Global Microbiome Therapeutics Market Size and Forecast – 2026 To 2033
The global microbiome therapeutics market is expected to grow from USD 57.4 Mn in 2026 to USD 310.6 Mn by 2033, registering a compound annual growth rate (CAGR) of 27.3%. The market for microbiome therapeutics is poised for significant expansion, fueled by rising global burden of antimicrobial resistance (AMR).
A study in The Lancet reports that antimicrobial resistance (AMR) was linked to an estimated 4.71 million deaths around the world in 2021, out of which 1.14 million have been caused by antimicrobial resistance (AMR) directly. Estimates suggest that deaths from antimicrobial resistance (AMR) will increase to nearly 1.91 million per year by 2050, with total deaths being around 8.22 million.
(Source: The Lancet)
Additionally, the World Bank estimates that antimicrobial resistance (AMR) could result in USD 1 trillion in additional healthcare costs by 2050 and annual global GDP losses ranging from USD 1 trillion to USD 3.4 trillion by 2030, highlighting its severe economic impact.
(Source: World Bank)
Key Takeaways of the Global Microbiome Therapeutics Market
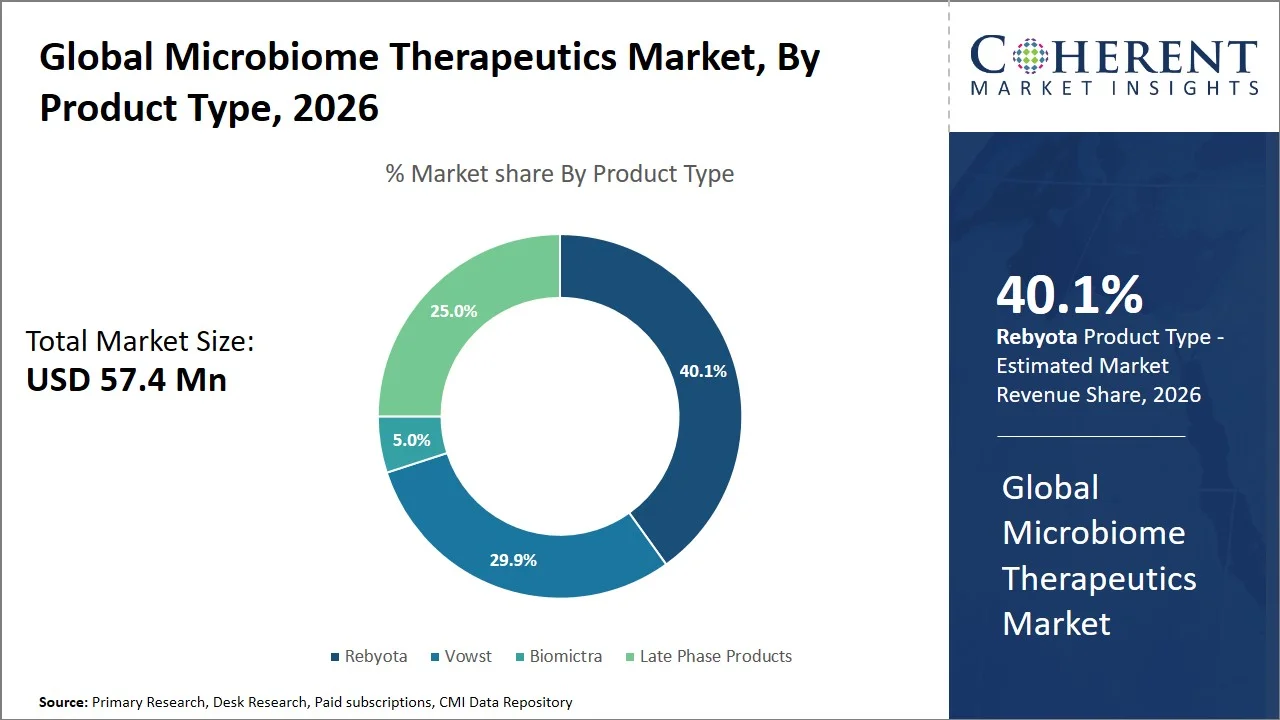
- Rebyota is projected to hold 40.1% of the global microbiome therapeutics market share in 2026, making it the dominant product type segment, especially in the U.S, owing to its first-mover advantage as the first U.S. FDA-approved microbiome drug and widespread use in the treatment of recurring infections. For instance, in November 2022, the U.S. Food and Drug Administration gave the green light to Rebyota, a product of Ferring Pharmaceuticals. This approval was significant, as it was the first microbiome-based therapy authorized in the U.S. The drug was designed to prevent the return of Clostridioides difficile infection in adults after they have been treated with antibiotics. (Source: Food and Drug Administration)
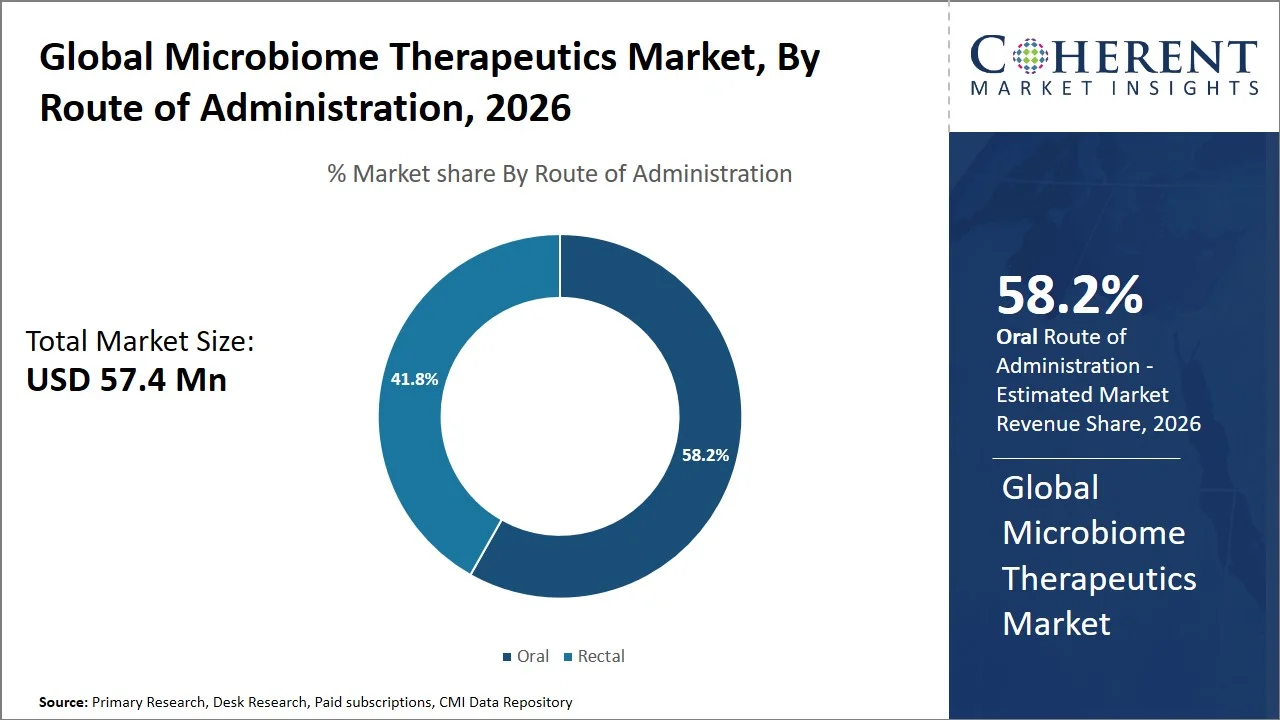
- Oral is projected to 58.2% of the global microbiome therapeutics market share in 2026, making it the dominant route of administration segment. North America and Europe are witnessing significant growth within the segment, fueled by enhanced patient compliance, non-invasive application, and growing number of approvals for oral microbiome drugs like Vowst. For instance, clinical trials evaluating Vowst showed significantly better patient outcomes and convenience. Specifically, the recurrence of Clostridioides difficile infection was about 12.4% with Vowst, compared to 39.8% with a placebo, at the eight-week mark. This indicates both strong effectiveness and better patient adherence, likely due to the oral, non-invasive method of administration. (Source: Food and Drug Administration)
- Hospitals segment is projected to hold 47.0%of the global microbiome therapeutics market share in 2026, making it the dominant care setting segment, having considerable dominance in North America because of the advanced infrastructure for healthcare facilities, greater uptake of recently launched biologics, and the requirement for controlled delivery of microbiome-based therapies in the clinical setting. For instance, the expansion of specialized healthcare and research facilities such as the Mayo Clinic and the establishment of dedicated microbiome research and treatment programs at institutions like Cleveland Clinic highlight the region’s strong clinical infrastructure.
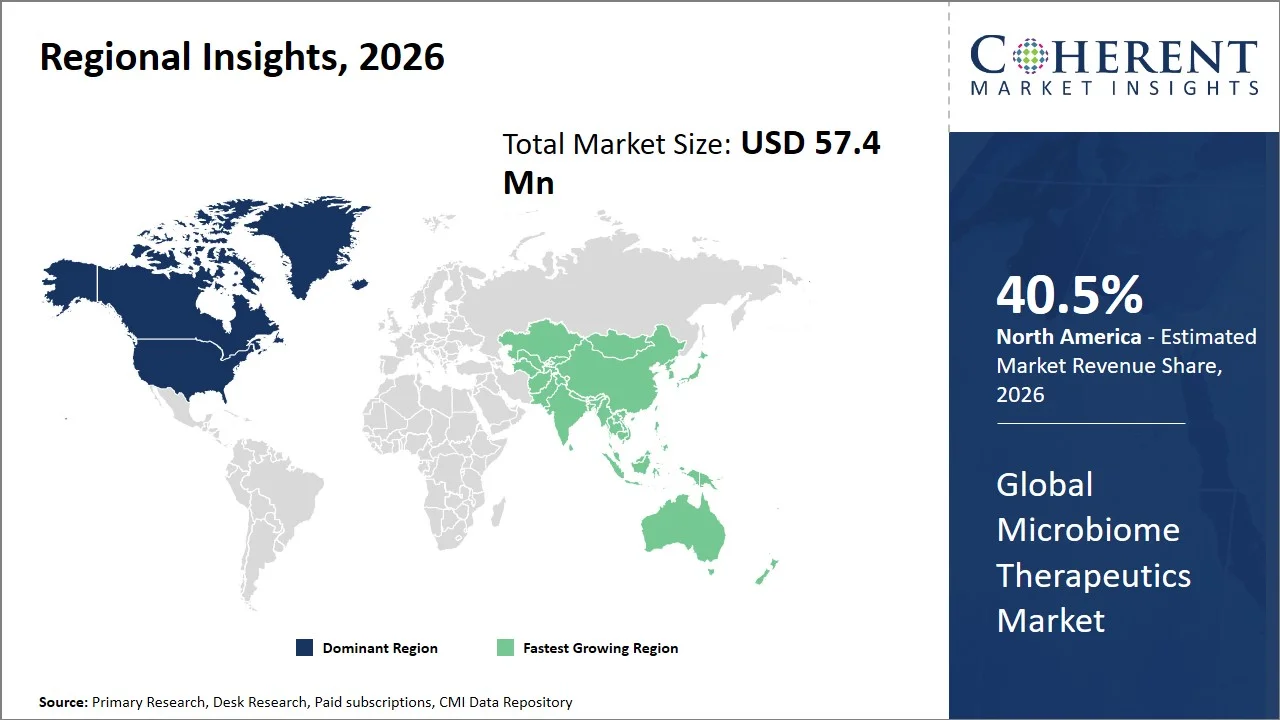
- North America maintains its dominance with an expected share of 40. 5% in 2026, primarily due to the early regulatory approval of microbiome-based treatments, robust research efforts, and increased use of sophisticated biological treatments. Also, the availability of a robust health care system, along with substantial microbiome-based research efforts supported by the government (e.g., Human Microbiome Project) helps to facilitate commercial success within the region.
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 22. 5% share in 2026, as a result of the increased emphasis by the government on biotechnology and precision medicine, increased capacity for conducting clinical trials, and a sizable patient pool, with an increasing number of patients with chronic diseases, advancements in regulatory infrastructure and funding towards genomics and microbiome research are propelling market growth. For instance, India's Genome India Project, a major precision medicine undertaking backed by the Department of Biotechnology, brings together over 20 institutions. The project has sequenced more than 10,000 genome samples, with the goal of creating a national genomic database. This database will serve as a resource for advanced therapeutics and microbiome research. (Source: Genome India)
- Expansion in Oncology as an Adjunct Therapy: Cancer immunotherapy is increasingly gaining attention as an area where microbiome therapeutics can improve outcomes. As a result, niches are emerging within combination therapy that focus on enhancing the efficacy of checkpoint inhibitors used in cancer treatments.
- Development of Oral Capsule-Based Microbiome Therapies: Moving from invasive fecal transplant procedures to standard oral capsule delivery is enhancing compliance and scaling. This development is creating niche areas for profitable and convenient microbiome-based medications.
Why Does Rebyota Dominate the Global Microbiome Therapeutics Market?
Rebyota is projected to hold a market share of 40.1% in 2026, substantiated by its established regulatory pathway and clinical validation as a standardized live biotherapeutic product (LBP), thereby establishing a model for subsequent microbiome-based therapies. The U.S. Food and Drug Administration (FDA) indicates that Rebyota is subject to biologics regulations, encompassing rigorous donor screening and manufacturing controls, which serve to uphold safety and quality benchmarks. (Source: U.S. Food and Drug Administration)
Furthermore, the increasing clinical challenges (such as high recurrence rates, antibiotic resistance, and limited effectiveness of conventional therapies) posed by recurrent infections, as underscored by the Centers for Disease Control and Prevention's focus on the widespread effects of healthcare-associated infections, has heightened the need for effective non-antibiotic interventions- where approximately 1 in 31 hospitalized patients, and 1 in 43 nursing home acquires at least one infection during care. (Source: Centers for Disease Control and Prevention) Consequently, this regulatory clarity and strong clinical evidence have fostered quicker acceptance and strengthened Rebyota's leadership position in the microbiome therapeutics market.
Why is Oral the Most Preferred Route of Administration in Global Microbiome Therapeutics Market ?
To learn more about this report, Download Free Sample
Oral is projected to hold a market share of 58.2% in 2026, owing to its easy patient acceptance, ease of application, and increasing regulatory support for orally administered microbiome treatments. The U.S. FDA approval of oral live biotherapeutic products, such as microbiome-containing capsules (Vowst), demonstrates that the gastrointestinal pathway is a feasible delivery route for viable bacteria. (Source: U.S. Food and Drug Administration)
Furthermore, in January 2026, research published by Springer Nature Limited has clinically shown that microbiome oral treatments are capable of rebalancing the intestinal flora and preventing infection recurrence. In fact, research shows that recurrence rates for C. difficile infection following antibiotic therapy could be as high as 20 to 30% in patients, hence the need for easy solutions (Source: Springer Nature Limited). These factors of simplicity, enhanced adherence rates, and clinical effectiveness have led to a clear preference for oral administration in the microbiome therapeutics market.
Hospitals Segment Dominates the Global Microbiome Therapeutics Market
The hospitals segment is projected to hold a market share of 47.0% in 2026, because of their prime importance in dealing with complicated medical cases where advanced therapies are required. Hospitals serve as key centers for the administration of therapies using the human microbiome. They also offer adequate infrastructure for the management of biological products such as storage, testing, and administration under controlled conditions. Additionally, the increased acceptance of newly approved therapies and improved reimbursement policies for hospitals (Medicare, Medicaid, and Diagnosis-Related Group (DRG) payment systems) makes hospitals a dominant segment.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Industry & Regulatory Push: Standardization of Microbiome Therapeutics (2025) |
|
|
Regulatory Evolution of Live Biotherapeutic Products (Regulatory + Industry Shift, 2024–2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Microbiome Therapeutics Innovation Group, U.S. Food and Drug Administration)
Global Microbiome Therapeutics Market Dynamics
To learn more about this report, Download Free Sample
Market Drivers
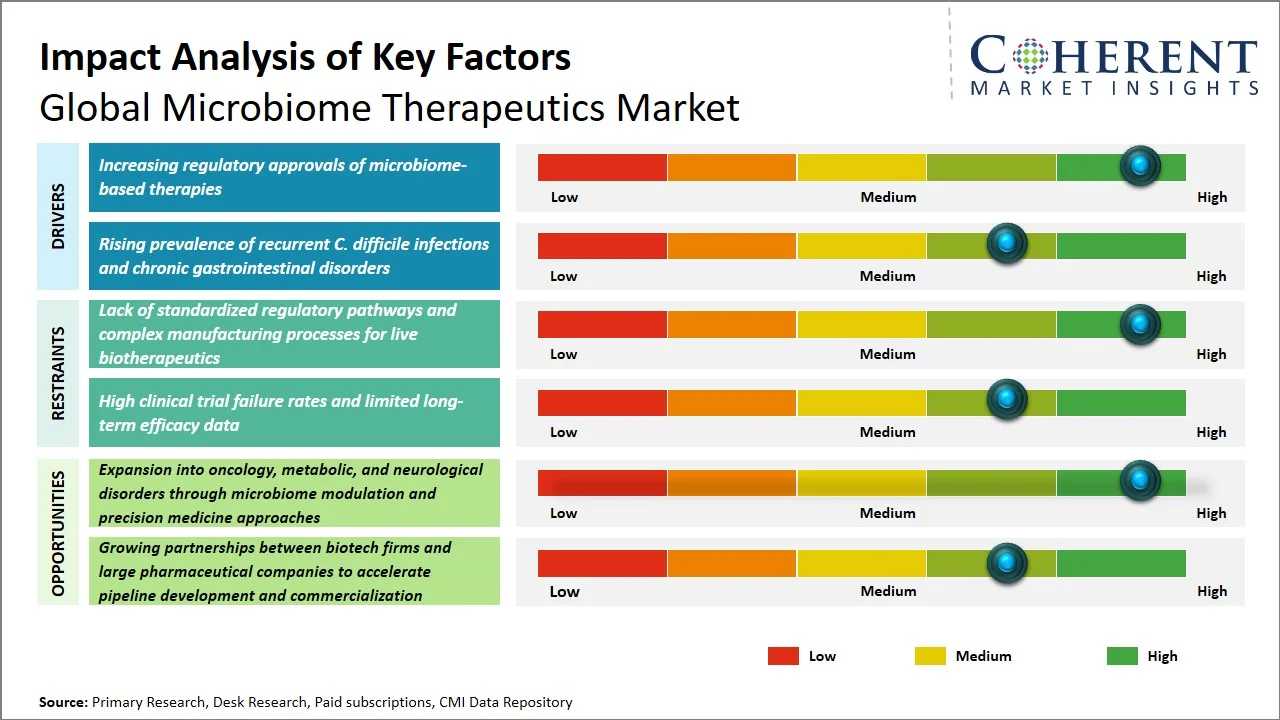
- Increasing regulatory approvals of microbiome-based therapies: S. FDA approved the world's first oral microbiome drug, Vowst, which is a significant breakthrough in the microbiome therapeutics market space (Source: U.S. Food and Drug Administration). The regulatory approvals have helped increase investment in research and development activities, thereby contributing to the market growth. In addition, it has improved investor confidence and spurred biopharmaceutical firms to develop their pipelines, resulting in an increase in clinical studies and product development initiatives. Thus, regulatory backing has become instrumental in speeding up the process of commercialization and innovation in the microbiome-based therapeutics industry.
- Rising prevalence of recurrent C. difficile infections and chronic gastrointestinal disorders: As reported by the Centers for Disease Control and Prevention, C. diff is estimated to cause almost half a million infections in the U.S. each year. (Source: Centers for Disease Control and Prevention) Moreover, the prevalence rate is estimated to be more than 117.2 out of every 100,000 people in 2023, emphasizing the continued challenge of the disease. (Source: Centers for Disease Control and Prevention) As the clinical burden grows, it hastens the use of microbiome-based therapies, which help restore the balance in the gut and provide a better alternative to conventional antibiotics.
Emerging Trends
- Shift Toward Precision Microbiome Therapies: The use of sequencing technologies and microbiome analysis has made it possible to design customized therapy programs based on the specific microbiomes of individuals. This approach has made therapy administration more effective, particularly for conditions such as cancer and metabolic diseases.
- Rising Focus on Live Biotherapeutic Products (LBPs): The transition from conventional probiotics to regulated Live Biotherapeutic Products (LBPs) is gaining momentum, propelled by accumulating clinical data and regulatory endorsement from entities like the U.S. FDA. Consequently, this shift is hastening the development of microbiome-based therapeutics, which are being engineered for specific medical uses.
Regional Insights
To learn more about this report, Download Free Sample
Why is North America a Strong Market for Microbiome Therapeutics?
North America leads the global microbiome therapeutics market, accounting for an estimated 40.5% share in 2026, owing to the high prevalence of antimicrobial resistance, and strong clinical research ecosystem. According to Centers for Disease Control and Prevention, antimicrobial resistance causes more than 2.8 million infections and around 35,000 deaths each year in the U.S. alone, making the development of an alternative treatment modality, including those based on microbiomes. (Source: Centers for Disease Control and Prevention)
Moreover, research on microbiomes are also greatly facilitated by the National Institutes of Health through projects such as the Human Microbiome Project which has generated comprehensive datasets and insights into microbial roles in human health, accelerating microbiome-based therapeutic development. (Source: National Institutes of Health) The presence of both significant medical demand and government-supported research programs helps establish the region’s dominance within the market.
Why Does the Asia Pacific Microbiome Therapeutics Market Exhibit High Growth?
The Asia Pacific microbiome therapeutics market is expected to exhibit the fastest growth with an estimated contribution of 22.5% share to the global market in 2026, because of the high rate of prevalence in microbiome-associated diseases, including infections like Clostridioides Difficile infection, gastrointestinal disorders, and metabolic diseases. Due to the increasing prevalence of health issues such as antibiotic resistance, the cases of IBD in countries such as China, Japan, and South Korea are quite high.
Also, there has been an inclination toward the clinical development and commercialization of microbiome solutions in the Asia Pacific region. This is supported by the increasing number of biotechnology innovations and clinical studies that have been carried out to develop solutions regarding microbiomes; China has established a robust microbiome R&D ecosystem. In addition, Japan has made it easier and faster for products involving microbiomes to gain regulatory approvals through Pharmaceuticals and Medical Devices Act (PMD Act) and Act on the Safety of Regenerative Medicine (ASRM) regulations.
Global Microbiome Therapeutics Market Outlook for Key Countries
Why are the U.S. Leading Innovation and Adoption in the Microbiome Therapeutics Market?
Innovation and adoption in the market for microbiome therapeutics are spearheaded by the U.S. because of the presence of robust clinical research infrastructure and the high number of clinical trials underway. According to information provided on ClinicalTrials.gov, there is a substantial number of clinical research trials involving microbiomes globally being conducted in the U.S., indicating that there is active research into multiple disease indications (Source: ClinicalTrials.gov).
Moreover, the Centers for Disease Control and Prevention is sounding the alarm about the rising tide of infections resistant to standard treatments. This situation is fueling interest in new approaches, including those that leverage the microbiome. The clear connection between ongoing clinical research and the pressing need for new solutions are, in turn, speeding up the development and acceptance of microbiome-based therapies across the U.S.
Is U.K a Favorable Market for Microbiome Therapeutics Market?
The U.K. presents a propitious environment for microbiome therapeutics, attributable to its robust regulatory framework, sophisticated research infrastructure, and increasing emphasis on novel therapeutic approaches. The Medicines and Healthcare products Regulatory Agency (MHRA) offers well-defined pathways for biologics and advanced therapies, thereby streamlining clinical development and approval processes.
Furthermore, the National Health Service (NHS) promotes the integration of innovative treatments through its healthcare delivery systems and associated funding mechanisms. The U.K. also benefits from substantial government-supported research endeavors (such as U.K. Research and Innovation (UKRI), National Institute for Health and Care Research (NIHR), and Innovate UK), with entities like U.K. Research and Innovation providing financial backing for microbiome and life sciences research. Consequently, this confluence of regulatory transparency, a strong public healthcare system, and active research and development support establishes the U.K. as a favorable and expanding market for microbiome therapeutics.
Is China Emerging as a Key Growth Hub for the Microbiome Therapeutics Market?
China appears to be a major growth hub for the microbiome therapeutics market propelled by strong government support, a vibrant biotech landscape, and a rising tide of health challenges. For instance, in 2023, China ranked first globally in most-cited research papers across multiple life science domains, including synthetic biology (61%), genomic sequencing and analysis (42%), novel antibiotics and antivirals (30%), and biological manufacturing (29%) highlighting its strong research output and innovation capacity that directly supports advancements in microbiome therapeutics. (Source: Mercator Institute for China Studies)
The China government, through the National Health Commission, has made biotechnology and precision medicine a priority, spurring innovation in microbiome research. Furthermore, China's clinical research landscape is expanding rapidly, with a noticeable increase in microbiome-focused studies and trials. The country's vast patient population and the rising incidence of chronic and metabolic diseases are also boosting the demand for new therapies. This blend of policy support, research advancements, and unmet clinical needs positions China as a promising market for microbiome therapeutics.
Why Does Germany Top the Europe Microbiome Therapeutics Market?
Germany dominates the Europe microbiome therapeutics market owing to its robust biotech environment and advanced healthcare sector. The country enjoys strict regulation by virtue of the existence of the Federal Institute for Drugs and Medical Devices, thereby helping to advance the research and development of advanced therapies. In addition, the country enjoys strong financial backing for research and development activities and participates actively in clinical studies of microbiomes. Moreover, the presence of major pharmaceutical and biotechnology firms (such as Bayer AG, BioNTech SE, Merck KGaA, and Evotec SE) helps speed up advancements.
Is the Microbiome Therapeutics Market Developing in France?
France is slowly but surely becoming a strong player in the world of microbiome therapeutics, buoyed by a surge in government-funded research and national programs. The "France 2030" initiative, which zeroes in on microbiome research, is a prime example, channeling roughly USD 68.4 million (€58 million) over seven years to fuel innovation by digging deeper into the links between the microbiome and health. (Source: France 2030) Furthermore, ambitious projects like the French Gut initiative are underway, aiming to gather data from 100,000 people and, in the process, bolster clinical research. (Source: Microbiome Foundation) Though not yet at the level of Germany and the U.S., the increasing development of research facilities is gradually establishing France as a rising market.
Regulatory Landscape Governing the Global Microbiome Therapeutics Market
|
Region |
Key Regulations |
Regulatory Overview |
Impact on Market |
|
North America |
U.S. FDA – Live Biotherapeutic Products (LBP) Guidelines; IND/BLA pathways |
Microbiome therapeutics are regulated as biologics, requiring IND approvals, clinical trials, and BLA submissions. FDA provides specific guidance for LBPs, including donor screening and manufacturing controls. |
High regulatory rigor ensures safety and efficacy, but increases development timelines and costs, favoring established players. |
|
Europe |
European Medicines Agency (EMA); Advanced Therapy Medicinal Products (ATMP) framework |
Regulated under biological medicinal products and ATMPs, requiring centralized approval. Strict guidelines on quality, safety, and traceability are enforced. |
Stringent regulations enhance product credibility, but create entry barriers and delay commercialization. |
|
Asia Pacific |
PMDA (Japan), NMPA (China), CDSCO (India) |
Regulatory frameworks are evolving, with increasing alignment toward biologics and cell/gene therapy pathways. Japan supports accelerated approvals under regenerative medicine laws. |
Faster approvals in some countries (e.g., Japan) drive innovation, while regulatory variability creates market entry challenges. |
|
Latin America |
ANVISA (Brazil), COFEPRIS (Mexico) |
Regulations are developing, often referencing U.S./EU frameworks. Approval pathways for microbiome therapies remain less defined. |
Moderate regulatory barriers, but lack of clarity may slow adoption and investment. |
|
Middle East & Africa |
SFDA (Saudi Arabia), SAHPRA (South Africa) |
Regulatory systems are emerging, largely based on international standards (FDA/EMA). Limited microbiome-specific guidelines. |
Growing market potential, but regulatory uncertainty and infrastructure gaps limit rapid growth. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the expansion into oncology, metabolic, and neurological disorders through microbiome modulation and precision medicine approaches creating new growth opportunities in the global microbiome therapeutics market?
The use of microbiome therapeutics to treat cancer, metabolic issues, and neurological disorders offers considerable growth opportunities, as it allows for personalized healthcare, taking into account the specific microorganisms linked to each condition. According to the World Health Organization, approximately 20 million new cancer cases and 9.7 million cancer-related deaths were reported globally in 2022. Additionally, the estimated number of people who were alive within 5 years following a cancer diagnosis was 53.5 million, suggesting the need for novel solutions. (Source: World Health Organization)
Furthermore, the studies supported by the National Institutes of Health, published in July 2023 have shown that the microbiome plays a crucial role in shaping immune responses, metabolic pathways, and neural communication systems (gut-brain axis) associated with oncological, metabolic, and neurological disorders (Source: National Institutes of Health). The increasing importance of the microbiome in health care settings would lead to innovations in providing targeted and personalized treatment services, making the microbiome a valuable therapeutic field.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Download Free Sample
Key Developments
- In February 2026, Metagen Therapeutics closed a Series B funding round, netting roughly USD 23.8 million (JPY 3.5 billion). This brought the company's total capital to more than USD 36.7 million (JPY 5.4 billion), a move designed to speed up global clinical trials for gut microbiome therapeutics developed in Japan. The new funds will help propel the development of microbiome-based drugs and broaden the company's clinical-stage pipeline.
- In February 2026, Evogene Ltd. and Lishan Biotech forged a licensing deal concerning BMC128 (LS-LBP-002), a cancer therapeutic candidate rooted in microbiome research. This partnership is designed to boost anti-tumor immune responses and expedite the advancement of microbiome therapeutics within the field of oncology.
Competitive Landscape
The microbiome therapeutics market is currently marked by significant competition, encompassing both established global pharmaceutical entities and nascent biotech companies, all propelled by a growing emphasis on the development of specialized therapies. This competitive environment is largely defined by technical proficiency, adherence to regulatory standards, and the pursuit of cost-effectiveness, mirroring the intricate and dynamic characteristics inherent in microbiome-based therapeutic approaches. Key focus areas include:
- Pipeline expansion and clinical trial advancement
- Strategic collaborations and partnerships
- Regulatory compliance and quality standards
- Development of advanced microbiome platforms and targeted therapies
Market Report Scope
Microbiome Therapeutics Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 57.4 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 27.3% | 2033 Value Projection: | USD 310.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Seres Therapeutics, Inc., Vedanta Biosciences, Inc., Finch Therapeutics Group, Inc., Enterome SA, 4D Pharma plc, Locus Biosciences, Inc., Rebiotix Inc., Second Genome Therapeutics, Microbiotica Ltd., and Synlogic, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The future of microbiome-based therapies will be significantly influenced by new evidence supporting live biotherapeutic products (LBPs), changes in regulatory guidelines, and the amount of investment in this area. With continued trends in pharmaceuticals towards precision medicine and biologicals, microbiome therapeutics will transition from experimental to commercialized treatment solutions. Also, as there is an emerging trend of outsourcing and collaboration in drug development, biotech companies and other development service providers will play an important role within the industry.
- Market opportunities will be significant for live biotherapeutics in gastrointestinal diseases (GI), especially Clostridioides difficile infection (CDI), and their use in oncology and immunology. For the U.S., growth is expected to be high due to the favorable regulatory landscape and the presence of supportive regulations, along with early approval of new products. For China, growth is expected to be high due to the increased investments in microbiome research and development of clinical programs.
- For gaining an advantage over their competitors, it is imperative that players in this market concentrate on developing their clinical proof by conducting late-stage studies, working on scalable production of living biotherapeutics, and creating a collaboration with major pharmaceutical corporations and scientific institutes. Moreover, getting acquainted with changing regulatory trends and entering emerging markets would be important.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Rebyota
- Vowst
- Biomictra
- Late Phase Products
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Oral
- Rectal
- Care Setting Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Specialty Pharmacies
- Physicians Clinics
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Seres Therapeutics, Inc.
- Vedanta Biosciences, Inc.
- Finch Therapeutics Group, Inc.
- Enterome SA
- 4D Pharma plc
- Locus Biosciences, Inc.
- Rebiotix Inc.
- Second Genome Therapeutics
- Microbiotica Ltd.
- Synlogic, Inc.
Sources
Primary Research Interviews
- Microbiome therapeutics developers and biotech firms
- Pharmaceutical companies and CDMOs involved in live biotherapeutics and microbiome-based drug development
- Clinical researchers and microbiome scientists specializing in gastrointestinal, metabolic, and immune-related diseases
- Regulatory experts focusing on biologics and live biotherapeutic product (LBP) approvals
Stakeholders
- Providers of microbiome therapeutics and live biotherapeutic products
- End-use Sectors
- Hospitals
- Specialty clinics (Gastroenterology, Oncology, Immunology)
- Research institutes and academic centers
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), National Institutes of Health (NIH), World Health Organization (WHO)
Databases
- U.S. FDA – Drug approvals and biologics database (Orange Book / biologics approvals)
- WHO – Global Health Observatory (GHO)
- NIH – Clinical trials and microbiome research database
- OECD Health Statistics – Biotechnology and pharmaceutical R&D data
Magazines
- Nature Reviews / Nature Portfolio – Microbiome regulatory frameworks and innovation trends
- BioPharma Dive – Microbiome drug development and funding trends
- Pharmaceutical Technology – Advancements in live biotherapeutics and microbiome manufacturing
Journals
- Journal of Pharmaceutical Sciences – Microbiome drug formulation studies
- International Journal of Pharmaceutics – Drug delivery systems for microbiome therapies
- Nature Biotechnology / Nature Microbiology – Microbiome innovation and translational research
- BMJ (British Medical Journal) – Clinical applications of microbiome-based therapies
Newspapers
- The Wall Street Journal – Biotech investments and microbiome startups
- Financial Times – Global microbiome and biotech market trends
- The Economic Times – India biotech and microbiome ecosystem developments
- Business Standard – Pharmaceutical and microbiome innovation coverage
Associations
- Microbiome Therapeutics Innovation Group (MTIG)
- Biotechnology Innovation Organization
- International Federation of Pharmaceutical Manufacturers & Associations
- Parenteral Drug Association
Public Domain Sources
- WHO – Microbiome and infectious disease frameworks
- NIH – Human microbiome research initiatives
- FDA – Live biotherapeutic product regulatory guidelines
- EMA – Microbiome-based medicinal product regulations
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients