Nigeria Pharmaceutical Market Size and Forecast – 2026-2033
The Nigeria pharmaceutical market is expected to grow from USD 3,342.7 Mn in 2026 to USD 5,824.4 Mn by 2033, registering a compound annual growth rate (CAGR) of 9.5%. The Nigeria pharmaceutical market is poised for significant expansion, fueled by the increasing burden of non-communicable diseases such as cardiovascular and chronic respiratory diseases, diabetes and cancer, that cause ~30% of deaths in the country, resulting in higher demand for medicines.
(Source: Development and Cooperation)
Key Takeaways of the Nigeria Pharmaceutical Market
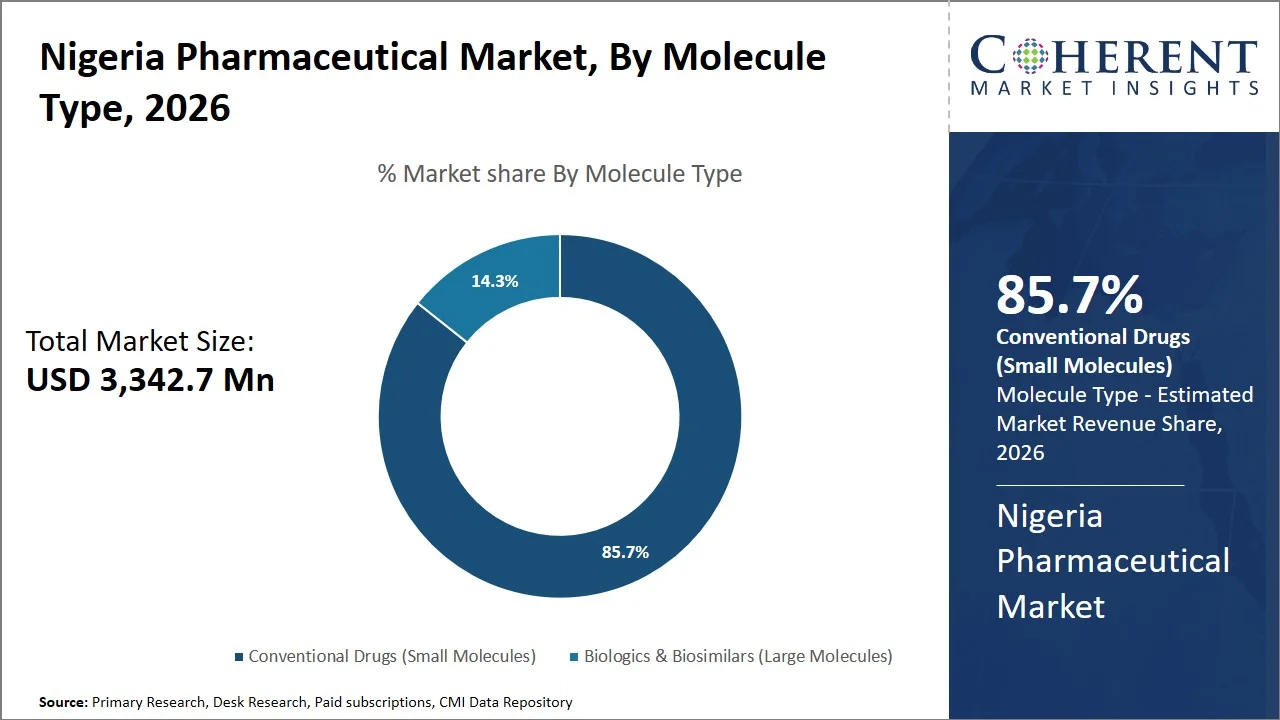
- Conventional drugs (small molecules) are projected to hold 85.7% of the Nigeria pharmaceutical market share in 2026, making it the dominant molecule type segment, owing to their compatibility with local manufacturing capabilities, lower production complexity compared to biologics, and suitability for treating high-burden conditions such as malaria, hypertension, and diabetes. For instance, a May 2025 report by World Health Organization stated Nigeria accounts for 27% of the global malaria burden, with 31% of global malaria deaths. (Source: World Health Organization)
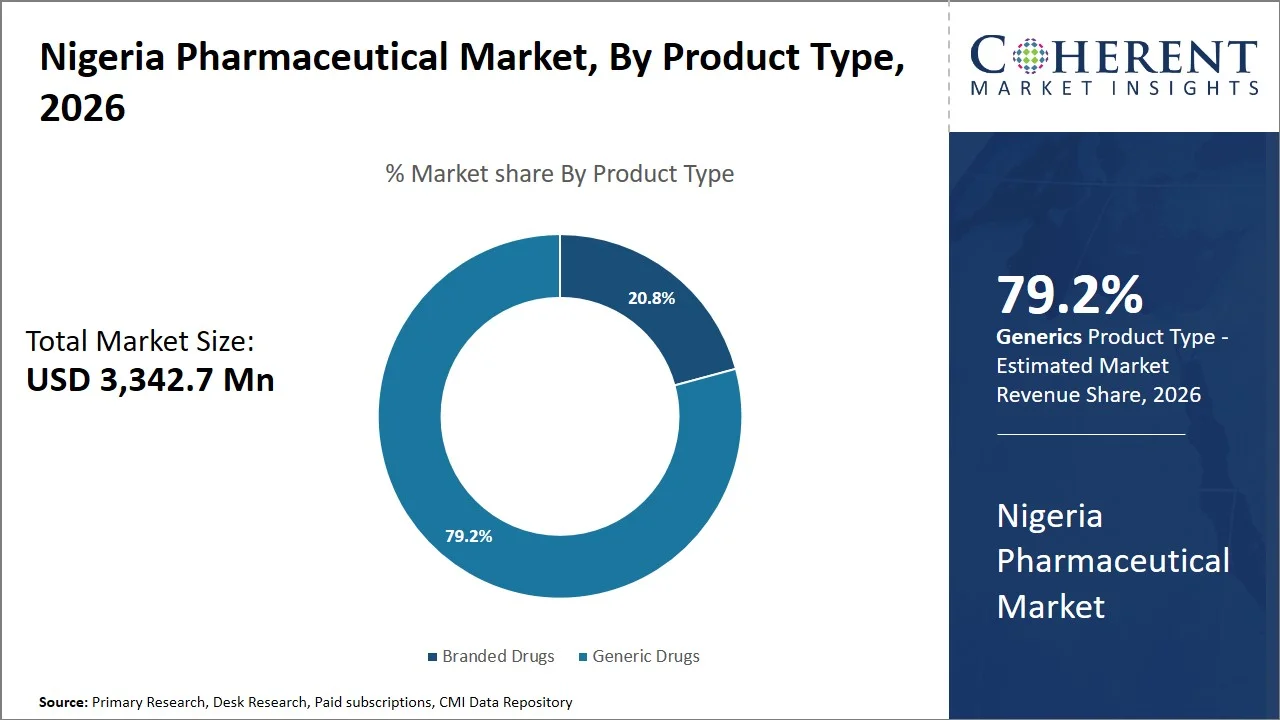
- Generic drugs are projected to hold 79.2% of the Nigeria pharmaceutical market share in 2026, making it the dominant product type segment driven by the institutional procurement practices favoring unbranded formulations (e.g., antimalarials, antibiotics, antiretrovirals- supplied through public health programs and donor-funded initiatives such as National Malaria Elimination Programme (NMEP)), rapid market entry post-patent expiry, and strong penetration across both urban pharmacies and rural drug outlets.
- Over-the-Counter (OTC) drugs are projected to hold 69.4% of the Nigeria pharmaceutical market share in 2026, making it the dominant drug type segment, due to widespread reliance on patent medicine vendors (such as community-based medicine retailers commonly operating under the regulation of the Pharmacists Council of Nigeria), limited physician access in semi-urban regions, and frequent demand for first-line treatment of malaria, pain, and minor infections without prescription dependency.
- Expansion of Regulatory Harmonization Across West Africa (ECOWAS Integration): With Nigeria’s integration into regional regulatory systems based on the ECOWAS Regional Pharmaceutical Plan, pharmaceutical firms now have an opportunity to gain access to wider markets in West Africa via one single approval process. This eliminates repetitive registration efforts and helps local producers expand their exports from Nigeria, thereby increasing competitiveness within the region.
- Growing Demand for Cold Chain and Specialty Drug Distribution Infrastructure: The increasing use of vaccines, biologics, and drugs that need to be kept cold is creating a growing need for cold chain The success of immunization campaigns conducted by bodies like the National Primary Health Care Development Agency is creating niche investment opportunities for the cold chain logistics industry in the country.
Why Do Conventional Drugs (Small Molecules) Dominate the Nigeria Pharmaceutical Market?
Conventional drugs (small molecules) are projected to hold a market share of 85.7% in 2026, owing to their relatively low cost, extensive generic presence, and compatibility with disease profiles of the nation. The healthcare sector of Nigeria operates in an out-of-pocket model, thus necessitating affordability of medication. In April 2025, study published by National Library of Medicine stated that Nigeria is significantly dependent on imports with around 70% of medication being imported, comprising both active pharmaceutical ingredients and final medications (Source: National Library of Medicine).
Furthermore, data from Trade Economics indicates that Nigeria spent around USD 642.98 million in pharmaceutical products in 2024. Thus, Nigeria relies heavily on imported, predominantly small molecule drugs. (Source: Trade Economics)
Why are Generic Drugs Most Preferred Product Type in Nigeria Pharmaceutical Market?
To learn more about this report, Request Free Sample
Generic drugs are projected to hold 79.2% of the market share in 2026, primarily because of their relative affordability, accessibility, and perfect representation of the requirements of the country’s medicine access program. The out-of-pocket health expenses in Nigeria are very high (~70–75% of total healthcare spending is out-of-pocket), which increases the necessity for affordable drugs, including generics.
According to the World Health Organization, essential medicines include those that address the basic health needs of the population, and they are formulated to be cheaper and more accessible, while being presented in the form of generics most of the time (Source: World Health Organization). Additionally, from the global perspective, the adoption of procurement policy within developing nations clearly indicates a preference for generics as part of improving access to essential medicines. (Source: National Library of Medicine)
Over-the-Counter (OTC) Drugs Segment Dominates the Nigeria Pharmaceutical Market
The Over-the-Counter (OTC) drugs segment is expected to hold 69.4% of the Nigeria pharmaceutical market share in 2026, primarily because of the easy availability of the drugs and lack of strict regulations for purchasing them, allowing customers to buy and receive treatment without a prescription.
This segment is favored by a well-developed retail pharmacy chain and patent medicine sellers, who provide the first point of contact for health care in Nigeria. According to World Health Organization, self-care measures, such as the consumption of OTC medicines, are vital in boosting health care access in underdeveloped and developing nations.
(Source: World Health Organization)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory & Policy Shift: Push for Local Drug Manufacturing (2023–2025) |
|
|
Industry Shift: NAFDAC “5+5” Policy Driving Local Manufacturing Transition (2023–2025) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: United Nations Industrial Development Organization, National Agency for Food and Drug Administration and Control)
Nigeria Pharmaceutical Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
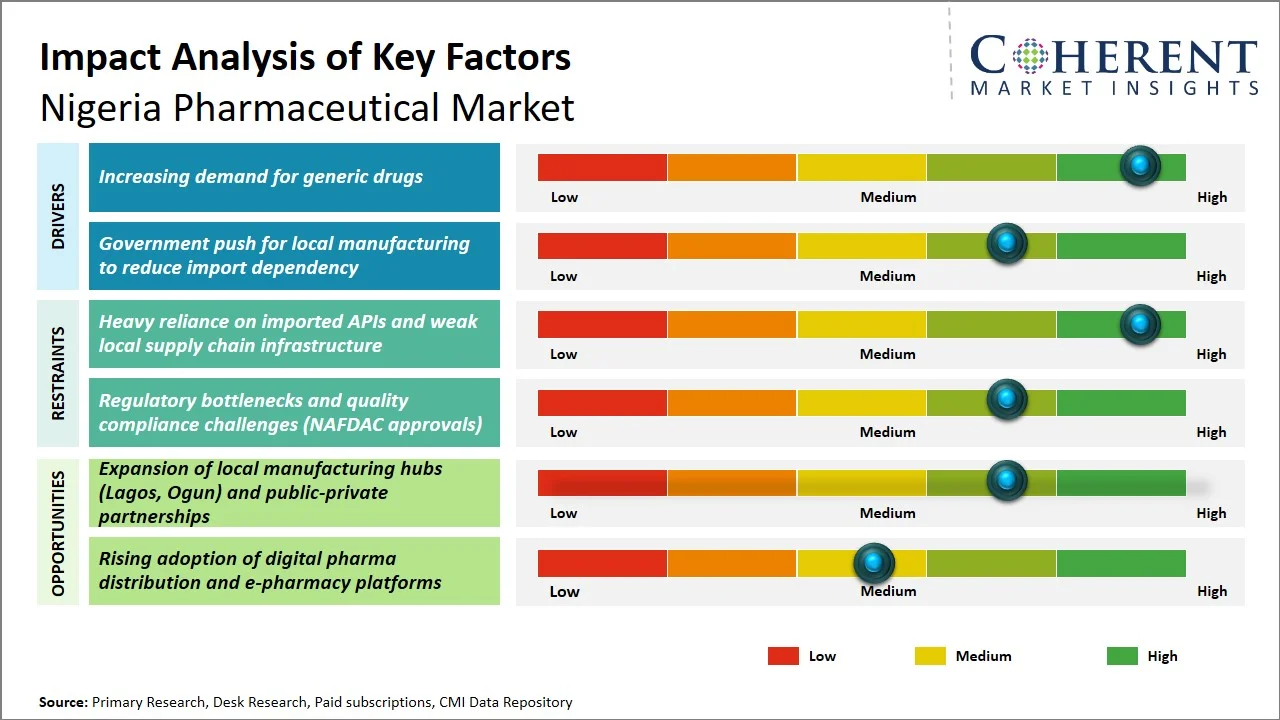
- Increasing Demand for Generic Drugs: Generics constitute a considerable percentage of the pharmaceutical industry in Nigeria due to their high popularity among patients looking for cheaper alternatives to save money within the health care industry. This is achieved with the support of the Federal Ministry of Health Nigeria in its National Drug Policy, where the use of generics is encouraged for health improvement at reduced costs (Source: Federal Ministry of Health Nigeria). According to the data presented by Pharma West Africa, generics constitute a considerable percentage of about 38.9% of the Nigeria pharmaceutical industry due to their high popularity among patients. (Source: Pharma West Africa)
- Government Push for Local Manufacturing to Reduce Import Dependency: The policies developed by the government of Nigeria have been focusing on encouraging local pharmaceutical manufacturing with an aim of minimizing imports and becoming self-reliant in the industry. As per the Federal Ministry of Health Nigeria, dependency on imports is one of the major challenges in this regard, and the National Drug Policy has continually been focusing on the importance of encouraging local production and backward integration (Source: Federal Ministry of Health Nigeria). Furthermore, some initiatives such as Presidential Initiative for Unlocking the Healthcare Value Chain have been implemented to enhance local manufacturing processes, ensure efficient supply chain management, and minimize foreign exchange issues. (Source: PVAC)
Emerging Trends
- Shift Toward Local Manufacturing & Import Substitution: Nigeria's pharmaceutical landscape is undergoing a notable transformation, with a marked increase in domestic production. This shift is fueled by government backing and the effects of foreign exchange controls on imported medications. The National Agency for Food and Drug Administration and Control, along with industry associations, are backing initiatives that boost capacity, thereby strengthening the competitive position of local manufacturers in the generic drugs.
- Growth of Private Healthcare & Digital Distribution Channels: The proliferation of private hospitals, retail pharmacy chains, and new e-pharmacy platforms is making drugs more accessible and improving last-mile delivery. This is especially evident in cities, where the demand for convenience, quicker access, and branded therapies is growing, driven by greater healthcare awareness.
Regulatory Framework Governing the Nigeria Pharmaceutical Market
|
Regulatory Body |
Description |
Key Role in Pharmaceutical Market |
|
National Agency for Food and Drug Administration and Control (NAFDAC) |
Primary regulatory authority responsible for drug approval, registration, and quality control |
Ensures safety, efficacy, and quality of pharmaceuticals before market entry; oversees importation and local manufacturing |
|
Pharmacists Council of Nigeria (PCN) |
Governing body for pharmacy practice and premises licensing |
Regulates pharmacists, licensing of pharmacies, and distribution channels |
|
Federal Ministry of Health Nigeria |
Develops national health policies and pharmaceutical guidelines |
Provides strategic direction, policy implementation, and public health programs |
|
National Health Insurance Authority (NHIA) |
Oversees health insurance schemes and reimbursement frameworks |
Expands access to medicines through insurance coverage and reimbursement policies |
|
National Drug Policy Nigeria |
Framework guiding drug development, distribution, and rational use |
Promotes local manufacturing, access to essential medicines, and regulatory harmonization |
|
Counterfeit and Fake Drugs Act |
Legislation targeting counterfeit and substandard drugs |
Strengthens enforcement against fake drugs and improves market integrity |
|
Standards Organization of Nigeria |
Sets standards for pharmaceutical packaging and labeling |
Ensures compliance with quality and safety standards in drug production and packaging |
|
ECOWAS Regional Pharmaceutical Plan |
Regional initiative to harmonize drug regulations across West Africa |
Facilitates cross-border trade and regulatory alignment within the region |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the expansion of local manufacturing hubs (Lagos, Ogun) and public-private partnerships creating new growth opportunities in the Nigeria pharmaceutical market?
The establishment of pharmaceutical manufacturing industries in Nigeria, particularly in Lagos and Ogun, has been facilitated significantly by government initiatives (e.g. National Drug Policy, Central Bank of Nigeria’s intervention funds for healthcare manufacturing through the Healthcare Sector Intervention Facility (HSIF)) and collaboration with private firms. According to National Agency for Food and Drug Administration and Control (NAFDAC) statistics, there are around 115 licensed pharmaceutical manufacturers, and many of them are concentrated in industry hubs such as Lagos and Ogun, which serve as the manufacturing hub of the country. (Source: National Agency for Food and Drug Administration and Control (NAFDAC))
The program within the framework of the National Drug Policy seeks to ensure that 70% or more of drug production is carried out locally, helped by the use of incentives such as restricted imports of specific drugs. In addition to this, there have been collaborations between government agencies and private producers, through groups such as PMG-MAN, helping expand capacity building, where Nigeria produces ~60% of West African pharmaceutical production. For instance, the Healthcare Sector Intervention Facility (HSIF) was launched by the Central Bank of Nigeria in March 2020, allocating USD 280 Million (₦100 billion) towards financing the local pharmaceutical manufacturing sector through partnership with industry associations like the Pharmaceutical Manufacturers Group of Manufacturers Association of Nigeria. (Source: Central Bank of Nigeria)
Market Players, Key Development, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- In November 2025, the European Union and Nigeria joined forces to strengthen local manufacturing within the health sector. The goal of this collaboration is to increase Nigeria's capacity to produce pharmaceuticals domestically, thereby lessening its dependence on imported medicines. This effort is anticipated to fortify the resilience of supply chains and foster sustained growth within Nigeria's pharmaceutical market.
- In November 2025, Nigeria and Brazil formalized a partnership, a memorandum of understanding, aimed at bolstering Nigeria's local pharmaceutical manufacturing. The core of the agreement centers on sharing technology and building capacity, with the goal of lessening Nigeria's dependence on imported medications. This collaborative effort is anticipated to boost domestic production and foster sustained expansion within Nigeria's pharmaceutical sector over the long haul.
- In October 2024, Micro Labs Ltd. acquired Swipha, a move designed to solidify its foothold in Nigeria and boost its local manufacturing. This purchase underscores the rising tide of foreign investment flowing into Nigeria's pharmaceutical industry, while also fostering the expansion of drug production within the country. The acquisition is anticipated to streamline supply chains and make medicines more accessible and affordable.
Competitive Landscape
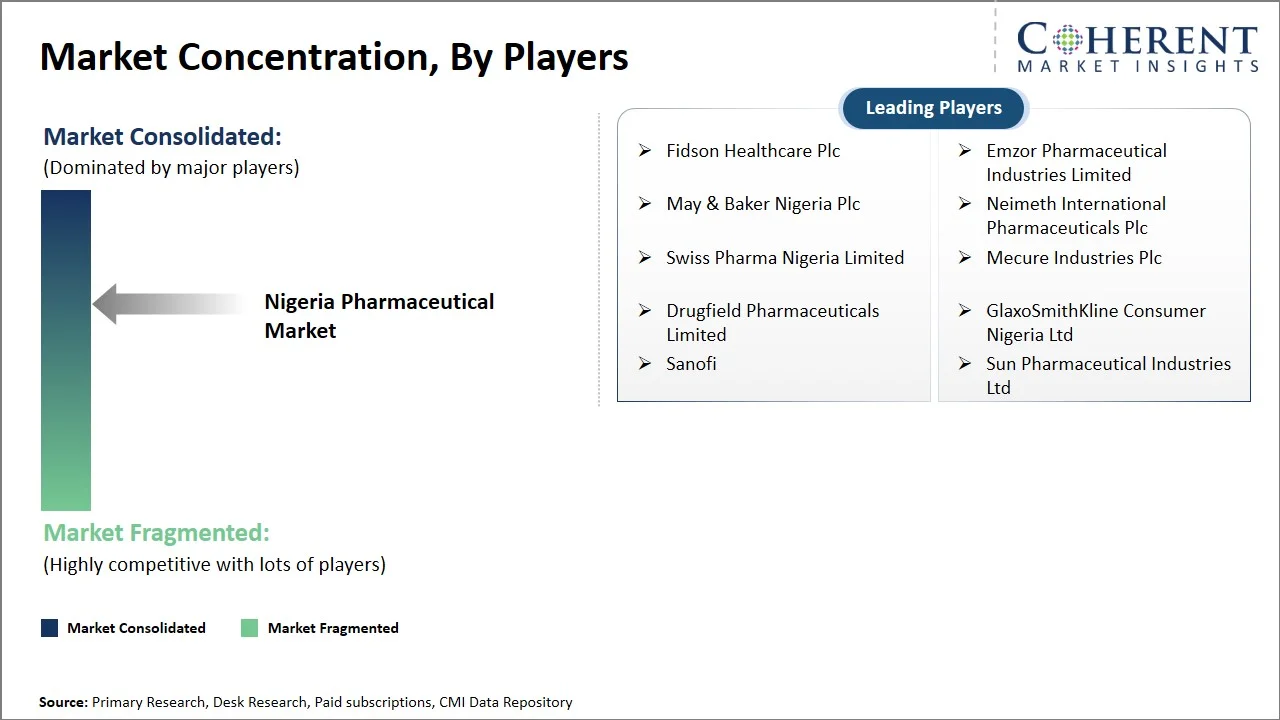
Nigeria pharmaceutical market operates in a state of moderate competition. It's a landscape where robust local manufacturers coexist with multinational corporations. The competitive dynamic hinges on several factors: pricing, how readily products are available, and the extent of distribution networks. Domestic companies, bolstered by regulatory support from the National Agency for Food and Drug Administration and Control, as well as industry associations like the Pharmaceutical Manufacturers Group of Manufacturers Association of Nigeria, hold a strong position in the generics sector. Meanwhile, multinational firms continue to have a presence, particularly in the branded and specialty drug segments. Key focus areas include:
- High-Volume Generic Drug Production (Anti-infectives & Chronic Therapies)
- Expansion of Local Manufacturing & Import Substitution
- Strengthening Distribution and Last-Mile Access
Market Report Scope
Nigeria Pharmaceutical Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3,342.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.5% | 2033 Value Projection: | USD 5,824.4 Mn |
| Segments covered: |
|
||
| Companies covered: |
Fidson Healthcare Plc, Emzor Pharmaceutical Industries Limited, May & Baker Nigeria Plc, Neimeth International Pharmaceuticals Plc, Swiss Pharma Nigeria Limited, Mecure Industries Plc, Drugfield Pharmaceuticals Limited, GlaxoSmithKline Consumer Nigeria Ltd, Sanofi, and Sun Pharmaceutical Industries Ltd |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- The new phase for the Nigeria pharmaceutical industry relies on localization, affordability, and favorable regulations to reduce its dependence on imports and develop an autonomous manufacturing base. High volume generic drugs production, better supply chain management, and incorporation of advanced treatments are key features of the future market. However, the use of small molecules will prevail because of their lower prices. On the other hand, selective use of biologics and vaccines in urban settings and private hospitals can be expected, creating two different markets in terms of mass and premium segments.
- The most favorable opportunity for the market lies in the area of small molecule generic drugs focusing on anti-infectives and chronic disease treatment drugs (diabetes, cardiovascular), and the market can easily be expanded to the West Africa sub-region. The need for the drugs is due to rising disease burden, population growth, and cost models that depend on out-of-pocket payments. Opportunities are seen in the area of vaccines due to collaboration with international organizations like World Health Organization (WHO) and United Nations Children's Fund (UNICEF).
- For an advantageous position in the market, the firms need to consider expanding their manufacturing capacity locally, adopt cost-leadership strategy and build strong distribution channels while working in tandem with industry associations like the Pharmaceutical Manufacturers Group of Manufacturers Association of Nigeria. Some of the areas that should form part of these firms’ strategies include backward integration, flexibility in dealing with regulations, technology transfers, and utilization of the digital route for reaching last mile consumers.
Market Segmentation
- Molecule Type Insights (Revenue, USD Mn, 2021 - 2033)
- Conventional Drugs (Small Molecules)
- Biologics & Biosimilars (Large Molecules)
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Branded Drugs
- Generic Drugs
- Drug Type Insights (Revenue, USD Mn, 2021 - 2033)
- Prescription Drugs
- Over-the-Counter (OTC) Drugs
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Oral
- Parenteral
- Topical
- Inhalation
- Others
- Formulation Insights (Revenue, USD Mn, 2021 - 2033)
- Tablets
- Capsules
- Injectables
- Suspensions
- Powders
- Sprays
- Others
- Age Group Insights (Revenue, USD Mn, 2021 - 2033)
- Adults
- Pediatrics
- Geriatric
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals
- Clinics
- Others
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Key Players Insights
- Fidson Healthcare Plc
- Emzor Pharmaceutical Industries Limited
- May & Baker Nigeria Plc
- Neimeth International Pharmaceuticals Plc
- Swiss Pharma Nigeria Limited
- Mecure Industries Plc
- Drugfield Pharmaceuticals Limited
- GlaxoSmithKline Consumer Nigeria Ltd
- Sanofi
- Sun Pharmaceutical Industries Ltd
Sources
Primary Research Interviews
- Managing Directors / Commercial Heads – Local pharmaceutical manufacturers
- Regulatory Affairs Heads – Companies dealing with National Agency for Food and Drug Administration and Control (NAFDAC) approvals
- Procurement Heads – Hospital networks & large pharmacies
- Distributors / Importers – Drug distribution companies across Lagos, Abuja
- Representatives – Pharmaceutical Manufacturers Group of Manufacturers Association of Nigeria (PMG-MAN)
- Pharmacists & Retail Chain Operators – Urban & semi-urban regions
Databases
- UN Comtrade
- World Health Organization (Global Health Observatory)
- National Bureau of Statistics Nigeria
Magazines
- BusinessDay Nigeria
- The Guardian Nigeria
- Premium Times Nigeria
- ThisDay Nigeria
- Pharma West Africa Magazine
- African Pharmaceutical Review
- Africa Health Magazine
- WHO Bulletin
- Fierce Pharma
- Pharmaceutical Technology
Journals
- The Lancet – Africa healthcare access studies
- BMJ Global Health – Drug affordability & access
- Journal of Pharmaceutical Policy and Practice – Nigeria pharma policy
Newspapers
- BusinessDay Nigeria
- The Guardian Nigeria
- Premium Times Nigeria
Associations
- Pharmaceutical Society of Nigeria
- Nigerian Medical Association
- West African Postgraduate College of Pharmacists
Public Domain Sources
- National Agency for Food and Drug Administration and Control
- Federal Ministry of Health Nigeria
- Central Bank of Nigeria
- World Bank
- United Nations Children's Fund
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients