Global Jet Injector Market Size and Forecast – 2026 to 2033
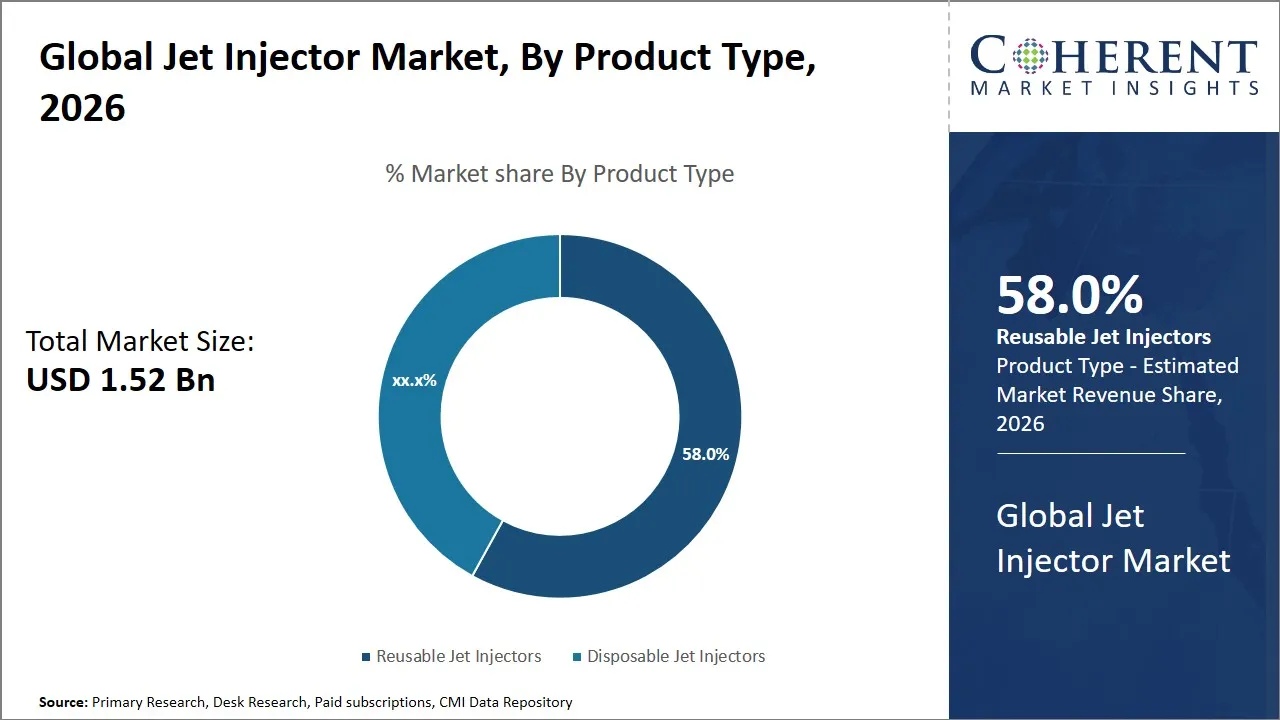
The global jet injector market is estimated to be valued at USD 1.52 Bn in 2026 and is expected to reach USD 2.91 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.72% from 2026 to 2033. This steady growth is fueled by the escalating demand for needle-free injections in the field of health care, immunization programs, and the increased awareness of the advantages of painless drug delivery systems in both developed and developing nations.
Key Takeaways of the Global Jet Injector Market
- The reusable jet injectors segment is expected to dominate the jet injector market, accounting for 58.0% share in 2026.
- The gas powered jet injectors segment is expected to dominate with 44.0% of the market share in 2026.
- The vaccine delivery segment is projected to capture 41.0% of the global jet injector market share in 2026.
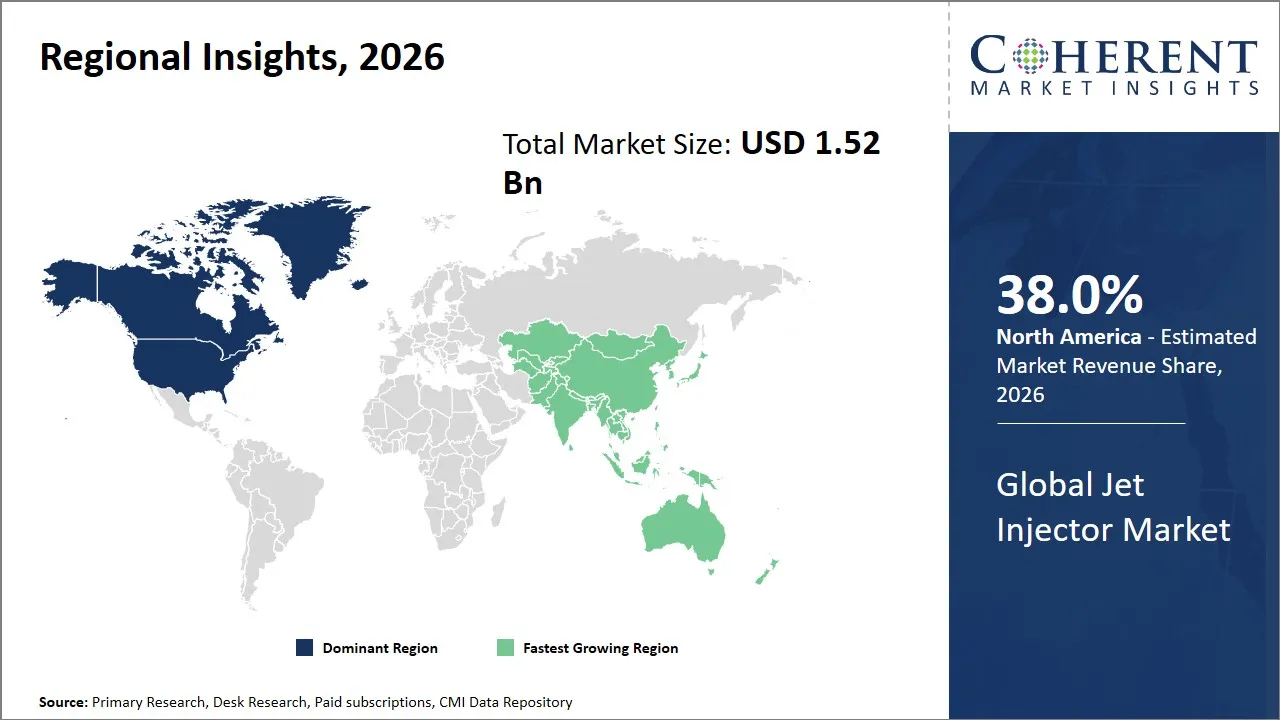
- North America is expected to dominate the global jet injector market in 2026, accounting for 38.0% share.
- Asia Pacific is anticipated to be the fastest growing region, with 28% share in 2026.
Market Overview
- Jet injectors are needle-free drug delivery devices that are widely used in medicine to inject drugs using a high-pressure jet. These injectors are used in various drug delivery procedures, such as vaccine delivery, insulin injection, and pain management therapies. The main advantage of using jet injectors is that they reduce the risk of needle-stick injury and increase patient compliance.
- Jet injectors are widely used in various drug delivery procedures, especially in vaccination drives, clinical settings, and home healthcare services. The growing prevalence of life-threatening diseases, the need to promote vaccination drives, and increased awareness about needle-free drug delivery techniques are key factors that contribute to the growth of this market. In addition, the risks associated with cross-contamination with needle injection techniques are also boosting this market.
- Additionally, the progress in the field of gas-powered as well as electronic jet injector technologies, along with the introduction of disposable jet injector products, is creating growth opportunities in the market. The growth of the biopharmaceutical markets, along with the self-administration trend, is supporting the market growth of jet injectors in developed as well as emerging markets.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Surging Demand for Needle-Free Drug Delivery Systems |
|
|
Advancements in Jet Injector Technologies |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Reusable Jet Injectors Segment Dominate the Global Jet Injector Market in 2026?
The reusable jet injectors segment is anticipated to hold a market share of 58% in 2026, owing to their substantial cost efficiency and sustainability. These devices are designed for multiple uses after proper sterilization. Therefore, there are substantial cost benefits in terms of saving on consumables. The cost efficiency of the jet injectors is also supplemented with environmental concerns. These devices are reusable and thus contribute to the minimal degradation of the environment and the infrastructure in handling biomedical wastes, which is an important issue in both developed and developing countries.
Additionally, there are substantial benefits in the form of increased confidence levels with respect to reusable jet injectors owing to the continuous improvements in technology that have increased their reliability, safety, and usability. Moreover, the use of reusable jet injectors in poor and developing countries, where there are issues in the supply chain, has increased their use and thereby their confidence levels. The issues of cross-contamination have also been alleviated with proper training and sterilization.
For instance, in February 2025, PharmaJet emphasized the use of its reusable intradermal jet injector, Tropis, in polio vaccination campaigns, in which thousands of doses were administered with the help of a single device, utilizing disposable syringes, thereby reducing medical waste as well as the overall cost of vaccination per dose.
(Source: PharmaJet)
Why Does the Gas Powered Jet Injectors Segment Dominate the Global Jet Injector Market in 2026?
The gas powered jet injectors segment is expected to hold the highest market share of 44% in the global jet injector market in 2026, owing to their ease of operation, reliability, and the absence of the need for the use of complex electronic equipment. The ease of operation is due to the ability of the device to utilize the pressure of the compressed gas, which could either be CO2 or air, to operate the jet injector. The ease of operation, coupled with the reliability of the device, makes it highly suitable for the administration of immunizations on a large scale, as well as in the military.
The cost-effectiveness of the gas powered jet injector is another reason that has contributed significantly to the popularity of the device in the market as of 2026. The gas powered jet injector is known to be highly cost-effective, as the maintenance of the device is relatively lower when compared with the electronic jet injector, thus making it highly suitable for the administration of immunizations on a large scale by governments around the world.
The Vaccine Delivery Segment Dominates the Global Jet Injector Market
Vaccine delivery segment forms the largest application segment of the global jet injector market contributing to an estimated market share of 41% in 2026, due to the increase in global immunization programs and the need to develop effective vaccine delivery systems. This is because governments and international health authorities are increasingly choosing vaccine delivery systems that are needle-free, as jet injectors are effective in preventing needle-stick injuries, reducing the chances of cross-contamination, and facilitating the disposal of used needles. This makes it easier for the patients as well as the medical staff, as it reduces the training period as well as for the patients, as it has been noted that patients who suffer from needle-phobia are more willing to take up the jet injectors.
Jet injectors are also effective in providing precise intradermal and subcutaneous injections, which is an added advantage in terms of vaccine efficiency, especially during times of vaccine shortages as well as during pandemics. Considering all these facts, as investments are being made in vaccine R&D and delivery, vaccine delivery takes the lead in the global jet injector market share.
Mass Vaccination Program Scale and Delivery Methods
Mass vaccination programs are implemented on a large scale with the aim of rapidly immunizing the public against different infectious diseases. Different techniques are employed to make vaccine delivery efficient, safe, and effective, and the governments and international health organizations are employing different techniques depending on the situation, with more emphasis on needle-free technology due to speed, safety, and increased compliance with the vaccine delivery system. The choice of vaccine delivery depends on factors such as accuracy, usability, and waste disposal.
Mass Vaccination Delivery Methods
|
Delivery Method |
Key Features |
Advantages |
Limitations |
|
Needle & Syringe |
Conventional intramuscular/subcutaneous injection using disposable needles |
Widely available, familiar to healthcare workers, high accuracy |
Needle-stick injuries, biohazard waste, higher training & disposal burden |
|
Jet Injectors (Needle-Free) |
Uses high-pressure stream to deliver vaccine through skin |
Eliminates needles, reduces cross-contamination risk, faster administration, improves compliance |
Higher initial device cost, requires maintenance/sterilization (for reusable types) |
|
Oral Vaccines |
Administered via drops or liquid orally (e.g., polio vaccines) |
Easy to administer, no trained personnel required, ideal for children |
Limited to specific vaccines, stability and storage challenges |
|
Microneedle Patches (Emerging) |
Patch-based delivery with tiny dissolvable needles |
Minimally invasive, self-administration potential, reduced cold-chain dependency |
Still in development, limited commercial availability |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Regional Insights
To learn more about this report, Request Free Sample
North America Jet Injector Market Analysis and Trends
The North America region is projected to lead the market with a 38% share in 2026, attributed to the well-developed healthcare infrastructure coupled with the rapid technological developments taking place in the field of drug delivery systems. The presence of major pharmaceutical and medical device companies like Becton Dickinson, PharmaJet, and Pfizer helps the market grow with innovative products, along with a highly developed healthcare system, favorable government policies, increased investments in vaccine delivery, and the emphasis on improving vaccination rates, which are major drivers of the market in this region, with the regulatory environment, headed by the FDA, ensuring the high safety and efficacy of the products, thereby promoting their use by the medical community.
For instance, in August 2024, Inovio Pharmaceuticals, a U.S.-based company that specializes in biotechnology, continued to advance its DNA medicine technology with the help of its CELLECTRA needle-free delivery device that utilizes electrical pulses to enhance intracellular uptake of active ingredients.
(Source: Inovio Pharmaceuticals)
Asia Pacific Jet Injector Market Analysis and Trends
Asia Pacific is anticipated to be the fastest growing region, with 28% share in 2026, driven by the development of health infrastructure, increased awareness of the advantages of needle-free technology, and the development of vaccination programs by the government. The countries in this region, such as China, India, Japan, and South Korea, are investing heavily in the development of health infrastructure and vaccination programs. The rise in the rate of chronic diseases and the outbreak of various diseases in this region also indicate the need for the development of quick and safe methods of drug delivery. This region also has the advantage of the presence of manufacturing facilities of companies such as Bioject and various new companies, which are providing cheap technology to this region.
For instance, in March 2025, PharmaJet announced that the World Health Organization facilitated the implementation of PharmaJet’s Tropis needle-free jet injector as part of the polio eradication initiative in Pakistan, which saw over 1.5 million children immunized. The initiative is a pointer to the increased acceptance of needle-free injection systems in the immunization drive across the Asia Pacific region.
(Source: PharmaJet)
Jet Injector Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Jet Injector Market?
The U.S. jet injector market is benefited from advanced R&D activities and early adoption of innovative healthcare technology. Renowned companies such as Becton Dickinson and PharmaJet have been actively working on introducing advanced sophisticated jet injection technology that caters specifically to vaccines and biologics. Moreover, large immunization programs and increased importance given to mass immunization campaigns indicate the vitality of the US market. Regulatory support and reimbursement practices ensure access to jet injector devices, thereby strengthening the US as an important market.
For instance: U.S. Food and Drug Administration emphasized the continued progress of needle-free drug delivery systems, such as jet injectors as part of the FDA’s combination product and drug-device innovation initiatives to develop innovative drug delivery systems for biologics in the U.S. via its guidance documents.
(Source: U.S. Food and Drug Administration)
How is China Helping in the Growth of the Jet Injector Market?
The driving forces of China jet injector market include rapid urbanization, increased public expenditure on healthcare, and vigorous vaccination programs. Domestic and multinational companies have shown increased cooperation in tailoring products for consumption by the local population. Some of the key players include Shanghai Needle-Free Injector Device Manufacturing, which contribute significantly to innovations in needle-free devices. The country’s approval process has also shown an upward trend, thereby propelling product availability and hence the market. Improved distribution channels and increased awareness also contribute to accelerating the jet injector market.
Key Drivers for the Growth of the Germany Jet Injector Market
Germany jet injector market is marked by a strong industrial footprint coupled with innovations in the manufacturing of medical devices, especially precision-engineered jet injectors by companies like Henke Sass Wolf, etc. The healthcare system of Germany is focused on developing advanced therapy solutions, especially needle-free injection technologies, with a robust framework that ensures the quality and safety of the products. The incentives for the innovation of medical device technologies, along with the demand for non-invasive treatments, contribute to the jet injector market size of Germany, which is a major contributor to the overall Europe jet injector market.
India Jet Injector Market Trends
The India jet injector market can be characterized by the high population base, the rising cases of infectious diseases, and the increased emphasis on vaccination programs such as Pulse Polio. The local market is supported by multinational companies in a collaborative effort to provide affordable jet injectors that can cover the rural and urban markets as well. The National Health Mission has also ensured greater access to healthcare services, which in turn has increased the usage of such innovative products. The association with global organizations to provide healthcare products highlights the rising importance of the jet injector market in India.
Regulatory Landscape for Needle-Free Injection Systems: Approvals, Safety Guidelines, and Adoption Frameworks
|
Regulatory Aspect |
Description |
Key Authorities / Guidelines |
Impact on Market |
|
Regulatory Approvals for Needle-Free Injection Systems |
Needle-free injectors (including jet injectors) are regulated as combination products (drug + device), requiring validation of safety, dose accuracy, performance, and biocompatibility before commercialization. |
U.S. Food and Drug Administration (Combination Product Guidance, Jet Injector Guidelines); European Medicines Agency |
Ensures high product quality and safety, builds trust among healthcare providers, and supports commercialization of advanced injection technologies |
|
Guidelines on Safe Injection Practices |
Establish protocols to minimize risks such as needle-stick injuries, cross-contamination, and improper disposal of sharps, while promoting safer alternatives including needle-free systems. |
World Health Organization (Safe Injection Global Network - SIGN); Centers for Disease Control and Prevention |
Drives shift toward needle-free technologies, especially in mass immunization and hospital settings, improving patient and healthcare worker safety |
|
Adoption Frameworks & Public Health Policies |
Governments and global organizations integrate needle-free technologies into immunization strategies and public health programs, supported by funding, procurement policies, and training initiatives. |
World Health Organization; Gavi, the Vaccine Alliance; National immunization programs |
Accelerates adoption in emerging markets, enhances vaccination coverage, and supports large-scale deployment of jet injectors |
|
Standards for Device Performance & Quality |
Includes technical standards for device reliability, sterility, durability, and repeat-use safety (for reusable injectors), along with post-market surveillance requirements. |
International Organization for Standardization; FDA Quality System Regulations |
Promotes continuous innovation, ensures long-term device performance, and reduces risks associated with repeated usage and sterilization |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In January 2025, Portal Instruments, Inc. announced the availability of its PRIME Nexus, a digital-enabled connected drug delivery system utilizing the company’s proprietary PRIME technology, which utilizes electromechanical actuation to accurately and rapidly administer high-viscosity biologics subcutaneously with volumes of up to ~1 mL.
- In December 2024, Crossject announced the relaunch of its joint research program with the United States Department of Defense to advance its ZENEO needle-free auto-injector technology. The collaboration was intended to improve the development of needle-free emergency drug delivery systems, which can facilitate quicker drug delivery with ease as opposed to conventional injection techniques.
Top Strategies Followed by Global Jet Injector Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market players emphasize continuous R&D to create advanced, high-precision needle-free jet injectors, as well as expanding the scope of vaccines and biologics. Emphasis on strategic partnerships and global expansion, especially in the emerging markets. |
In September 2024, PharmaJet announced the expansion of the scope of its Tropis intradermal jet injection technology in polio vaccination programs in Africa. |
|
Mid-Level Players |
Mid-level players focus on targeting the price-sensitive segment by providing affordable and reliable jet injectors, and emphasize strategic partnerships with manufacturers/distribution companies. |
In March 2025, Zogenix continued the development and positioning of alternative drug delivery systems to improve patient compliance in rare diseases. |
|
Small-Scale Players |
Small-scale players emphasize niche innovation and advanced technologies, including ingestible or specialized jet technology-based systems. Additionally, they emphasize differentiation through niche applications and early-stage technological innovations. |
In 2024, Bioject Institute continued support for various research programs related to the development of needle-free injection technology to improve vaccine delivery with minimum risks of cross-contamination. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Jet Injector Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.52 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.72% | 2033 Value Projection: | USD 2.91 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
PharmaJet, CROSSJECT, Portal Instruments Inc, NuGen Medical Devices, Medical International Technology MIT Canada, INOVIO Pharmaceuticals, Aijex Pharma International Inc, IntegriMedical, Micron Biomedical Inc, and Halozyme Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Jet Injector Market Dynamics
To learn more about this report, Request Free Sample
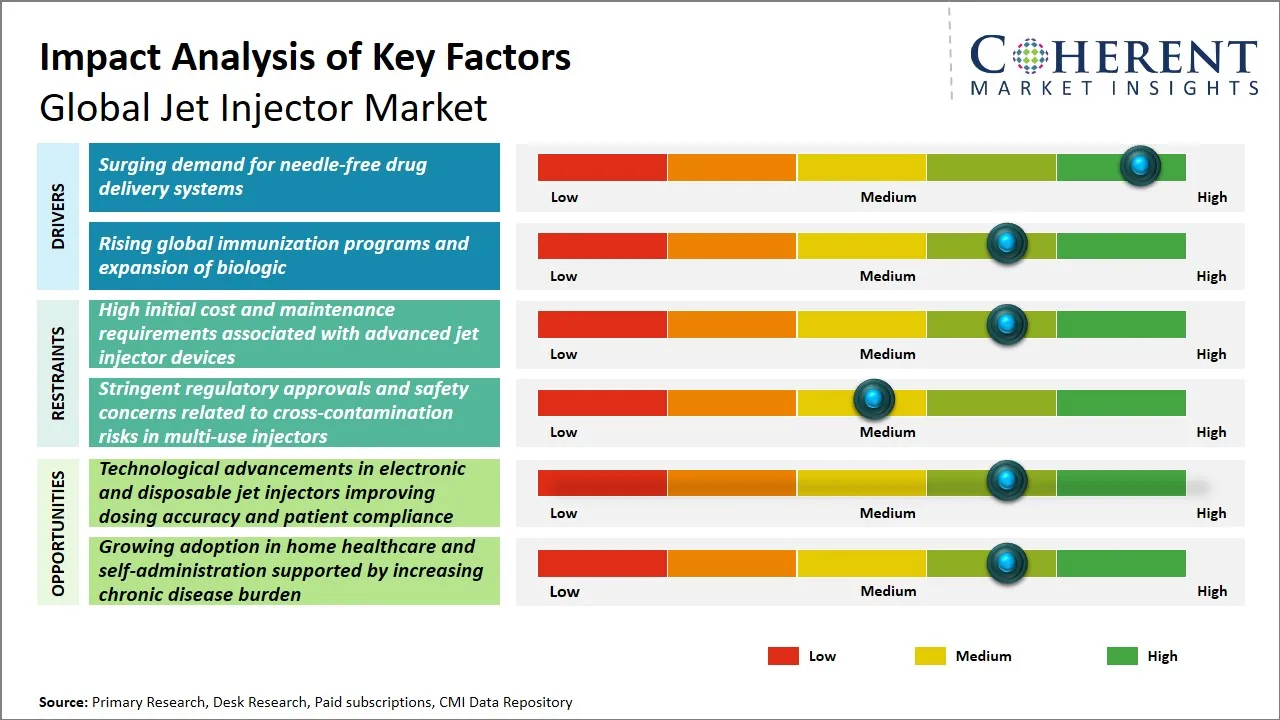
Global Jet Injector Market Driver - Surging Demand for Needle-Free Drug Delivery Systems
The surging demand for needle-free drug delivery systems is a major factor that is greatly contributing to the growth of the jet injector market in the overall healthcare industry. Patients, as well as medical practitioners, are look for alternatives that are not only pain-free, eliminate the problem of needle phobia, and minimize the chances of needlestick injuries as well as cross-contamination.
The jet injector, which makes the best use of high-pressure jets of drugs that are injected into the skin without the help of needles, is the best solution that meets the needs of the hour, as they are not only safe but are also pain-free. In addition, the rise of various diseases that require frequent injection, as well as vaccination, has created a need for effective drug delivery systems, which has been met by the technological advancements that have made jet injectors not only efficient, effective, as well as accurate.
For instance: In November 2024, Massachusetts Institute of Technology (MIT) and Novo Nordisk scientists designed microjet capsules based on squid, which can inject insulin, GLP-1, and RNA-based medicines into the GI tissue with the help of jet propulsion technology inspired by cephalopods, similar to the efficacy of subcutaneous injection with the absence of needles. This helps to improve patient compliance with macromolecule-based therapies.
(Source: Massachusetts Institute of Technology (MIT)
Global Jet Injector Market Opportunity - Technological Advancements in Electronic and Disposable Jet Injectors Enhancing Dosing Accuracy and Patient Compliance
The jet injector market is growing at a rapid pace globally, driven by the technological developments in electronic as well as disposable jet injectors, which improve the accuracy of the dose administered, thereby increasing the overall patient compliance. The electronic jet injectors now employ microprocessor-based control systems, along with smart sensors, that enable the administration of highly accurate doses, reducing the overall variability of the dosing process, which is inherent with mechanical jet injectors.
In addition, the overall flexibility of the dosing, along with the overall ergonomic design, is making the jet injectors accessible to a wide range of patients, including children as well as the elderly. Furthermore, the increased adoption of disposable jet injectors is helping to overcome the long-standing issues related to hygiene and cross-contamination, especially in the realm of both clinical and home healthcare services. The use of disposable jet injectors has improved safety and made it easier to administer injections, which has led to better patient adherence to their prescribed medications.
For instance, in June 2025, PharmaJet announced that the company had signed a clinical development agreement with Immuno Cure to utilize its needle-free injection system (Tropis) to deliver its HIV therapeutic DNA vaccine (ICVAX) in human clinical trials. This announcement pointed to an increased emphasis within the pharmaceutical industry on the development of safe and efficient ways of delivering drugs.
(Source: PharmaJet)
Analyst Opinion (Expert Opinion)
- The popularity of jet injector is on the rise as the demand for needle-free drug delivery systems is on the increase. Jet injectors make use of high-pressure columns of liquids for injecting drugs into the bodies of patients. This method is more comfortable and secure for patients since there is no possibility of prick accidents.
- The rise in the occurrence of chronic diseases, such as diabetes and autoimmune diseases, for which patients require injections, has increased the demand for needle-free systems. This has made patients seek alternative, painless, and easier methods of treatment, thus encouraging medical practitioners to make use of advanced injection systems, such as jet injectors.
- Furthermore, the research activities being carried out on the accuracy of injections, dosages, and safety of injectors are strengthening the technological advancement of jet injectors. The development in microjet and high-pressure technology has increased the possibility of using jet injectors in various applications.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2021 - 2033)
- Reusable Jet Injectors
- Disposable Jet Injectors
- Technology Insights (Revenue, USD Bn, 2021 - 2033)
- Spring Loaded Jet Injectors
- Gas Powered Jet Injectors
- Laser Powered Jet Injectors
- Application Insights (Revenue, USD Bn, 2021 - 2033)
- Vaccine Delivery
- Insulin Delivery
- Pain Management
- Other drug delivery applications
- End User Insights (Revenue, USD Bn, 2021 – 2033)
- Hospitals and Clinics
- Home Care Settings
- Research Laboratories
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- PharmaJet
- CROSSJECT
- Portal Instruments Inc
- NuGen Medical Devices
- Medical International Technology MIT Canada
- INOVIO Pharmaceuticals
- Aijex Pharma International Inc
- IntegriMedical
- Micron Biomedical Inc
- Halozyme Inc
Sources
Primary Research Interviews
Industry Stakeholders
- Medical device manufacturers
- Pharmaceutical and biotechnology companies
- Vaccine manufacturers and immunization program coordinators
- Healthcare professionals (physicians, nurses, vaccination specialists)
- Regulatory and compliance experts
- Distributors and suppliers of drug delivery devices
End Users
- Hospitals and clinics
- Vaccination centers and public health organizations
- Home healthcare users
- Research and clinical trial organizations
- Specialty care centers
Government and International Databases
- World Health Organization – Immunization and vaccination programs
- Centers for Disease Control and Prevention – Vaccination coverage and disease surveillance
- U.S. Food and Drug Administration – Medical device approvals and guidelines
- National Institutes of Health – Clinical research and funding
Trade Publications
- BioPharma Dive
- Pharmaceutical Technology
- Medical Device Network
- Fierce Pharma
- MedTech Dive
Academic Journals
- Journal of Controlled Release
- Drug Delivery and Translational Research
- International Journal of Pharmaceutics
- Advanced Drug Delivery Reviews
Reputable Newspapers
- The New York Times – Health Section
- The Guardian – Science & Health
- Financial Times – Healthcare
- The Wall Street Journal – Health & Medicine
Industry Associations
- Advanced Medical Technology Association (AdvaMed)
- Biotechnology Innovation Organization (BIO)
- International Federation of Pharmaceutical Manufacturers & Associations (IFPMA)
Public Domain Resources
- ClinicalTrials.gov
- WHO Reports and Fact Sheets
- CDC Publications
- NIH Publications
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients