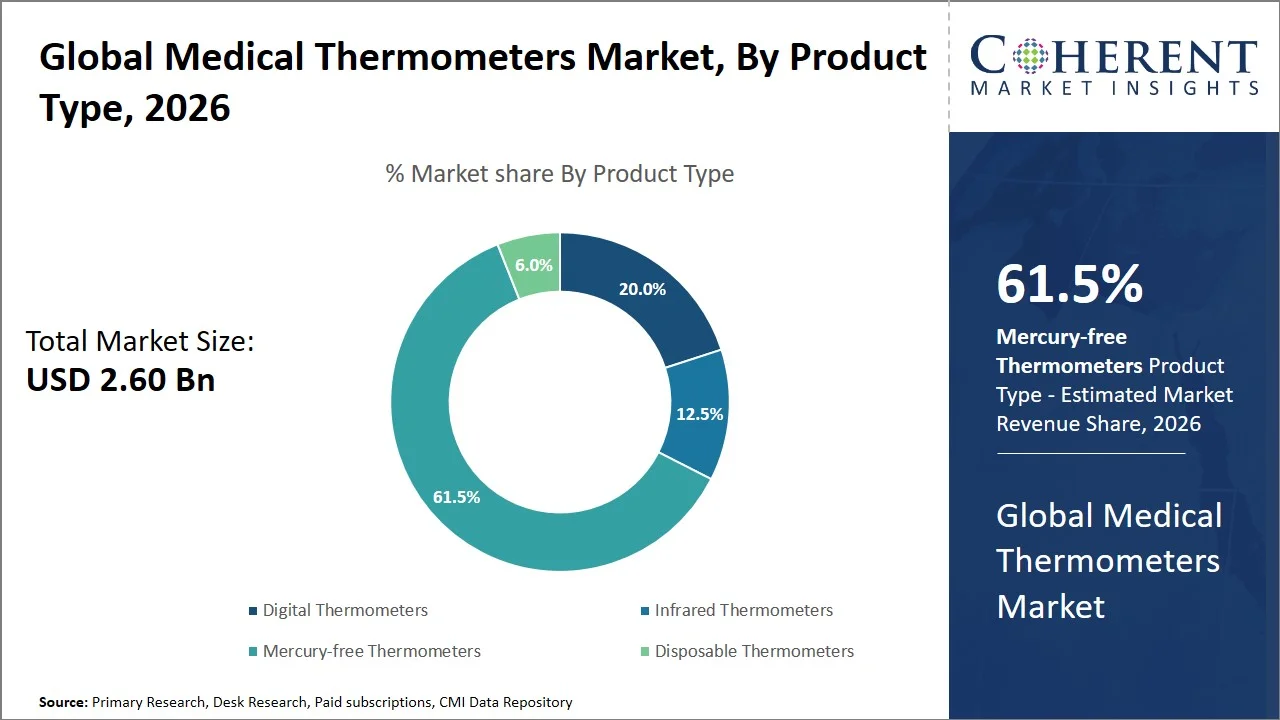
Global Medical Thermometers Market Size and Forecast: 2026-2033
The global medical thermometers market is expected to grow from USD 2.60 Bn in 2026 to USD 4.69 Bn by 2033, registering a compound annual growth rate (CAGR) of 8.8%. The market for global medical thermometers are poised for significant expansion, fueled by soaring burden of communicable diseases globally.
According to the World Health Organization, communicable diseases continue to impose a significant global burden, with 1.3 million new HIV infections and 10.7 million tuberculosis cases reported worldwide in 2024, highlighting the critical need for routine temperature monitoring tools for early detection and disease management.
(Source: World Health Organization)
Key Takeaways of the Global Medical Thermometers Market
- Mercury-free thermometers are projected to hold 61.5% of the global medical thermometers market share in 2026, making it the dominant product type segment, with strong dominance in Europe primarily because of strict regulations surrounding the use of mercury-based medical instruments, forcing healthcare facilities to adopt safer technology like digital and infrared thermometers. For instance, under directive 2007/51/EC, the sale of fever thermometers and other measuring instruments containing mercury is not allowed within the European Union for use by consumers. This directive has resulted in the phasing out of mercury thermometers in healthcare facilities in the European Union, hastening the adoption of digital and infrared thermometers. (Source: European Union)
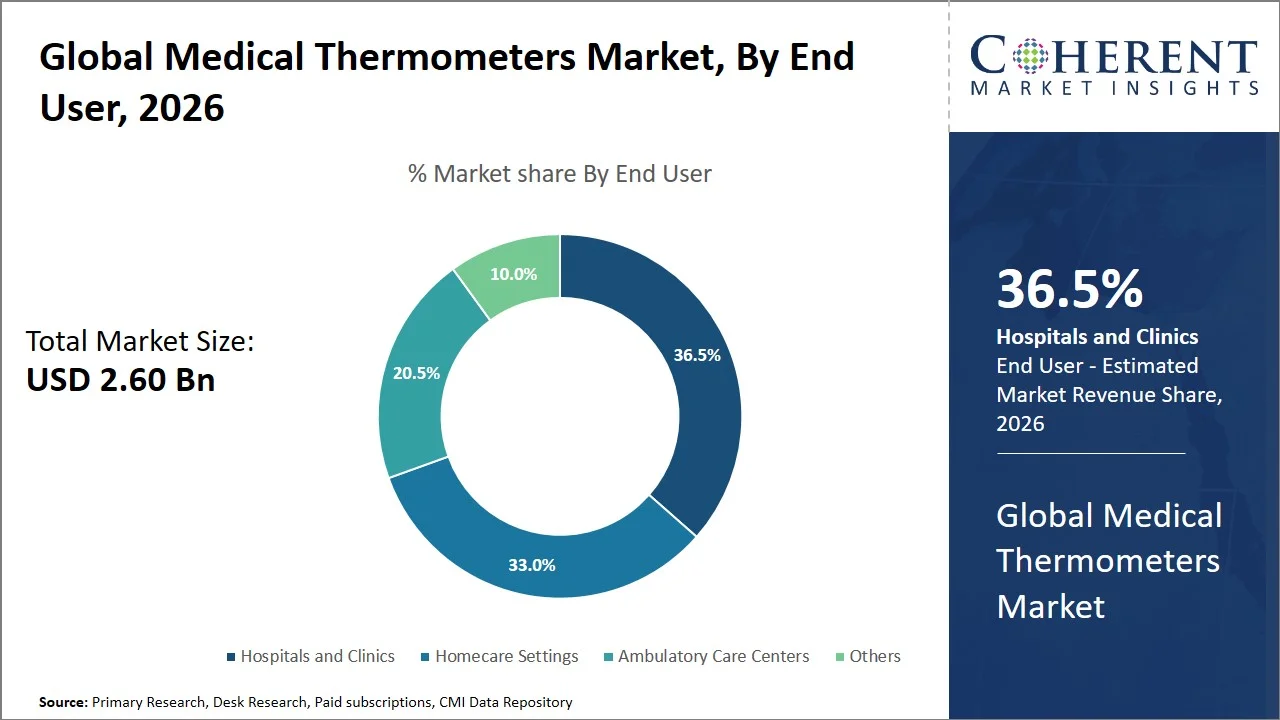
- Hospitals and clinics are projected to hold 36.5% of the global medical thermometers market share in 2026, making it the dominant end user segment, with dominance in North America due to advanced healthcare infrastructure and high patient volume (139.8 million emergency department visits annually in the U.S.). The segment benefits from the requirement of continuous monitoring, stringent infection control measures (such as routine vital sign monitoring, patient triaging protocols, isolation screening, and prevention of hospital-acquired infections (HAIs)), and standardization in clinical processes, requiring frequent temperature checks. For instance, according to the Centers for Disease Control and Prevention, the guidelines for the prevention and control of infections include regular monitoring of vital signs, such as body temperature, in order to detect the presence of infections and prevent hospital-acquired infections (HAIs). This necessitates constant use of thermometers in in-patient and out-patient facilities, thereby increasing demand in hospitals and clinics. (Source: Centers for Disease Control and Prevention)
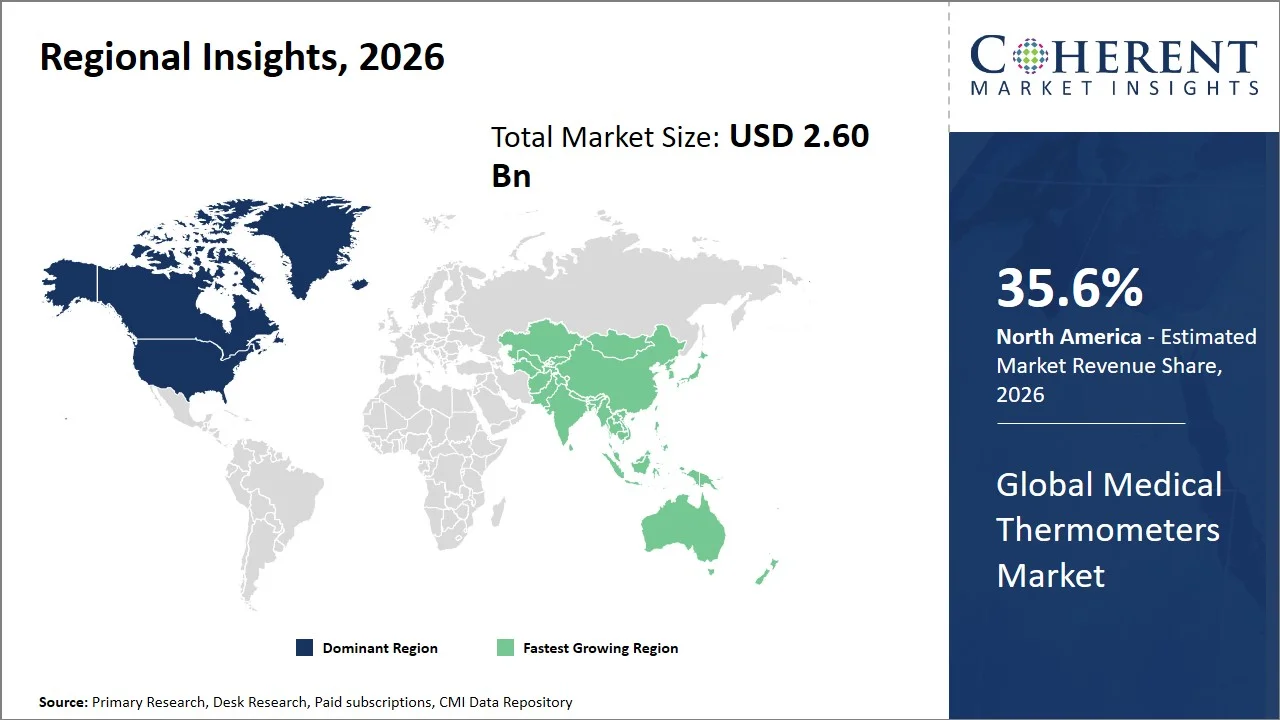
- North America maintains dominance with an expected share of 35.6%in 2026, backed by stringent infection control policies (such as routine vital sign monitoring, patient triaging protocols, isolation screening, and prevention of hospital-acquired infections (HAIs)), and high prevalence of healthcare-acquired infections (HAIs), that require continuous patient monitoring. For instance, according to Centers for Disease Control and Prevention, about 1 in 31 hospitalized patients in the U.S. suffers from a healthcare-associated infection on any given day, which requires the adoption of stringent practices in monitoring infections, especially the need for regular temperature screening. This ensures the constant demand for medical thermometers across hospitals and clinical settings. (Source: Centers for Disease Control and Prevention)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 32.5% share in 2026, driven by large-scale public health screening programs and the expansion of primary healthcare services in high-population countries like India and China. For instance, Integrated Disease Surveillance Programme (IDSP) by the National Centre for Disease Control is a real-time monitoring of epidemic-prone diseases, including fever-based conditions like dengue and chikungunya, by systematic temperature screening and reporting at the district and state levels, which has led to a substantial increase in the use of thermometers in public healthcare systems. (Source: National Centre for Disease Control)
- Expansion of Continuous & Wearable Temperature Monitoring in Remote Care: With the rising adoption of remote patient monitoring (RPM) and telemedicine services, there is an increasing demand for wearable devices capable of tracking body temperature in real-time. Compared to traditional thermometers, smart sensors provide a constant channel of communication that can be very useful in the treatment of chronic diseases, post-operative recovery, and elderly people. Moreover, high compatibility with mobile apps, AI-powered alerts, and early-stage disease detection add to the attractiveness to the medical thermometers market.
- Rising Demand for Non-Contact Infrared Thermometers in Infection Control Protocols: The post-COVID healthcare and public health measures have resulted in the use of non-contact infrared thermometers being common in hospitals, airports, work places, and schools. Non-contact infrared thermometers offer benefits such as minimal risks of contamination and minimized time required for handling patients. The increase in the demand from developing countries within the Asia Pacific region, Latin America, and Africa will be driven by health and public safety drives funded by governments. Advancements in sensors and declining cost will play a significant role in sustaining this market niche as an engine for growth.
Why Does the Mercury-free Thermometers Segment Dominate the Global Medical Thermometers Market?
Mercury-free thermometers are projected to hold a market share of 61.5% in 2026, owing to the policy-led transition away from hazardous materials and a clear shift in healthcare procurement toward safer, compliant devices. International agreements like the Minamata Convention have pushed for the elimination of mercury-containing medical equipment, leading to an increasing replacement of traditional thermometers with digital and infrared alternatives. (Source: Minamata Convention on Mercury)
Moreover, the segment’s dominance is further enhanced through institutional replacement cycles and changing trends in medicine, which emphasize infection prevention and greater efficiency in operations. The health practitioners' preference for mercury-less thermometers is based on their suitability with the guidelines of hygiene, lack of contamination risks, and ease of maintenance. The technological advancements in sensing systems and the availability of easily operable digital thermometers further promote the usage of mercury-less thermometers.
For instance, Omron Healthcare has developed state-of-the-art digital and infrared thermometers, including the MC-720 forehead thermometer, which employs non-contact infrared sensing technology to give accurate readings in one second, thus increasing efficiency and maintaining hygienic conditions in clinical as well as home settings. (Source: Omron Healthcare)
Why do Hospitals and Clinics Represent the Largest End User Segment in the Medical Thermometers Market?
To learn more about this report, Request Free Sample
Hospitals and clinics are projected to hold a market share of 36.5% in 2026, due to the standardized and frequent necessity for the measurement of body temperature that is involved in triage, inpatient examination, surgical interventions, and infection detection processes. Body temperature testing is an obligatory element of patient examination procedure that needs to be performed several times per day, thus increasing usage rates in institutional environments. Moreover, continuous monitoring of temperature levels plays a key role in the detection of fever symptoms and their consequences.
The segment’s dominance is further reinforced through clinical guidelines and protocols that insist upon constant temperature monitoring for diagnostics and monitoring purposes. For instance, the World Health Organization in its Integrated Management of Childhood Illness (IMCI) guidelines highlights the importance of conducting fever diagnosis and insists on taking the patient’s temperature. (Source: World Health Organization) Likewise, the National Institute for Health and Care Excellence (NICE) guidelines emphasize on measuring vital signs, including body temperature, in order to determine the severity of an acute illness. (Source: National Institute for Health and Care Excellence) Such standardized guidelines and practices ensure the continued and consistent use of thermometers, thereby cementing their segment’s supremacy in the medical thermometers market.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Exemption Update for Clinical Electronic Thermometers (2025 – Ongoing Implementation) |
|
|
EU MDR 2025 Amendment Strengthening Supply & Transparency Requirements |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Federal Register, European Commission)
Global Medical Thermometers Market Dynamics
To learn more about this report, Request Free Sample
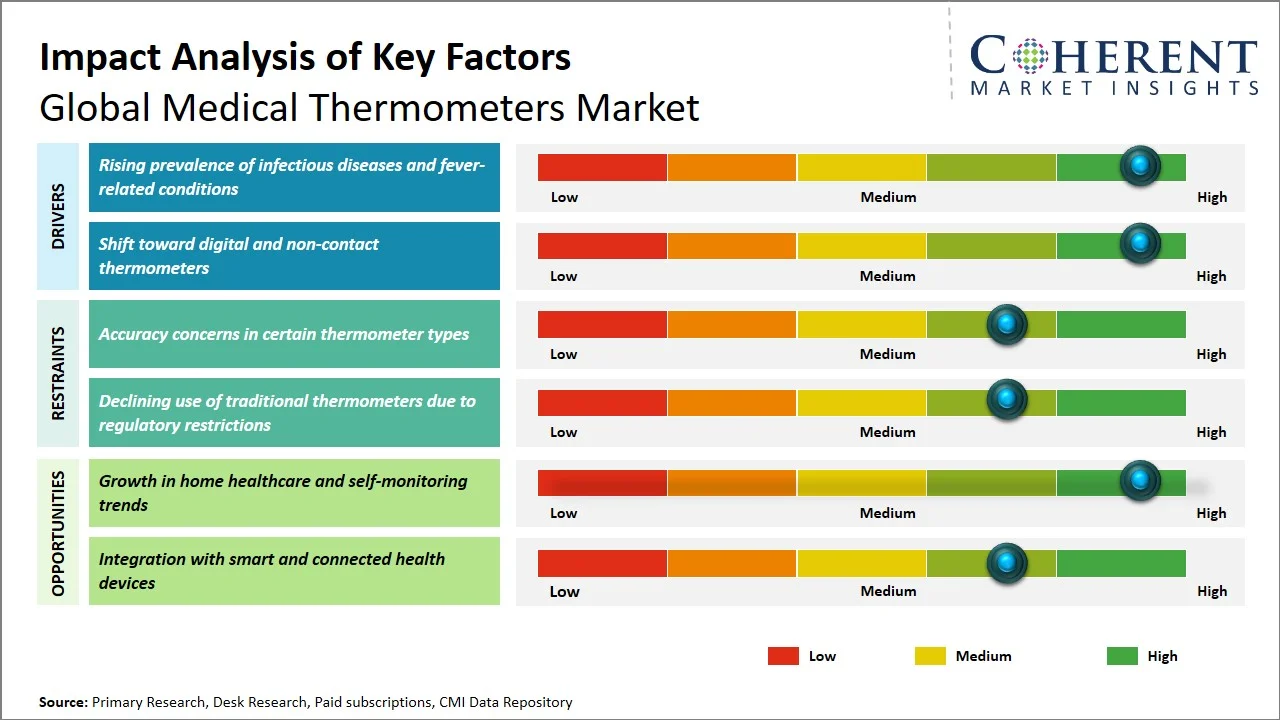
Market Drivers
- Rising prevalence of infectious diseases and fever-related conditions: The industry is spurred by an increasing incidence of infectious disease and fever-related ailments, as fever is still the most prevalent symptom that requires clinical examination. According to the World Health Organization, vector-borne diseases alone contribute to more than 17% of all infectious diseases and lead to over 700,000 fatalities each year, with diseases like malaria (249 million cases) and dengue (96 million symptomatic cases) being commonly associated with fever. (Source: World Health Organization) Additionally, typhoid fever causes about 9 million cases and more than 110,000 deaths annually, which reinforces the prevalence of diseases characterized by fever. (Source: World Health Organization). The increasing incidence of fever-related illness will require regular temperature measurement to detect and manage such diseases, hence contributing to increased demand for medical thermometers across healthcare settings.
- Shift toward digital and non-contact thermometers: There is a distinct trend toward digital and contactless thermometers in the marketplace due to increased safety measures, improved performance, and flexibility in adapting to non-infectious environments. Compared to conventional mercury thermometers, the latest thermometers offer enhanced protection from toxic materials, as well as taking one’s body temperature without having to come into contact with anything, thus reducing chances of infections spreading in the clinic. For instance, U.S. Food and Drug Administration, highlights the increasing use of non-contact infrared thermometers in fast temperature checks, especially in circumstances that require minimal physical contact. (Source: S. Food and Drug Administration) Moreover, there is an increased trend among health practitioners towards using digital thermometers owing to their compatibility with electronic health records systems and portability, making it easier to monitor patients efficiently through hospitals and even at home care centers. Such trends have contributed to increasing demands for digital and infrared thermometers globally.
Emerging Trends
- Integration of Multi-Parameter Monitoring in Thermometer Devices: Medical thermometers are transitioning from single-parameter monitors to multi-functional devices that can measure other vital signs such as pulse rate, respiration rate and SpO₂ levels. This trend is fueled by the surging demand for miniaturized and integrated systems combining various functionalities into one small device. Such a demand is driven by the need for a more efficient assessment during triage in hospitals or at home using portable health kits.
- Growing Shift Toward Eco-Friendly and Mercury-Free Thermometer Alternatives: Environmental concerns regarding mercury pollution and laws have led to mercury-free thermometers being the better option available today. This is because mercury thermometers have been largely phased out due to the increased environmental threats associated with handling hazardous wastes. Manufacturers of mercury thermometers have spent money on ensuring that their products are environmentally-friendly, recycled, and energy efficient.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Medical Thermometers?
North America leads the global medical thermometers market, accounting for an estimated 35.6% share in 2026, attributed to its well-developed healthcare system and effective infection control practices. The healthcare facilities in North America comply with the laid down regulations that prescribe routine monitoring of essential vital signs, including body temperature, in order to detect possible sources of infections and treat them. Additionally, infection prevention and control measures have been recognized by the Centers for Disease Control and Prevention as essential components of quality care, and as such, they require proper monitoring and resource allocation to mitigate the risk of infections. (Source: Centers for Disease Control and Prevention)
Moreover, the region has embraced modern temperature monitoring systems, such as zero contact thermometers and infrared devices, to enhance patient flow and prevent cross-contamination. For instance, the growing utilization of zero-contact infrared thermometers within healthcare institutions in the U.S. facilitates quick triage and follows infection control procedures within high-volume settings. (Source: TriMedika) The advancements, together with the robust infrastructure and rigorous clinical protocols, have continued to strengthen North America’s dominance in the global medical thermometers market.
Why Does the Asia Pacific Medical Thermometers Market Exhibit High Growth?
The Asia Pacific medical thermometers market is expected to exhibit the fastest growth with an estimated contribution of 32.5% share to the market in 2026, attributed to the high prevalence of infectious diseases (South-East Asia accounts for ~45% of global tuberculosis cases and a significant share of dengue and malaria burden), and an increase in government-run screening and primary health care initiatives (such as, Integrated Disease Surveillance Programme (IDSP) in India; National Basic Public Health Service Program in China) within the highly populated nations of the region such as India, and China.
The World Health Organization has highlighted that communicable diseases have continued to exert a considerable burden on the world, necessitating regular checks. For instance, in February 2026, World Health Organization identified a positive case of Nipah virus infection in Bangladesh where the infected individual exhibited symptoms such as fever and neurological manifestations, and several contacts were placed under surveillance. (Source: World Health Organization) The structured and high volume of screening processes, along with the improvement in the healthcare infrastructure, will keep on generating robust demand for medical thermometers in the Asia Pacific region.
Global Medical Thermometers Market Outlook for Key Countries
Why is the U.S. Leading Innovation and Adoption in the Medical Thermometers Market?
The U.S. emerges as an innovation hub and adopter in the medical thermometers market owing to its swift incorporation of connected health technologies into its clinical processes as well as having robust regulations on the use of advanced diagnostic devices (such as U.S. FDA 510(k) premarket clearance pathway; U.S. FDA Quality System Regulation (21 CFR Part 820); Unique Device Identification (UDI) system). There is widespread use of non-contact infrared thermometers and digital connected devices that connect with electronic health record systems, thereby facilitating clinical decision-making and monitoring of patients. Technology integration, regulatory facilitation, and clinical digitization together put the U.S. in a favorable position to lead innovation and widespread adoption within the global medical thermometers market.
Is Japan a Favorable Market for Medical Thermometers?
Japan emerges as an ideal market for medical thermometers owing to the demographic composition of the country as well as the high frequency of routine health monitoring. Japan boasts of an advanced healthcare sector where the use of both digital and infrared thermometers is widespread in hospitals and homes. Moreover, because of the nation’s focus on prevention of diseases, the need for regular temperature checks becomes increasingly important in order to detect any illness early. Furthermore, the presence of leading manufacturers and continuous innovation in compact, high-precision devices (such as., Omron Healthcare, Terumo Corporation, Citizen Systems Japan) further strengthens the market growth in the country.
Is China Emerging as a Key Growth Hub for the Medical Thermometers Market?
China stands out as one of the most prominent countries with respect to the future growth of the medical thermometers market owing to country's effective public health system, and rising focus on monitoring and preventing diseases. Routine temperature testing has been incorporated into primary healthcare and community health screenings, thus greatly enhancing the usage of such devices in both urban and rural areas. Production capacities for both digital and infrared medical thermometers in addition to low cost of production have played their role in attaining this objective. Furthermore, government-sponsored programs that were directed towards boosting the growth of hospitals within the region have assisted in promoting these thermometers. For instance, Healthy China 2030 strategy emphasizes enhancing the capacity of hospitals, developing primary health care networks, and adopting preventive care strategies, which include routine patient monitoring such as temperature checks. (Source: ScienceDirect)
Why Does Germany Top the Europe Medical Thermometers Market?
Germany is the leader in the Europe market for medical thermometers due to the organized nature of the health care industry in the nation, which makes it necessary to have stringent clinical monitoring procedures that will ensure that there is an extensive use of diagnostic tools in the healthcare sector. Germany derives advantage from the compulsory infection control measures and standard assessment processes that require the use of temperature monitors, hence ensuring constant use of thermometers. Additionally, with its vast number of medical device manufacturing companies (such as., B. Braun, Beurer GmbH) and high-quality healthcare devices, Germany is poised to embrace advanced thermometers in healthcare settings.
Is the Medical Thermometers Market Developing in India?
The market for medical thermometers is rapidly growing in India attributed to the expanding public healthcare infrastructure, and growing emphasis on prevention and early diagnosis. Public health programs such as the Ayushman Bharat initiative are reinforcing the process of improving healthcare through the use of Health and Wellness Centers, where regular monitoring of important health parameters, including body temperature monitoring, is carried out.
Moreover, the National Vector Borne Disease Control Program targets vector-borne diseases such as malaria and dengue, which necessitate fever-based testing and surveillance both in the community and at hospitals. (Source: Ministry of Health and Family Welfare) The transition towards digital and mercury-free devices, along with increasing adoption of home healthcare, is further driving the market growth in India.
Regulatory Landscape Governing the Global Medical Thermometers Market
|
Region
|
Key Regulatory Bodies |
Framework |
Impact on Market |
|
North America |
U.S. Food and Drug Administration (U.S.), Health Canada |
FDA 21 CFR (e.g., 880.2910), 510(k) pathway; ISO 13485; ASTM standards |
High regulatory rigor ensures product accuracy and safety, but increases time-to-market; recent exemptions improve innovation and entry for basic devices |
|
Europe |
European Commission, Notified Bodies |
EU Medical Device Regulation (MDR) 2017/745, CE Marking; EN/ISO standards |
Stringent MDR increases compliance costs and documentation, but enhances traceability, transparency, and patient safety |
|
Asia Pacific |
NMPA (China), CDSCO (India), PMDA (Japan), TGA (Australia) |
Country-specific frameworks; e.g., India Medical Device Rules 2017, China NMPA regulations |
Regulatory fragmentation creates market entry complexity, but supportive policies and local manufacturing incentives drive regional growth |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico), INVIMA (Colombia) |
Country-specific frameworks aligned with global standards (FDA/EU MDR references) |
Regulatory evolution improving market standardization, but bureaucratic processes may delay approvals |
|
Middle East & Africa |
SFDA (Saudi Arabia), SAHPRA (South Africa), MoH authorities |
Mix of local regulations + reliance on EU CE / FDA approvals |
Moderate regulatory stringency; reliance pathways enable faster product approvals, but limit domestic innovation ecosystems |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is growth in home healthcare and self-monitoring trends creating new growth opportunities in the global medical thermometers market?
The trend towards increasing adoption of home healthcare and self-care has led to potential growth avenues in the global medical thermometers market through turning the temperature testing process into an integral and convenient aspect of daily health assessment performed at home. The growing trend towards telehealth consultations has contributed to the increased demand for easy-to-use, reliable, and responsive digital thermometers that consumers can use at home.
For instance, QuickCare Smart Thermometer from Kinsa Inc. is an illustration of how telemedicine technology can support the delivery of quality care. This product communicates with smartphones using the Bluetooth technology to connect with the mobile application that records the body temperature and other symptoms, medications, and illnesses in relation to individual family members. (Source: Kinsa Inc.) These advancements underscore the importance of thermometers becoming increasingly interconnected devices rather than standalone diagnostic equipment, which underlines their significance in decentralized healthcare systems and promotes consistent growth within the global medical thermometers market.
Market Players, Key Development, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- In December 2025, Smart Meter introduced its new invention of iDigiTemp – the world's first cellular-enabled thermometer designed for remote patient monitoring. The product records and delivers real-time temperature data through cellular connection without the use of Wi-Fi or Bluetooth connectivity. This breakthrough will revolutionize healthcare technology by making remote patient monitoring simpler and more reliable.
- In November 2025, Withings launched a high-tech multifunctional thermometer with integrated temperature, ECG, and stethoscope functionality. This development reflects the trend towards intelligent thermometers, which contribute to the transformation of the medical thermometer industry.
Competitive Landscape
The global medical thermometers market will likely stay highly competitive due to continuous innovation and significant competition from established companies in the medical devices sector. Companies in the medical thermometers market is focusing on innovating advanced digital and infrared thermometers that are very precise, efficient, and easy to use, to create a competitive edge for themselves. Innovation, product launches, and market expansion will define the competitive structure of this industry. Moreover, affordability, regulatory compliance, and connectivity with health technology platforms will also give companies an advantage. Key focus areas include:
- Advancement in non-contact and infrared sensing technologies
- Development of smart and connected thermometers (app/EHR integration)
- Product innovation in accuracy, response time, and user-friendly design
- Expansion into emerging markets through cost-effective solutions
- Compliance with regulatory standards and shift toward mercury-free devices
Market Report Scope
Medical Thermometers Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.60 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 8.8% | 2033 Value Projection: | USD 4.69 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Omron Corporation, Terumo Corporation, Exergen Corporation, Microlife Corporation, A&D Company, Limited, Geratherm Medical AG, BPL Medical Technologies, Rossmax International Ltd., Welch Allyn, and 3M |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the medical thermometers market will depend on the trends associated with the shift towards intelligence, connectivity, and touchless technology in the process of measuring body temperatures due to the rising focus on infection prevention, self-diagnosis, and technology utilization. The future of medical thermometers will therefore lie in their incorporation into telemedicine applications, as patient monitoring and preventive measures are becoming increasingly significant.
- Most opportunities are envisaged in the segment of digital and non-contact thermometer devices in the Asia Pacific region, especially in China and India, where there is high prevalence of infectious diseases coupled with expansion of the primary healthcare sector, which is leading to the creation of huge demand. It would be worthwhile for the industry to invest in community-level and domestic screening technology.
- For gaining any competitive advantage in the market, companies need to focus on innovations in their products related to Internet of Things (IoT), making them more effective and accurate while conforming to the new standards for achieving a competitive advantage. In addition, companies may consider local production, collaboration with hospitals, and exploring government purchasing markets. The importance of cost-effective and easy-to-use devices along with their inclusion in digital health care solutions cannot be understated.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2021 - 2033)
- Digital Thermometers
- Infrared Thermometers
- Mercury-free Thermometers
- Disposable Thermometers
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals and Clinics
- Homecare Settings
- Ambulatory Care Centers
- Others
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Omron Corporation
- Terumo Corporation
- Exergen Corporation
- Microlife Corporation
- A&D Company, Limited
- Geratherm Medical AG
- BPL Medical Technologies
- Rossmax International Ltd.
- Welch Allyn
- 3M
Sources
Primary Research Interviews
- Medical device manufacturers and product managers from leading thermometer companies
- Hospital procurement managers and clinicians (pediatricians, general physicians) utilizing thermometers in routine diagnosis
- Distributors and suppliers involved in retail and e-commerce sales of digital and infrared thermometers
- Telehealth platform providers and remote patient monitoring solution experts integrating home-use diagnostic devices
Stakeholders
- Medical Thermometer Manufacturers & Solution Providers (e.g., consumer health device companies, infrared sensor manufacturers, digital health solution providers)
- End-use Sectors:
- Hospitals and Clinics
- Home Healthcare Users
- Diagnostic Centers
- Ambulatory Care Settings
- Regulatory & Health Bodies: U.S. Food and Drug Administration – device approvals and classification standards, European Commission – Medical Device Regulation (MDR) compliance, World Health Organization – medical device safety guidelines and mercury-free initiatives, Central Drugs Standard Control Organization – medical device registration and compliance in India
Databases
- FDA Product Classification Database – device classification and regulatory pathways
- EUDAMED (European Database on Medical Devices) – device registration and tracking
- WHO Global Health Observatory – healthcare infrastructure and device usage insights
- OECD Health Statistics – healthcare access and medical device utilization data
Magazines
- Medical Device Network – coverage on device innovation and regulatory updates
- MedTech Dive – insights on digital health devices and wearable monitoring trends
- Healthcare Business Today – industry developments in diagnostic and home healthcare devices
Journals
- Journal of Medical Devices
- BMJ Innovations
- Journal of Clinical Monitoring and Computing
- Sensors (MDPI) – research on infrared and digital sensing technologies
Newspapers
- Financial Times – coverage on global healthcare investments and device markets
- The Wall Street Journal – reporting on medical device companies and innovation
- The Economic Times (India) – updates on healthcare infrastructure and device demand in India
- Business Standard (India) – coverage on domestic medical device manufacturing and policy
Associations
- Advanced Medical Technology Association (AdvaMed)
- Medical Device Manufacturers Association (MDMA)
- European Medical Technology Industry Association
- Association for the Advancement of Medical Instrumentation
Public Domain Sources
- Centers for Disease Control and Prevention – fever screening and infection monitoring guidelines
- National Institutes of Health – research on diagnostic technologies
- Ministry of Health and Family Welfare – public healthcare programs and device usage
- European Commission – funding and regulatory initiatives in medical devices
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients