Global Human-Derived Exosomes Skincare Market Size and Forecast – 2026-2033
The global human-derived exosomes skincare market is expected to grow from USD 164.0 Mn in 2026 to USD 362.6 Mn by 2033, registering a compound annual growth rate (CAGR) of 12%. The global human-derived exosomes skincare market is poised for significant expansion, fueled by rising aging population.
According to the World Health Organization, by 2030, 1 in 6 people globally will be aged 60 years or over, with the population increasing from 1 billion in 2020 to 1.4 billion, and expected to reach 2.1 billion by 2050.
(Source: World Health Organization)
Key Takeaways of the Global Human-Derived Exosomes Skincare Market
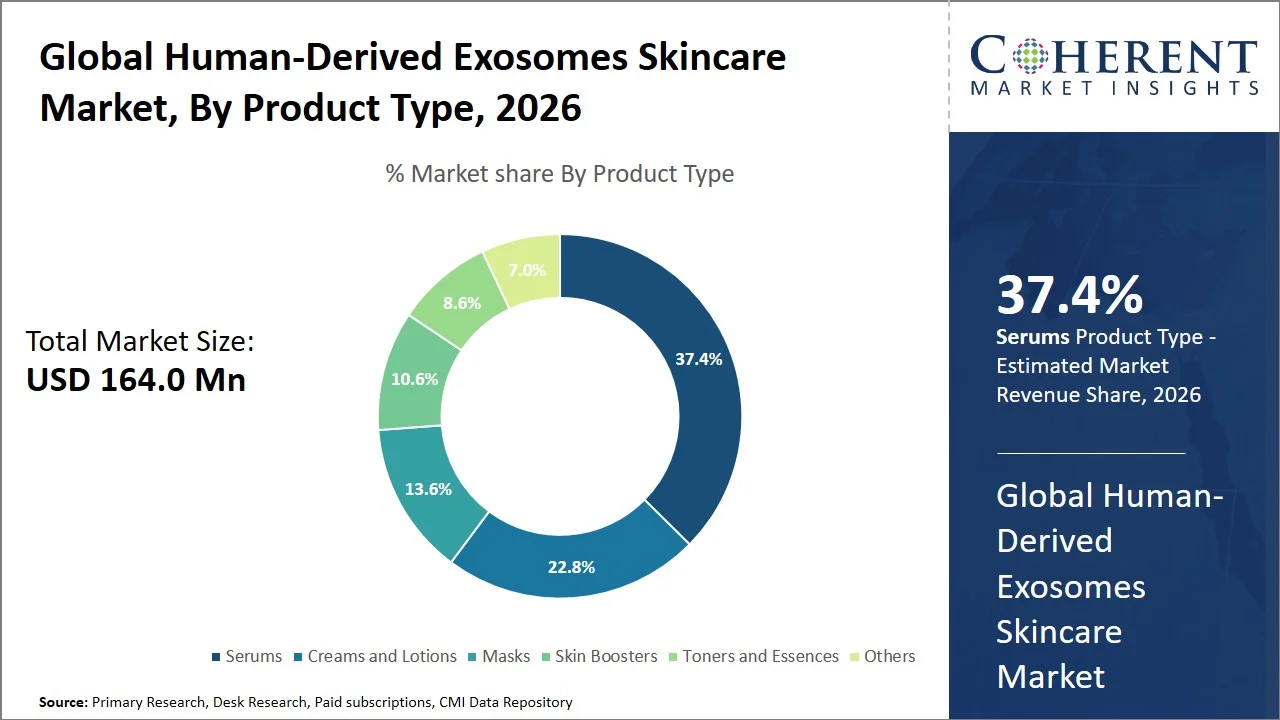
- Serums are projected to hold 37.4% of the global human-derived exosomes skincare market share in 2026, making it the dominant product type segment, especially in Asia Pacific, where customers have a liking for lightweight and quickly absorbed formulas and K-beauty and J-beauty regimens focus on potent serums that help in tackling aging issues. For instance, the Ministry of Food and Drug Safety (MFDS) requires regulatory evaluation for functional cosmetics. This includes products designed to improve skin, such as serums and those with anti-aging properties, to verify their effectiveness and safety. This regulation reflects the strong consumer demand for high-performance skincare products that contain active ingredients, as outlined in the MFDS Functional Cosmetics Guidelines. (Source: Ministry of Food and Drug Safety)
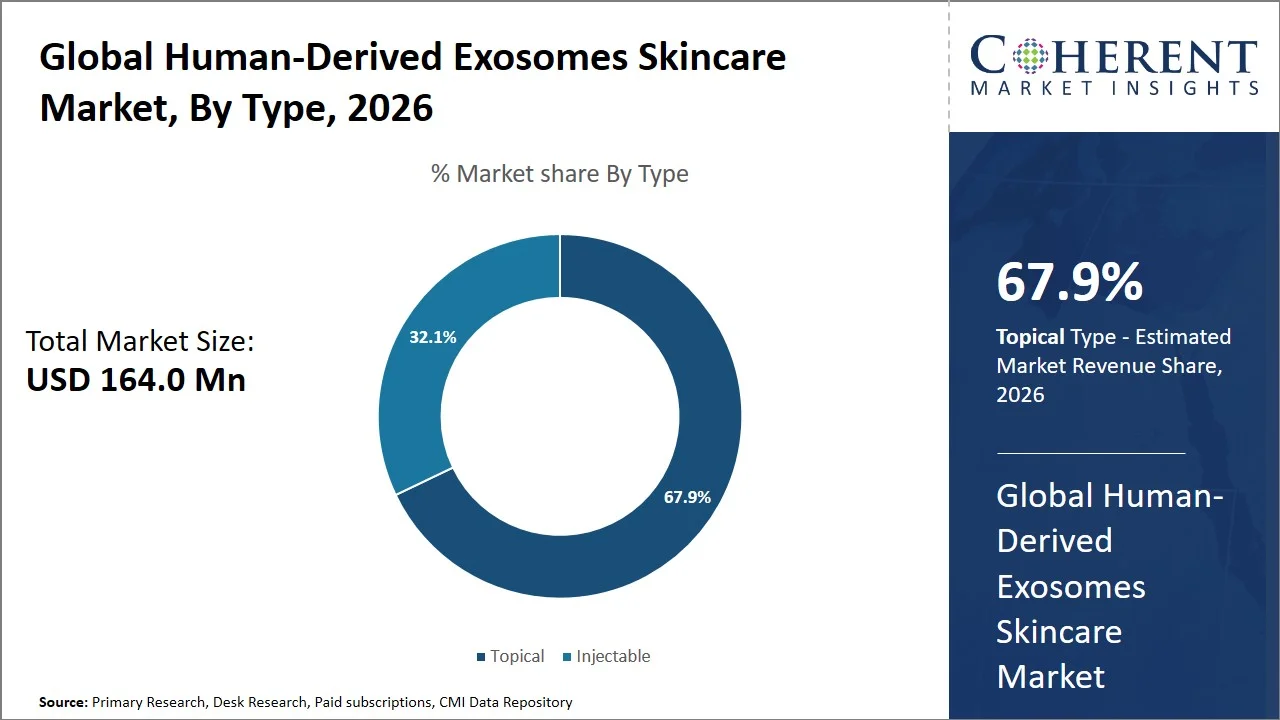
- Topical is projected to hold 67.9% of the global human-derived exosomes skincare market share in 2026, making it the dominant type segment, particularly in North America, owing to stringent regulatory oversight for injectables (e.g., oversight by the U.S. Food and Drug Administration (FDA) for biologics and injectable products), a greater consumer preference for minimally invasive procedures, and ease of incorporating them into their routine skincare programs.
- Mesenchymal Stem Cell (MSC)-Derived Exosomes segment is projected to hold 77.3% of the global human-derived exosomes skincare market share in 2026, making it the dominant exosome sources segment, especially in Europe, which is dominated by ingredients that have been clinically validated, safe, and research backed, thus making MSC-derived exosomes the most favorable because of their anti-inflammatory and regenerative properties. For instance, the European Medicines Agency (EMA) classifies exosome-based products as Advanced Therapy Medicinal Products (ATMPs). This classification requires a thorough evaluation, including compliance with Good Manufacturing Practices (GMP), safety studies, and clinical validation. These requirements support the credibility and use of MSC-derived exosome-based applications in regenerative dermatology.
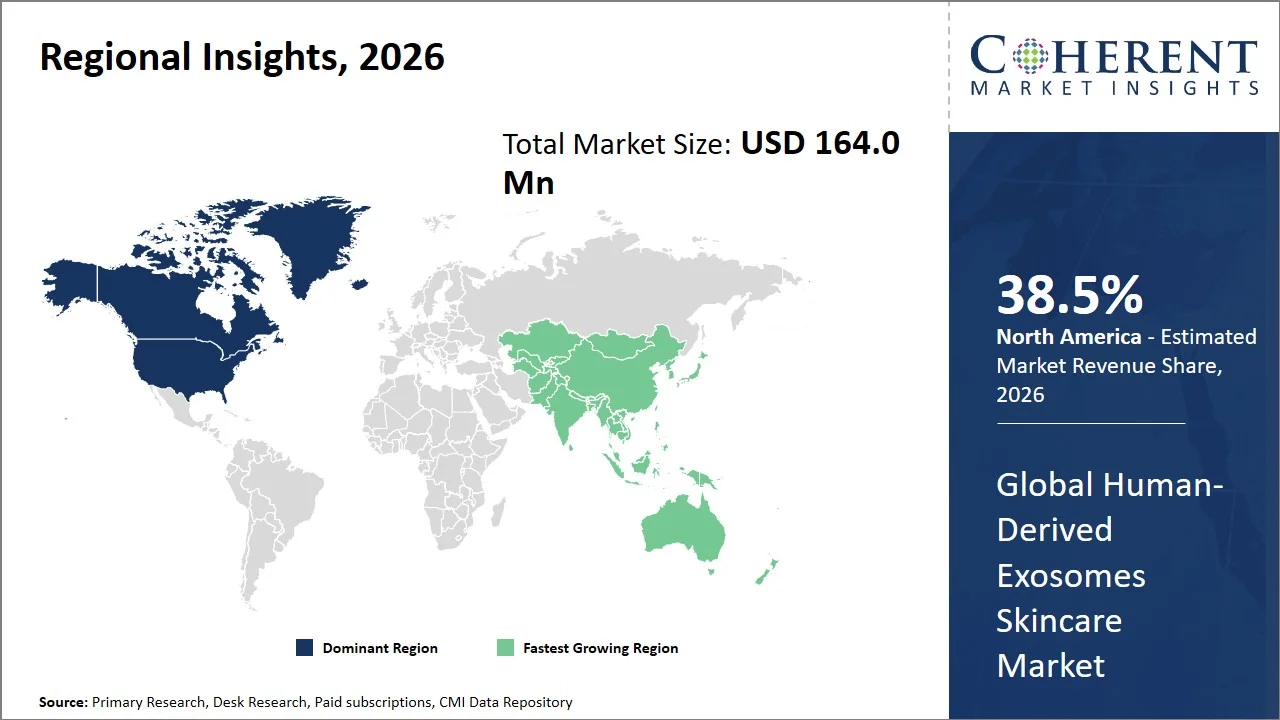
- North America market maintains dominance with an expected share of 38.5% in 2026, due to the prevalence of advanced cosmetic clinics, higher levels of consumer spending on luxury skincare (e.g., personal consumption expenditures on personal care products in the U.S. exceeded USD 90 billion in recent years, reflecting strong premium skincare demand as per the Bureau of Economic Analysis (BEA)) and stringent regulatory environment encouraging the use of proven products.
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 32.7% share in 2026, increasing popularity of innovation in skin care, quick growth of K-beauty and J-beauty fads, rising discretionary income levels (e.g., Asia Pacific accounted for over 40% of global middle-class consumption growth, supported by income expansion trends reported by the World Bank), and the increasing use of regenerative skin care treatments in various countries such as China, Japan, and South Korea.
- Expansion in Post-Procedure Skincare (Laser, Microneedling, Peels): Exosome-based therapies are gaining traction in post-aesthetic procedure recovery. They offer the promise of faster healing, less inflammation, and improved skin regeneration. This opens a specialized avenue for dermatology clinics and medical spas looking to provide cutting-edge recovery options.
- Rising Demand for Stem Cell-Derived Cosmetic Ingredients: Increasing popularity of regenerative and stem cell technology in skincare formulations are pushing demand for exosomes as an innovative bioactive component. The similarity of their actions to the physiological processes of wound healing makes exosomes the most promising substitute for conventional actives.
Why Do Serums Dominate the Global Human-Derived Exosomes Skincare Market?
Serums are projected to hold the market share of 37.4% in 2026, owing to their high efficacy rates in delivering their active ingredients and clinically proving effective in skin hydration and regeneration. In December 2024, as per research published in the National Library of Medicine, topicals have shown significant improvement in skin hydration, and the effect could be seen 12-24 hours after the treatment, along with lower rates of trans-epidermal water loss, thus implying higher levels of skin barrier functionality (Source: National Library of Medicine). Additionally, in May 2025, a study published in the National Library of Medicine has shown noticeable improvements in the quality and hydration of the skin using serum formulations, which shows their high potential in delivering active ingredients into deeper layers of skin (Source: National Library of Medicine). The superior clinical performance, along with higher concentrations of actives and better absorption rates, make serum the optimal choice for advanced skincare formulations that use exosomes for regeneration.
Why is Topical the Most Preferred Type in Global Human-Derived Exosomes Skincare Market?
To learn more about this report, Request Free Sample
Topical is projected to hold a market share of 67.9% in 2026, owing to non-invasive nature, easy to use, and have proven effective in enhancing various skin conditions. According to research published by the National Library of Medicine in December 2024, topical skincare formulations have shown a remarkable enhancement of hydration and barrier function during a short period of application. Moreover, the method of topical administration excludes any risks related to invasive treatment, which also makes it more appealing to the target market, since people want to be confident that the procedure will not have negative effects on their health (Source: National Library of Medicine). Moreover, being non-invasive, this method is free from any hazards and complications that may occur during the application of other methods and is therefore much preferred by many customers looking for safe and reliable means to slow down aging processes and improve skin condition. Considering the high consumer interest in convenient treatments, topical exosomes are dominating the market.
Mesenchymal Stem Cell (MSC) Derived Exosomes Segment Dominates the Global Human-Derived Exosomes Skincare Market
The mesenchymal stem cell (MSC)-Derived Exosomes segment is projected to hold a market share of 77.3% in 2026, due to the enhanced regenerative abilities and anti-inflammatory qualities, making them extremely effective in skin repair and rejuvenation applications. Exosome produced from mesenchymal stem cell (MSC) contain growth factors, cytokines, and other signaling molecules that help boost the number of fibroblasts along with enhancing collagen generation, thereby increasing skin elasticity. In October 2025, research published by Elsevier has shown that the presence of mesenchymal stem cell (MSC) exosomes can help boost the rate of wound healing up to 30-50% while ensuring lower levels of inflammation and tissue remodeling. In addition, their lower immunogenicity and higher biocompatibility also increase safety when used cosmetically. (Source: Elsevier)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
FDA Safety Notification on Exosome Products (Regulatory | U.S.) |
|
|
EU Strengthening Cosmetic Ingredient Safety Regulations (Regulatory | Europe) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: U.S. Food and Drug Administration, European Commission)
Global Human-Derived Exosomes Skincare Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
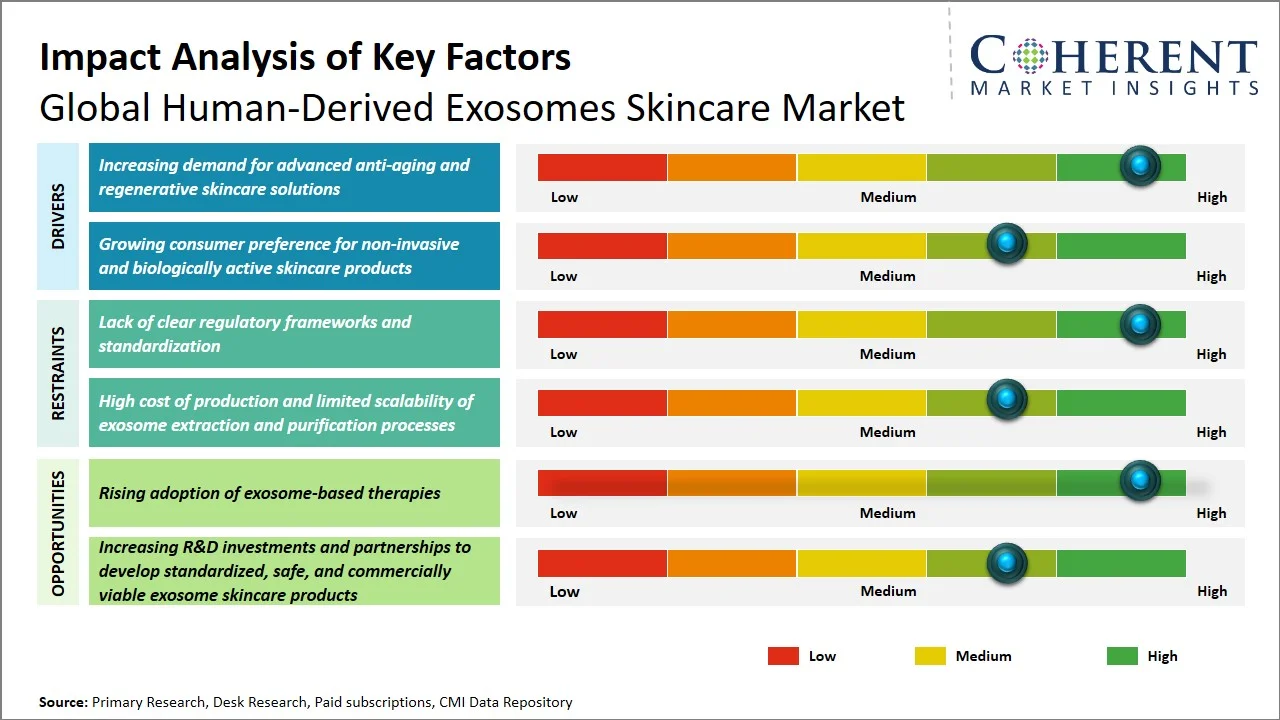
- Increasing demand for advanced anti-aging and regenerative skincare solutions: The burgeoning human-derived exosomes skincare market is primarily fueled by the escalating need for sophisticated anti-aging and regenerative skincare products, a trend underscored by the swift aging of the global populace. The aging process is characterized by a decreased rate of collagen synthesis, delayed skin repair, and a higher incidence of skin diseases. As a result, this growing concern in healthcare is driving the need for bio-advanced skin care products, with the rising acceptance of exosomes being driven by their ability to repair, regenerate, and reverse aging cells.
- Growing consumer preference for non-invasive and biologically active skincare products: Increasing consumer preference for non-invasive and bioactive skincare products are primary growth drivers for the human-derived exosomes skincare market as more people are opting for safe skin rejuvenation methods that can be done without undergoing surgical cosmetic treatments. Based on statistics released by the International Society of Aesthetic Plastic Surgery, more than 34.9 million aesthetic surgery operations have been performed worldwide in 2023, out of which, roughly 19.1 million operations are non-invasive procedures while 15.8 million are surgical procedures. (Source: International Society of Aesthetic Plastic Surgery) As a consequence, there is a growing demand for sophisticated skin care products that provide regenerative effects without the need for surgery, making the use of exosomes a logical choice.
Emerging Trends
- Shift Toward Personalized Exosome-Based Skincare: Personalized medicine is gaining popularity with companies developing exosomes based on the individual’s skin type, genetics, and any particular condition affecting the skin. The development has significantly improved efficacy and the use of high-end products in cosmetic medicine.
- Advancements in Exosome Stabilization and Delivery Technologies: Encapsulation, lyophilization, and nanotechnology delivery methods have improved the stability, longevity, and absorption ability of exosomes. The development has enabled mass production and use of exosomes in topical applications.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Human-Derived Exosomes Skincare?
North America leads the global human-derived exosomes skincare market, accounting for an estimated 38.5% share in 2026, attributable to the significant prevalence of dermatological ailments, a high degree of clinical awareness, and a sophisticated healthcare system that facilitates the integration of biologically active skincare solutions. The American Academy of Dermatology reports that acne affects more than 50 million Americans each year, while conditions like eczema impact nearly one in ten individuals, thereby underscoring a considerable need for efficacious skin treatments. (Source: American Academy of Dermatology) Furthermore, the National Institutes of Health highlights the expanding significance of regenerative and cell-based methodologies in skin healing and tissue repair, thus fostering the adoption of biologically active skincare innovations (Source: National Library of Medicine). Consequently, this convergence of a substantial disease burden, heightened awareness, and a robust research environment fuels the demand for advanced skincare technologies, including exosome-based products, within the North America market.
Why Does Asia Pacific Human-Derived Exosomes Skincare Market Exhibit High Growth?
The Asia Pacific human-derived exosomes skincare market is expected to exhibit the fastest growth with an estimated contribution of 32.7% share to the global market in 2026, owing to a large demographic population, high disease burden, and increased consumer demands for modern dermatological and aesthetic care services. As per World Health Organization reports, the South East Asia region represents about a quarter (approximately 2 billion consumers) of the total world population making Asia Pacific the most suitable market for skincare and cosmetic procedures. (Source: World Health Organization). Moreover, the growing prevalence of non-communicable diseases, along with the increasing number of older adults, makes the Asia Pacific region a key market.
Why are the U.S. Leading Innovation and Adoption in the Human-Derived Exosomes Skincare Market?
The U.S. is the most advanced country in human-derived exosomes skincare market owing to its robust research environment and early strides in regenerative medicine. The U.S. FDA has issued its regulatory guidelines for safety notices for exosomes-based products indicating increasing oversight and recognition of this emerging category (Source: U.S. Food and Drug Administration). With this blend of scientific research, regulatory involvement, and early adoption of innovative skincare products, the country is poised to be a leader in the human-derived exosomes skincare market.
Is China a Favorable Market for Human-Derived Exosomes Skincare Market?
China emerges as a promising yet cautiously optimistic market for the human-derived exosomes skincare industry, which witness high demand and innovation, but is limited by regulations. China ranks among the top markets globally for the beauty industry, with more investments in biotechnology-based skin care and reforms from the National Medical Products Administration (NMPA) to support innovation and expedite cosmetic ingredient approvals (Source: Dezan Shira & Associates). However, exosomes from human sources are presently not allowed as cosmetic ingredients in China because the Chinese regulatory body clearly forbids the inclusion of any human cells or tissues in cosmetics. This poses a huge challenge to the usage of exosomes from human sources, thus forcing firms to explore other avenues for their products. Although China represents an ideal market in terms of consumer demand, R&D, and market size, the strict regulatory policy makes it a conditional market rather than an open one.
Is Japan Emerging as a Key Growth Hub for the Human-Derived Exosomes Skincare Market?
Japan is shaping up to be a promising growth cluster in the field of exosomes skincare from humans, more so because of innovation rather than regulatory approval. Japan has a sound background in regenerative medicine and aesthetic dermatology and is also flexible in its regulation, such that physicians can apply exosome therapy in the clinic before regulatory pre-approval (under the Act on the Safety of Regenerative Medicine (ASRM)). For instance, clinical treatments using exosomes have been administered without the standard approval route in clinics in Japan, as noted in research papers and institutional reports because these treatments are administered on a trial basis within institutions and not yet approved through pharmaceutical regulation. (Source: Center for iPS Cell Research and Application, Kyoto University)
However, there are no defined regulations in Japan for exosomes and no exosome-based products have been approved by regulatory authorities for cosmetic purposes. Also, biological materials used in exosome therapies have to meet certain criteria under the Ministry of Health framework, which makes the commercialization of human-derived exosomes difficult in over-the-counter (OTC) skin care. Therefore, Japan can be considered to be a promising market in terms of innovation and early adoption, yet with a few grey areas in the regulatory context, and thus a promising growth cluster.
Why Does Germany Top the European Human-Derived Exosomes Skincare Market?
Germany leads the market of human-derived exosomes skincare in Europe owing to country’s excellent research facilities in the medical field, dermatological infrastructure, and regulations concerning medically-oriented innovations (e.g., EU Medical Device Regulation (MDR) and German Medicines Act (Arzneimittelgesetz, AMG)). The country is center for biotechnological development and regenerative medicine, where companies like the Paul Ehrlich Institute and the Federal Institute for Drugs and Medical Devices (BfArM) monitor the quality of biological materials used in treatment. Although stringent regulations limit the application of human-derived ingredients in cosmetics, they establish credibility, making people trust in the physician-driven medical aesthetic treatment. Besides, the country has robust research and development initiatives with collaboration from biotechnology firms and educational institutions to make advancements in skin care, such as the Fraunhofer Society and top universities have promoted translational research in regenerative medicine and extracellular vesicles, aiding the development of novel exosomes for dermatology.
Is Human-Derived Exosomes Skincare Market Developing in South Korea?
South Korea is emerging as a key player in the human-derived exosome skincare market, fueled by its robust K-beauty scene and cutting-edge biotechnology. Dermatology clinics and aesthetic practices are increasingly incorporating these treatments, especially for anti-aging and regenerative purposes. Yet, the Ministry of Food and Drug Safety (MFDS) has tightened regulations (e.g., 2025 ban on “exosome” claims in cosmetic advertising and restrictions on human-derived ingredient references), imposing limits on "exosome" claims and the use of human-derived ingredients in cosmetics. This has, in turn, constrained widespread commercialization within the mass skincare sector.
Regulatory Landscape Governing the Global Human-Derived Exosomes Skincare Market
|
Region |
Regulatory Authority |
Regulatory Status |
Key Regulation |
Impact on Market |
|
North America |
U.S. Food and Drug Administration |
No specific exosome cosmetic approvals; regulated under biologics if therapeutic |
Federal Food, Drug, and Cosmetic Act (FD&C Act) |
Exosomes may be classified as biologics requiring strict clinical validation; no FDA-approved exosome products currently |
|
Europe |
European Commission / European Medicines Agency |
Use of human-derived materials in cosmetics is highly restricted |
EU Cosmetic Regulation (EC) No. 1223/2009 |
Strict regulatory framework limits the use of human-derived exosomes, restricting commercialization in cosmetic applications |
|
Asia Pacific |
National Medical Products Administration (China), Ministry of Food and Drug Safety (South Korea), MHLW (Japan) |
Varies by country; generally restricted or tightly regulated |
Cosmetic Supervision and Administration Regulation (CSAR - China), Cosmetic Act (Korea), Pharmaceutical Affairs Law (Japan) |
Regulatory fragmentation; South Korea and Japan allow controlled innovation, while China maintains stricter restrictions, impacting uniform market growth |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico) |
No specific exosome guidelines; regulated under general cosmetic/biologic frameworks |
General Health Law (Mexico), ANVISA Cosmetic Regulations |
Lack of clear exosome-specific regulation creates uncertainty but allows gradual market entry under existing cosmetic frameworks |
|
Middle East & Africa |
SFDA (Saudi Arabia), South African Health Products Regulatory Authority |
Emerging regulatory oversight; limited specific guidance |
SFDA Cosmetic Regulations, SAHPRA guidelines |
Developing regulatory landscape with increasing focus on safety and compliance, offering early-stage growth opportunities |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the growing investments and government funding in biotechnology, personalized medicine, and biobanking initiatives creating new growth opportunities in the human-derived exosomes skincare market?
There is an increase in government expenditure and investments in biotechnology, personalized medicines, and biobanks around the world, which is generating a positive situation for the human-derived exosomes skincare market, considering the requirement of a strong infrastructure regarding biospecimen storage and long-term preservation infrastructure. For instance, the National Institutes of Health invested nearly USD 48 billion budget in medical research for the American people, which includes funds for precision medicines and biobank-related projects (Source: National Institutes of Health).
Concurrently, the European Commission is investing USD 109.2 billion (EUR 93.5 billion) under its Horizon Europe program (2021–2027) (Source: European Commission). Additionally, country-wide genome projects that are being supported through government funding have generated huge amounts of biological material (e.g., UK Biobank, All of Us Research Program, GenomeIndia), which requires advanced methods of cryopreservation. Consequently, increased investments by the government in these programs have resulted in increased demand for ultra-low temperature storage and automated biobanking technologies.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In November 2025, Pluri Inc. joined forces with Miss Universe Skincare to launch exosome-enhanced skincare products designed to regenerate. This move represents a major leap toward bringing advanced, biotech-driven cosmetics to market. The partnership underscores the rising trend of incorporating biotechnology into high-end skincare, as well as the growing consumer appetite for effective anti-aging treatments.
- In April 2024, dsm-firmenich collaborated with ExoLab Italia to create plant-based exosome technology for skincare. This partnership underscores a significant industry trend: a move away from exosomes sourced from humans. The shift is largely driven by regulatory hurdles and safety worries surrounding human-derived exosomes.
Competitive Landscape
The human-derived exosomes skincare market is highly competitive due to fast innovation within the field of biotechnology as well as increased demand for efficient and innovative regenerative skincare products. Competitive advantage is being taken up by companies that have the ability to innovate and process their product using highly sophisticated technology and techniques. There is also an increasing trend towards incorporating cold chains and storage facilities into the supply chain of such products. Key focus areas include:
- Advanced preservation & stabilization techniques
- Standardization and quality control protocols
- Integration of advanced delivery systems
- Strategic collaborations and R&D investments
Market Report Scope
Human-Derived Exosomes Skincare Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 164.0 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12% | 2033 Value Projection: | USD 362.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
ExoCoBio, Amorepacific Corporation, GFC Life Sciences, ExoStemTech, Prostemics, Rion Aesthetics, Creative Biolabs, Advancexo, Wigmore Medical, and MCCM Medical Cosmetics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The trajectory of the human-derived exosomes skincare market seems promising, owing to the merging of advanced biotechnology with high-end skincare products. With growing validation and regulatory clearances, there will be an increased use of such products in the near future. The trend of science-backed and regenerative skincare products, which does not involve any invasive techniques, is on the rise among consumers.
- The maximum potential for growth will be seen in non-invasive or topical application-based product formulations in the anti-aging and dermatology segments within North America and Asia Pacific regions, especially the U.S., South Korea, China, and India. The reason behind this is the high degree of consumer awareness, advanced skincare markets, and increasing focus on novel cosmetics using biologics.
- For competitive advantage, one needs to emphasize on clinical testing and regulatory approval, invest in scalable and standardized methods for producing exosomes, and pursue strategic partnerships with dermatology clinics and aesthetic centers. Moreover, product differentiation, which can be achieved through efficacy of the product, high pricing, and safety assurance.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Serums
- Creams and Lotions
- Masks
- Skin Boosters
- Toners and Essences
- Others
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Topical
- Injectable
- Exosome Sources Insights (Revenue, USD Mn, 2021 - 2033)
- Mesenchymal Stem Cell (MSC)-Derived Exosomes
- Platelet-Derived Exosomes
- Others
- Application Insights (Revenue, USD Mn, 2021 - 2033)
- Anti-aging
- Skin Hydration
- Skin Pigmentation
- Wound Healing / Skin Repair
- Acne and Inflammation
- Others
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Online
- Offline
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- ExoCoBio
- Amorepacific Corporation
- GFC Life Sciences
- ExoStemTech
- Prostemics
- Rion Aesthetics
- Creative Biolabs
- Advancexo
- Wigmore Medical
- MCCM Medical Cosmetics
Sources
Primary Research Interviews
- Human-Derived Exosomes Skincare solution providers
- Cryopreservation and biobanking service providers
- Hospital administrators and laboratory managers
- Life sciences researchers and regenerative medicine experts
Stakeholders
- Providers of Human-Derived Exosomes Skincare Solutions
- End-use Sectors
- Biobanks
- Hospitals and Clinics
- Research Institutes
- Pharmaceutical & Biotechnology Companies
- Regulatory & Health Bodies: World Health Organization, U.S. Food and Drug Administration, European Medicines Agency
Databases
- Data World Bank Healthcare Expenditure Database
- World Health Organization Global Health Observatory (GHO)
- OECD Health Statistics
- National Institutes of Health (NIH) Research Funding Database
Magazines
- Genetic Engineering & Biotechnology News – Biopreservation and cell therapy trends
- Lab Manager Magazine – Laboratory storage and cryopreservation technologies
- Healthcare Business Today – Life sciences infrastructure developments
- BioPharma Dive – Biologics and cell therapy market trends
- Pharmaceutical Technology – Cold chain and biologics preservation
Journals
- Nature Biotechnology – Cryopreservation and regenerative medicine advancements
- Cryobiology Journal – Preservation techniques and innovations
- Journal of Translational Medicine – Cell and gene therapy storage
- The Lancet – Biobanking and clinical research applications
- Stem Cell Research & Therapy – Stem cell preservation technologies
Newspapers
- The Wall Street Journal – Biotechnology investments and innovation
- Financial Times – Global life sciences market trends
- Business Standard – Growth in biotech infrastructure
- The Economic Times – India biopharma and research ecosystem
Associations
- International Society for Biological and Environmental Repositories (ISBER)
- American Association of Tissue Banks (AATB)
- Society for Cryobiology
- Alliance for Regenerative Medicine
Public Domain Sources
- World Health Organization – Global Strategy on Digital Health (2020–2027)
- National Institutes of Health (NIH) – Biobanking and research initiatives
- U.S. Food and Drug Administration – Cell and gene therapy regulations
- European Commission – Life sciences and biobanking regulatory framework
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients