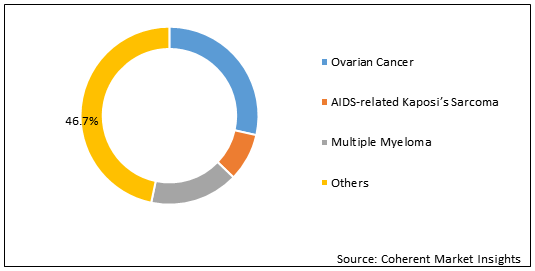
Liposomal doxorubicin is a chemotherapy drug, which is administered intravenously to treat AIDS-related Kaposi's sarcoma, breast cancer, ovarian cancer, and other solid tumors. Liposome drug delivery systems are able to modify pharmacokinetics and bio-distribution of cytostatic agents, it increases the concentration of the drug released to neoplastic tissue and reduces the exposure of normal tissue with the drug. This drug is a type of anthracycline antitumor antibiotic, also known as Doxil and doxorubicin hydrochloride liposome. Dosage of doxorubicin hydrochloride liposome depends on several factors including, height and weight, general health or other health problems, and the type of cancer such as ovarian cancer, AIDS-related kaposi's sarcoma, and multiple myeloma.
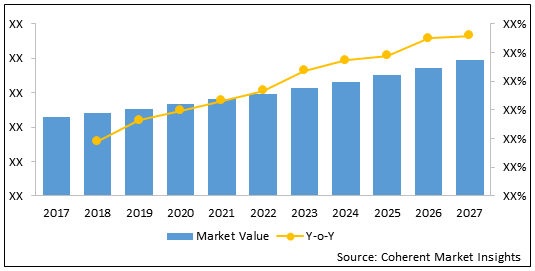
The U.S. liposomal doxorubicin market is estimated to be valued at US$ 140.0 Million in 2020 and is expected to reach US$ 192.9 Million by 2027, exhibiting a CAGR of 4.7% over the forecast period (2020-2027).
Figure 1. U.S. Liposomal Doxorubicin Market Value (US$ Million) Analysis and Forecast and Y-o-Y Growth (%), 2020
To learn more about this report, Request Free Sample
The increasing approval of generic products is expected to drive the U.S. liposomal doxorubicin market growth
The increasing approval of generic products of liposomal doxorubicin from regulatory authorities is expected to boost demand for liposomal doxorubicin and this is further expected to drive growth of the U.S. liposomal doxorubicin market. For instance, in March 2017, Dr. Reddy’s Laboratories Ltd announced that it received the U.S. Food Drug and Administration approval to launch doxorubicin hydrochloride liposome injection in the U.S., which is a generic version of Janssen Products’ Doxil (doxorubicin hydrochloride liposome injection). Doxorubicin hydrochloride liposome injection is indicated for the treatment of ovarian cancer and administered intravenously in the patients.
U.S. Liposomal Doxorubicin Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2019 | Market Size in 2020: | US$ 140.0 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2020 to 2027 |
| Forecast Period 2020 to 2027 CAGR: | 4.7% | 2027 Value Projection: | US$ 192.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Johnson & Johnson, Baxter International Inc., Sun Pharmaceutical Industries Ltd., and Dr. Reddy’s Laboratories, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Figure 2. U.S. Liposomal Doxorubicin Market Share (%), By Cancer Type 2020
To learn more about this report, Request Free Sample
U.S. Liposomal Doxorubicin Market– Impact of Coronavirus (COVID-19) Pandemic
Since the COVID-19 virus outbreak in December 2019, the disease has spread to over 100 countries across the globe and the World Health Organization declared it a public health emergency. The private healthcare sector is one of the sectors, which has been majorly impacted by the pandemic. The lockdowns in various countries have created an economic burden on the private healthcare sector. Healthcare providers are facing challenges with regards to manpower, equipment, consumables, and other resources to ensure safety of patients with other diseases, and declining outpatients’ visits, among others. Moreover, the pandemic has negatively impacted the development, production, and supply of drugs, and affected growth of the liposomal doxorubicin market and healthcare segment of various companies across the globe. This has led to closure of industrial establishments, except manufacturing of essential commodities and disruption in supply chain of products. Thus, the Covid-19 pandemic is expected to slightly disrupt the R&D activities of healthcare sector as well as in the liposomal doxorubicin market.
U.S. Liposomal Doxorubicin Market: Restraint
Some side effects associated with liposomal doxorubicin drug is expected to hamper the growth of market. The side effects include increased risk of infection, breathlessness and pale complexion, bruising, bleeding gums and nosebleeds, tiredness and weakness (fatigue) during and after treatment, redness, soreness, and peeling of the hands and feet, loss of weight, and diarrhea among others.
Key Players
Major players operating in the U.S. liposomal doxorubicin market include Johnson & Johnson, Baxter International Inc., Sun Pharmaceutical Industries Ltd., and Dr. Reddy’s Laboratories, Inc.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients