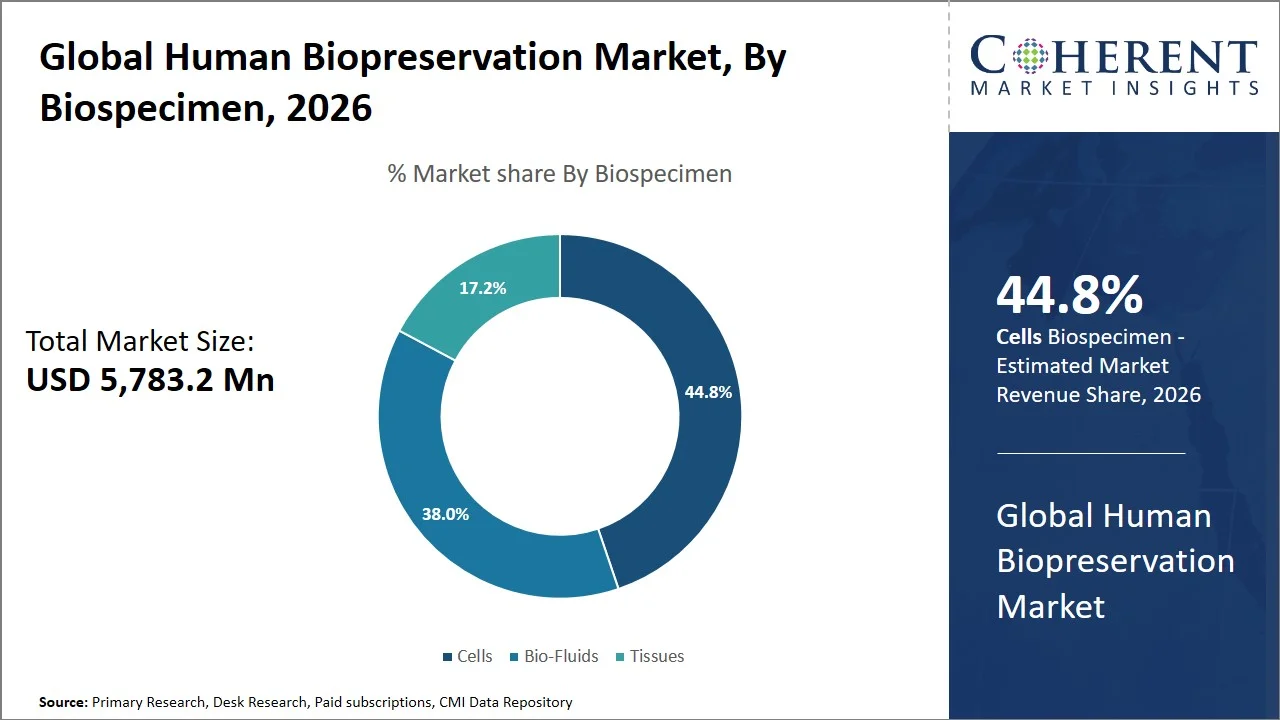
- Cells are projected to hold 44.8% of the global human biopreservation market share in 2026, making it the dominant biospecimen segment. Cell-based sample types are widely employed in North America and Asia Pacific due to significant activities in regenerative medicine and increasing cell therapies pipelines, especially in the U.S., China, and Japan. For example, there are about 2,700 clinical trials that utilize cell therapy in China alone, whereas over 500 stem cell clinical trials are currently taking place in Japan and the Asia Pacific region. (Source: National Library of Medicine)
- Cryopreservation segment is projected to hold 66.3% of the global human biopreservation market share in 2026, making it the dominant preservation method segment. This technique is widely favored in North America and Europe, where clinical and research purposes demand long-term, high viability storage techniques for biological materials.
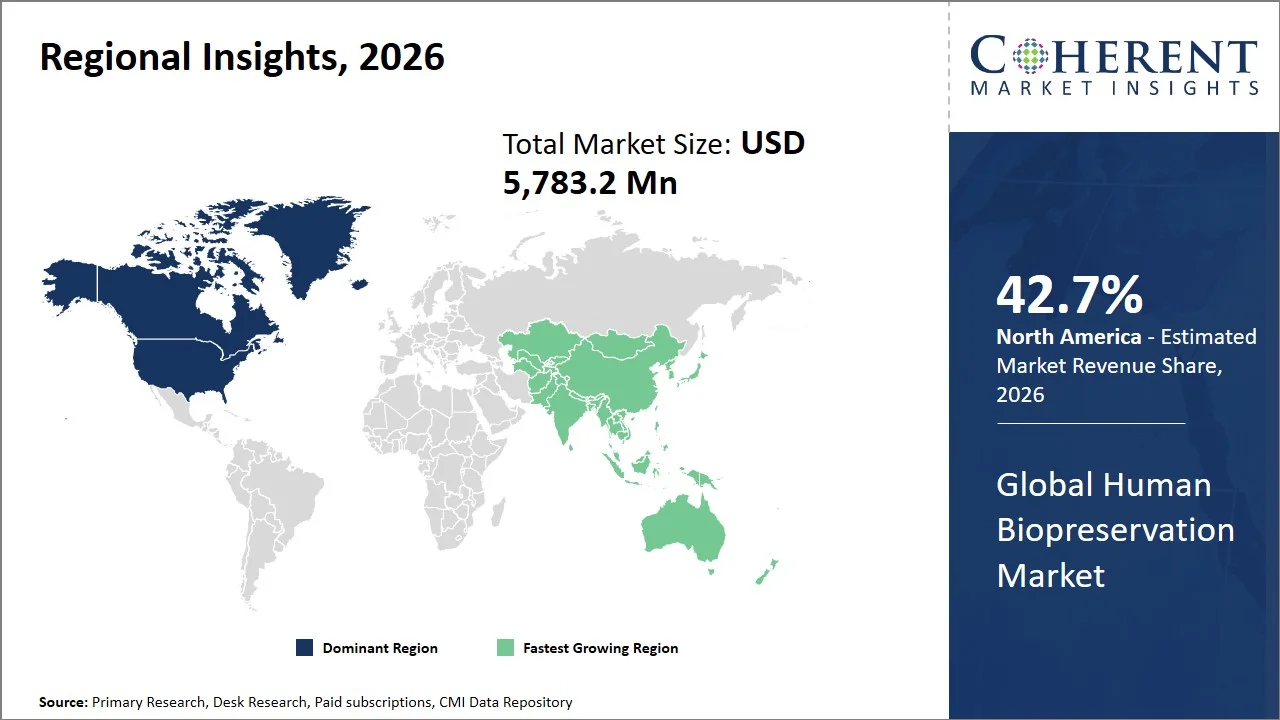
- The North America market maintains dominance with an expected share of 42.7%in 2026, bolstered by the region’s highly developed health care infrastructure, robust scientific research environment, and pioneering preservation techniques. The availability of biobanks in the region reinforces local demand.
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 21.0% share in 2026, owing to the growing sophistication of the biotechnology sector, rising clinical studies, and increasing interest from governments in genomics and precision medicine.
- Expansion of Personalized Medicine and Cell-Based Therapies: The rising application of personalized medicine approaches such as stem cells and genetic therapies has increased the demand for accurate biopreservation of biological samples. There is a specific market opportunity for innovative cryobiological techniques that guarantee optimal cell survival and treatment efficiency.
- Rising Demand for Fertility Preservation and Reproductive Biobanking: The rising popularity of fertility preservation programs among cancer patients and those at an advanced age for having children has increased the demand for gametes and embryos banking. There is a market opportunity for innovative cryobiological techniques to preserve biological samples.
Why Do Equipment and Instruments Dominate the Global Human Biopreservation Market?
Equipment and instruments are projected to hold the market share of 51.1% in 2026, because of the key function they play in ensuring proper temperature control and preservation of the biological specimens for longer periods of time. Biobanking and biopreservation initiatives by governments (e.g., UK Biobank, All of Us Research Program, GenomeIndia, China Kadoorie Biobank, FinnGen, and Qatar Biobank) have contributed to this demand. For instance, the UK Biobank has gathered and preserved more than 500,000 participant samples, which requires massive storage and management of the samples. (Source: UK Biobank)
Additionally, All of Us Research Program by National Institutes of Health, aims to collect and study health data from one million or more people living in the U.S. (Source: National Institutes of Health). Such programs indicate how biopreservation relies heavily on equipment, where cryogenic tanks, rate-controlled freezing chambers, and automated biobanks are needed in order to ensure that the specimens are preserved, thus making it a major player in the global human biopreservation market.
Why are Cells the Most Preferred Biospecimen?
To learn more about this report, Request Free Sample
Cells are projected to hold a market share of 44.8% in 2026, owing to its pivotal significance in furthering medical science research, especially in such areas like genomics, drug discovery, and personalized medicine. According to National Library of Medicine, biospecimens, like cells play a key role in the comprehension of how diseases develop and in finding targeted treatment approaches, through biobanks' capacity for conducting extensive analysis on cellular levels (Source: National Library of Medicine). Different reasons exist for the popularity of cells in experiments, starting with their capacity to multiply, be preserved for prolonged periods, and be used in repeated experiments unlike other biosamples. Moreover, cellular models play an integral role in creating treatments like gene therapy and immunotherapy, thus further establishing their significance as the most employed biological sample for both medical and experimental purposes.
Cryopreservation Segment Dominates the Global Human Biopreservation Market
The cryopreservation segment is projected to hold a market share of 66.3% in 2026, considering the importance of cryopreservation in terms of sustaining and guaranteeing efficiency and effectiveness of biological substances such as stem cells, reproductive cells and tissues during complicated procedures and researches.
Regenerative medicine and biobanks initiatives sponsored by governments have become an important aspect in popularizing the cryopreservation techniques. The National Health Service encourages the large-scale use of cryopreservation in fertility programs and stem cell preservation programs. Also, the initiatives managed by Health Resources and Services Administration include national programs in collecting umbilical cord blood banks that are crucial for transplants and researches. (Source: National Health Service)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory & Macro Shift: EU AI Act Impacting AI-enabled Biobanking & Digital Health (2024–2025) |
|
|
Geopolitical & Infrastructure Shift: Expansion of Biobanking and Cryogenic Storage Capacity (2024–2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: European Commission, UK Biobank Limited)
Global Human Biopreservation Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
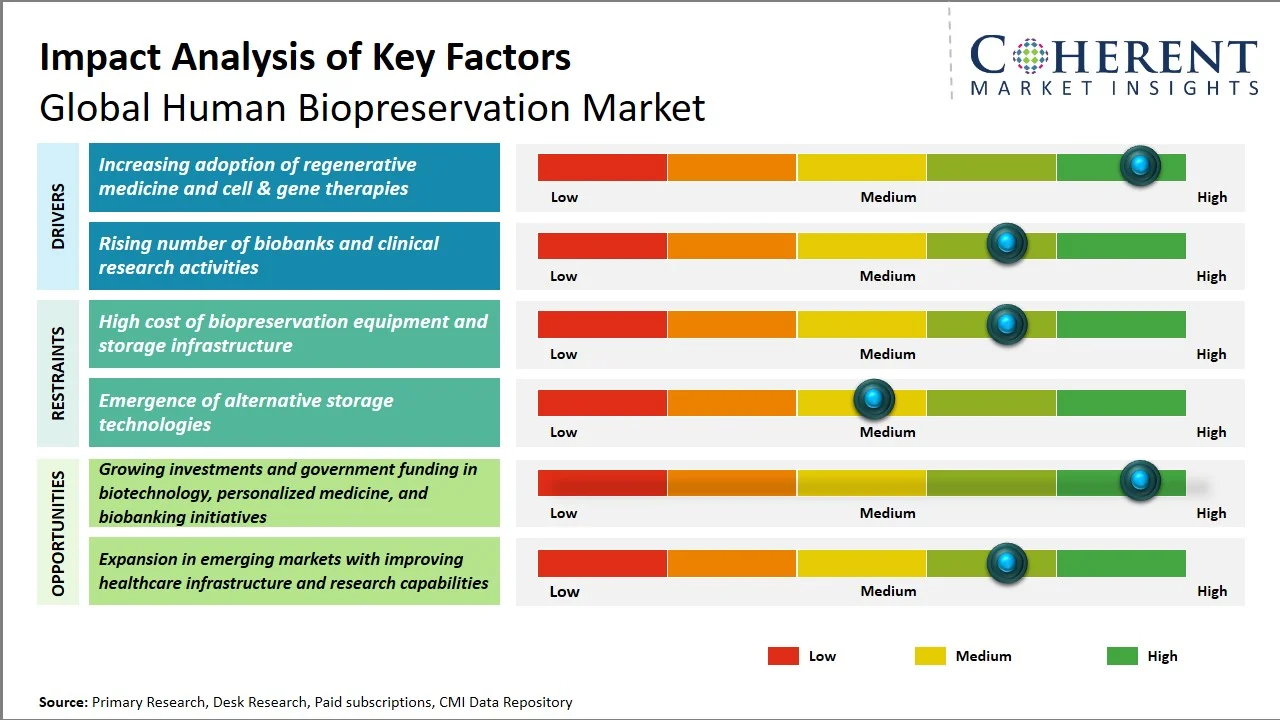
- Increasing adoption of regenerative medicine and cell & gene therapies: These innovative treatments demand meticulous preservation of living cells at every stage: during their creation, storage, and eventual transport. The U.S. Food and Drug Administration has observed the rapid advancement of cell and gene therapies, a trend reflected in the numerous products currently undergoing clinical trials and receiving regulatory clearance (Source: U.S. Food and Drug Administration). Furthermore, the National Institutes of Health reports more than 5,000 gene therapy clinical trials underway worldwide, a clear sign of the growing research focus (Source: National Library of Medicine). This boom in advanced therapies naturally boosts the demand for cryopreservation, controlled storage, and cold chain logistics. Consequently, biopreservation technologies are becoming essential for the successful development of these next-generation therapeutics.
- Rising number of biobanks and clinical research activities: The escalating proliferation of biobanks and clinical research endeavors worldwide is a key factor propelling the demand for human biopreservation solutions, given that large-scale investigations necessitate the systematic acquisition and prolonged storage of biological specimens. The ClinicalTrials.gov database, overseen by the U.S. National Library of Medicine, functions as a central repository for global clinical studies, thereby mirroring the persistent expansion of research activities on a global scale with over 2 million study records having posted cumulatively since its inception, reflecting sustained growth in clinical research submissions. Moreover, governmental genomic and biobanking initiatives are fostering a swift increase in the volume of stored biospecimens, with numerous studies mandating standardized preservation protocols. As a result, the increasing complexity of clinical trials and population health research has created a demand for automation, ultra-cold storage technology, and preservation techniques, leading to increased adoption of advanced biopreservation technology in academic and healthcare institutions.
Emerging Trends
- Advancements in Cryopreservation Technologies: The recent advancements in technology, including vitrification, cryo-storing technology, and controlled-rate freezing technology, have tremendously increased viability in the preservation process of cells and tissues. The need for advancements in these fields has been fueled by the increasing demand for cell and gene therapies.
- Expansion of Biobanking and Regenerative Medicine: The growing need for personalized and regenerative medicines has led to the expansion of biobanks around the world. This will create a further increase in the need for standardized biopreservation techniques.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Human Biopreservation?
North America leads the global human biopreservation market, accounting for an estimated 42.7% share in 2026, considering the abundance of biobanks, the presence of a well-developed scientific research infrastructure, and the existence of a vast biomedical ecosystem supported by government agencies.
The National Institutes of Health, along with other federal bodies, has facilitated the development of large-scale biological sample collection and storage. According to estimations, there are currently over 307 million biological specimens collected from approximately 178 million cases preserved within biobanks in the U.S., demonstrating the enormous need for preserving samples (Source: National Library of Medicine). Additionally, almost 46% of the world’s life sciences R&D expenditures take place in the U.S. This combination of factors makes North America a major and mature market for human biopreservation.
Why Does Asia Pacific Human Biopreservation Market Exhibit High Growth?
The Asia Pacific human biopreservation market is expected to exhibit the fastest growth with an estimated contribution of 21.0% share to the global market in 2026, owing to extensive infrastructure buildup of biomedical research facilities driven by the government initiatives, as well as an increase in clinical research studies in the region. For instance, the National Health Commission of the People’s Republic of China has been instrumental in fostering rapid growth in biobank infrastructure in China, with evidence showing that biobanks are prevalent in tertiary hospitals and research organizations, creating one of the largest biobanking infrastructures in the world. (Source: National Library of Medicine)
In India, the Indian Council of Medical Research has created a national biobank facility for conducting genomic studies (for instance, the establishment of the National Biobank (ICMR) and initiatives under the GenomeIndia Project). the Ministry of Health, Labour and Welfare in Japan is pushing forward with its regenerative medicine agenda (including frameworks such as the Act on the Safety of Regenerative Medicine and the Pharmaceuticals and Medical Devices Act). These factors collectively drive strong adoption of human biopreservation solutions across the region.
Why are the U.S. Leading Innovation and Adoption in the Human Biopreservation Market?
The U.S. is the most advanced country in human biopreservation owing to the advanced biomedical infrastructure backed by the U.S government, large-scale use of biospecimen banks, and effective translational research processes. The efforts of the National Cancer Institute on building extensive biobanking and biospecimen banks include millions of annotated biological specimens collected from patients to advance cancer studies, and therefore increase the demand for innovative biopreservation tools (Source: National Cancer Institute).
Additionally, other government agencies like the Centers for Disease Control and Prevention (CDC) provide national specimen collections as a part of the NHANES project that systematically collects and preserves biological specimens. With such active participation of the government and investments into biopreservation technology development and advancement, coupled with strict regulation in place, the U.S. ecosystem can be described as very mature and advanced regarding innovations in cryobiology and biobanking.
Is China a Favorable Market for Human Biopreservation Market?
China presents a rapidly expanding and potentially lucrative market for the human biopreservation, driven by governmental backing, a robust biomedical research environment, and advancements in healthcare infrastructure. The nation has cultivated its life sciences infrastructure via national initiatives centered on precision medicine, biobanking, and clinical research (such as the China Precision Medicine Initiative, Healthy China 2030 Plan, and the 14th Five-Year Plan for Biotechnology Development).
For instance, the National Health Commission of the People’s Republic of China has enabled widespread development of biobanking infrastructure, with studies indicating that biobanks are extensively established across tertiary hospitals and research institutions, forming one of the largest networks globally (Source: National Library of Medicine). Furthermore, the Personal Information Protection Law (PIPL) of China continues to ensure proper regulation of biological data management.
Is India Emerging as a Key Growth Hub for the Human Biopreservation Market?
India has become an emerging market for human biopreservation owing to the fast-growing developments in biotechnology, genomics, and clinical research activities that are being undertaken by government bodies. The Indian government's Department of Biotechnology is spearheading substantial projects like GenomeIndia. This initiative seeks to establish a national genomic database, housing more than 10,000 genome samples. Consequently, there's a considerable need for biobanking and cryopreservation infrastructure (Source: Department of Biotechnology).
Additionally, India has created a national biobank system in connection with government-funded research projects (e.g., GenomeIndia, Indian Biological Data Centre, National Biobank Consortium, CSIR Biorepository Network). Together with an increase in clinical studies and government policies favoring innovation in biotech, these factors have made India an emerging market for developing new biopreservation technologies especially in relation to genomics and personalized medicine.
Why Does Germany Top the European Human Biopreservation Market?
Germany leads the market of human biopreservation in Europe owing to the presence of an advanced research system, good regulatory environment, and substantial investment in the areas of life sciences and healthcare (with R&D spending reaching ~3.1% of GDP). There is considerable encouragement by Germany towards activities related to biobanking and biomedical research in line with the High-Tech Strategy 2025 and extensive funding for translational research (Source: Statistisches Bundesamt (Destatis)).
Moreover, the country spends a considerable amount on healthcare as health spending has accounted for more than 13.2% of the Gross Domestic Product in the last few years (Source: Statistisches Bundesamt (Destatis)). The availability of a robust data protection policy in Germany, including the General Data Protection Regulation (GDPR), assures safe storage and processing of biological materials, thereby making people feel more confident about their privacy.
Is Human Biopreservation Market Developing in Japan?
The human biopreservation market in Japan is gaining traction, fueled by significant progress in regenerative medicine and stem cell research. Japan has established extensive biobanks, such as BioBank Japan, to support the long-term preservation of biological samples. Furthermore, the nation's regulatory framework is proving favorable for cell and gene therapies, accelerating their transition into clinical practice.
Regulatory Landscape Governing the Global Human Biopreservation Market
|
Region |
Key Regulations |
Regulatory Overview |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (21 CFR Part 1271), HIPAA, AATB guidelines |
Comprehensive regulatory framework governing HCT/Ps with strict donor eligibility, traceability, and Good Tissue Practices (GTP) requirements along with strong patient data protection laws |
Ensures high product quality and safety, boosting adoption; however, increases compliance costs and time-to-market for new entrants |
|
Europe |
European Medicines Agency, EU Tissue & Cells Directive (2004/23/EC), GDPR, ISO 20387 |
Harmonized regulations across member states focusing on donor consent, traceability, storage standards, and cross-border exchange of biospecimens |
Facilitates regional collaboration and biobanking networks but adds regulatory complexity and certification burden |
|
Asia Pacific |
Central Drugs Standard Control Organization, National Medical Products Administration, Pharmaceuticals and Medical Devices Agency |
Fragmented and evolving regulatory environment with increasing focus on stem cell research oversight, ethical approvals, and clinical application restrictions |
Creates entry challenges due to variability but offers strong growth potential driven by regulatory maturation and rising investments |
|
Latin America |
Agência Nacional de Vigilância Sanitária, Federal Commission for the Protection against Sanitary Risk |
Developing regulatory frameworks focusing on biospecimen handling, tissue banking standards, and clinical research compliance |
Moderate regulatory enforcement may limit standardization but supports gradual market expansion and foreign investment opportunities |
|
Middle East & Africa |
Saudi Food and Drug Authority, Department of Health – Abu Dhabi, National health authorities |
Emerging regulatory landscape emphasizing ethical biobanking, donor consent, and infrastructure development |
Improving regulatory clarity is attracting international partnerships and investments, supporting early-stage market growth |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How are growing investments and government funding in biotechnology, personalized medicine, and biobanking initiatives creating new growth opportunities for the human biopreservation market?
There is an increase in government expenditure and investments in biotechnology, personalized medicines, and biobanks around the world, which is generating a positive situation for the human biopreservation market, considering the requirement of a strong infrastructure regarding biospecimen storage and long-term preservation infrastructure. For instance, the National Institutes of Health invested nearly USD 48 billion budget in medical research for the American people, which includes funds for precision medicines and biobank-related projects. (Source: National Institutes of Health)
Concurrently, the European Commission is investing USD 109.2 billion (EUR 93.5 billion) under its Horizon Europe program (2021–2027) (Source: European Commission). Additionally, country-wide genome projects that are being supported through government funding have generated huge amounts of biological material (e.g., UK Biobank, All of Us Research Program, GenomeIndia), which requires advanced methods of cryopreservation. Consequently, increased investments by the government in these programs have resulted in increased demand for ultra-low temperature storage and automated biobanking technologies.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In February 2026, CaseBioscience introduced CaseCryo CTG DMSO and CaseStor HSS Hypothermic Storage Solution at the Advanced Therapies Week (ATW) 2026, furthering their biopreservation product range for cell therapy. These innovative products have been developed to improve the long-term viability, efficacy, and consistency of cells used in cell therapy manufacturing processes.
- In May 2025, BioIVT unveiled its VivoSTART cryopreserved GMP leukopaks. These are intended to bolster cell therapy development, offering a reliable, high-quality foundation for both research and clinical manufacturing. This new offering streamlines workflows and allows for greater scalability within advanced therapy pipelines, highlighting the critical role of biopreservation in cell-based therapies.
- In April 2025, BioLife Solutions revealed its acquisition of PanTHERA CryoSolutions. This move was designed to broaden its range of cryopreservation media and solidify its standing as a dedicated bioproduction consumables provider. The purchase bolsters BioLife's ability to support cell and gene therapy processes, further cementing its position as a leader in the biopreservation sector.
Competitive Landscape
The human biopreservation market is highly competitive due to the fast-growing innovations within biotechnology, rising requirement for biobanking, and rise in cell & gene therapy usage. Well-established market players that have an advantage in their R&D capacity and high-grade cryopreservation techniques are expanding their business and managing the complexities of regulations and clinical processes. Key focus areas include:
- Advanced cryopreservation techniques such as vitrification and controlled-rate freezing
- Integration of automated storage systems and digital sample tracking technologies
- Expansion of cold chain logistics and ultra-low temperature storage solutions
- Strategic collaborations with biopharma companies and research institutions
Market Report Scope
Human Biopreservation Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 5,783.2 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.3% | 2033 Value Projection: | USD 11,698.0 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Thermo Fisher Scientific Inc., Merck KGaA, Azenta Inc., BioLife Solutions Inc., Cryoport Inc., Panasonic Healthcare Co. Ltd., Lonza Group AG, Sartorius AG, Eppendorf AG, and Chart Industries Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The trajectory of the human biopreservation market will likely be substantially influenced by advancements within the cell and gene therapy sector, regenerative medicine, and personalized healthcare. Given the increasing emphasis on biologics and the customization of medical treatments, the demand for advanced cell and tissue preservation technologies is poised to expand. Furthermore, innovations in automation and AI-integrated biopreservation solutions are anticipated to materialize.
- The potential revenue streams can be identified to lie in the areas of cell therapy and regenerative medicine in the U.S. and China, due to their well-established R&D environment along with financial support. In addition, the biobank market expansion in Europe (Germany, U.K.) and development in the clinical research infrastructure in India can be regarded as high-growth markets. In terms of application, stem cell storage and CAR-T cell therapy storage can be considered the largest segments for revenue generation.
- In order to create competitive advantage, players in the market should concentrate their efforts on innovation in technologies used in cryopreservation processes such as vitrification and automation. The creation of strategic alliances with pharmaceutical firms, research institutions, and healthcare providers is essential in order to improve the market positioning of the firm.
Market Segmentation
- Offerings Insights (Revenue, USD Mn, 2021 - 2033)
- Equipment and Instruments
- Media and Kits
- Consumables and Accessories
- Software / Digital Systems
- Biospecimen Insights (Revenue, USD Mn, 2021 - 2033)
- Cells
- Bio-Fluids
- Tissues
- Preservation Method Insights (Revenue, USD Mn, 2021 - 2033)
- Cryopreservation
- Vitrification
- Hypothermic Storage
- Others
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Biobanking
- Regenerative Medicine
- Research
- Other Applications
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Biopharmaceutical Companies
- Contract Research Organizations (CROs)
- Academic and Research Institutes
- Hospitals and Biobanks
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Thermo Fisher Scientific Inc.
- Merck KGaA
- Azenta Inc.
- BioLife Solutions Inc.
- Cryoport Inc.
- Panasonic Healthcare Co. Ltd.
- Lonza Group AG
- Sartorius AG
- Eppendorf AG
- Chart Industries Inc.
Sources
Primary Research Interviews
- Human biopreservation solution providers
- Cryopreservation and biobanking service providers
- Hospital administrators and laboratory managers
- Life sciences researchers and regenerative medicine experts
Stakeholders
- Providers of Human Biopreservation Solutions (e.g., Thermo Fisher Scientific, Merck KGaA, Avantor, BioLife Solutions, Chart Industries)
- End-use Sectors
- Biobanks
- Hospitals and Clinics
- Research Institutes
- Pharmaceutical & Biotechnology Companies
- Regulatory & Health Bodies: World Health Organization, U.S. Food and Drug Administration, European Medicines Agency
Databases
- Data World Bank Healthcare Expenditure Database
- World Health Organization Global Health Observatory (GHO)
- OECD Health Statistics
- National Institutes of Health (NIH) Research Funding Database
Magazines
- Genetic Engineering & Biotechnology News – Biopreservation and cell therapy trends
- Lab Manager Magazine – Laboratory storage and cryopreservation technologies
- Healthcare Business Today – Life sciences infrastructure developments
- BioPharma Dive – Biologics and cell therapy market trends
- Pharmaceutical Technology – Cold chain and biologics preservation
Journals
- Nature Biotechnology – Cryopreservation and regenerative medicine advancements
- Cryobiology Journal – Preservation techniques and innovations
- Journal of Translational Medicine – Cell and gene therapy storage
- The Lancet – Biobanking and clinical research applications
- Stem Cell Research & Therapy – Stem cell preservation technologies
Newspapers
- The Wall Street Journal – Biotechnology investments and innovation
- Financial Times – Global life sciences market trends
- Business Standard – Growth in biotech infrastructure
- The Economic Times – India biopharma and research ecosystem
Associations
- International Society for Biological and Environmental Repositories (ISBER)
- American Association of Tissue Banks (AATB)
- Society for Cryobiology
- Alliance for Regenerative Medicine
Public Domain Sources
- World Health Organization – Global Strategy on Digital Health (2020–2027)
- National Institutes of Health (NIH) – Biobanking and research initiatives
- U.S. Food and Drug Administration – Cell and gene therapy regulations
- European Commission – Life sciences and biobanking regulatory framework
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients