Global Nipah Virus Infection Market Size and Forecast: 2026-2033
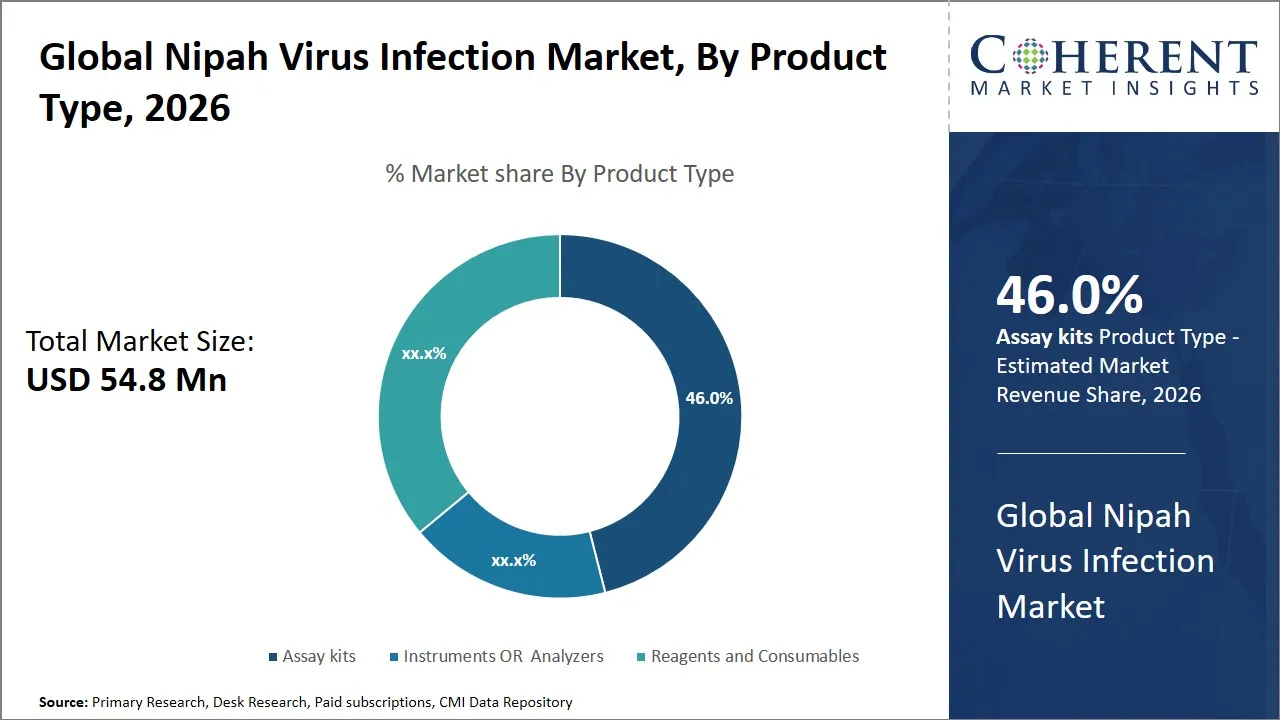
The global Nipah virus infection market is estimated to be valued at USD 54.8 Mn in 2026 and is expected to reach USD 123.6 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 12.32% from 2026 to 2033. The market growth can be attributed to the rising awareness of Nipah virus infection, recurring nature of the disease, and increased investments in the development of diagnostics, vaccines, and therapeutic treatments for Nipah virus infections.
The overall burden of human infections and mortality has a significant impact on the market size for Nipah virus infections. For instance, as per a study published by International Society for Infectious Diseases in 2024, the overall human infection count worldwide has surpassed 750, with over 400 deaths.
Key Takeaways of the Global Nipah Virus Infection Market
- The assay kits segment is expected to dominate the global Nipah virus infection market by product type, accounting for approximately 46.0% of the total market share in 2026.
- The molecular tests segment is expected to dominate the global Nipah virus infection market by test type, accounting for approximately 68.0% of the total market share in 2026.
- The PCR-based testing segment is expected to dominate the global Nipah virus infection market by technology platform, accounting for approximately 72.0% of the total market share in 2026.
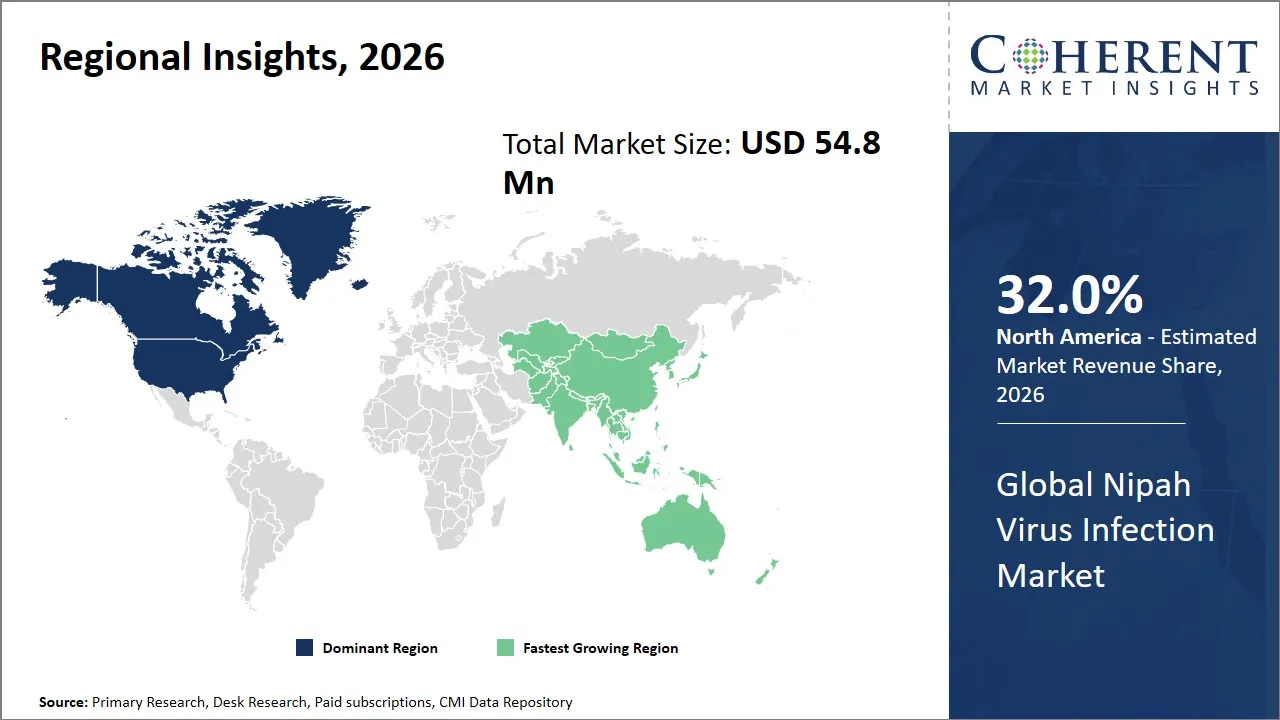
- North America is expected to dominate the global Nipah virus infection market in 2026, accounting for approximately 32.0% of the total market share.
- Asia Pacific is anticipated to witness the fastest growth, holding a market share of 28.0% in 2026.
Market Overview
- Nipah virus is a zoonotic virus, and it belongs to the Henipavirus family. This virus is transmitted from animals to humans, and it leads to respiratory and brain infections with high mortality.
- The Nipah virus infection market growth is primarily driven by diagnostic test, with molecular methods such as RT-PCR widely used for accurate disease detection.
- Testing carried out in the Nipah virus infection market is majorly done in high-containment labs like BSL-3 and BSL-4 labs with the help of public health organizations.
- The Nipah virus infection market is primarily driven by public health initiatives, with demand largely dependent on surveillance and monitoring activities.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
WHO prioritization of Nipah virus under R&D Blueprint for epidemic-prone diseases |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Assay Kits Segment Dominate the Global Nipah Virus Infection Market in 2026?
The assay kits segment is anticipated to hold a market share of 46.0% in 2026, because of the growing need for rapid and precise diagnostic tests. These kits are particularly valuable for pinpointing the Nipah virus, leveraging proven biochemical and immunological methods. This approach guarantees high sensitivity and specificity, which are crucial for distinguishing Nipah virus infections from other illnesses with overlapping symptoms.
The popularity of assay kits can be further supported by their ease of use, the potential for large-scale production, and technological advancements. Assay kits are designed to be easy to use, regardless of whether they are to be used in a sophisticated lab or a point-of-care facility, and they do not require a great deal of sample preparation and expertise, allowing them to be implemented even in areas with scarce resources and a high risk of outbreak.
For instance, a study published in October 2023 and indexed by the National Library of Medicine, identified real-time polymerase chain reaction (RT-PCR) as the most important and sensitive method for diagnosing early Nipah virus infections. This technology is already in widespread use, via RT-PCR-based assay kits. These kits are purpose-built for swiftly and accurately identifying the virus. The study's findings demonstrated the kits' exceptional sensitivity and specificity, enabling the detection of the virus even at extremely low concentrations, with results available within hours. This rapid turnaround and high degree of precision have established them as the favored choice among healthcare professionals and public health authorities, thereby solidifying their preeminence in the marketplace.
(Source: National Library of Medicine)
Why Does the Molecular Tests Segment Dominate the Global Nipah Virus Infection Market in 2026?
The molecular tests segment is expected to dominate with 68.0% of the global Nipah virus infection market share in 2026, attributed to the high precision offered by the tests in detecting the viral genome. The high sensitivity offered by molecular tests helps in the early detection of the virus, even in low viral loads, and hence is very important in the early stage of the infection, before the appearance of severe symptoms.
Moreover, the importance of molecular tests are also related to the role they play in the genomic analysis of the virus, using next-generation sequencing (NGS) techniques, as the emergence of infectious diseases, such as Nipah virus infection, is an important factor in the global market. Additionally, the use of molecular diagnostic tests can be further enhanced by the integration of these tests into automated and high-throughput diagnostic platforms.
For example, there has been a tremendous expansion in global genomic surveillance activities following the COVID-19 pandemic. According to the World Health Organization, global efforts currently are focused on ensuring that all 194 member states have access to genomic sequencing technologies by 2032, in recognition of the rapid scale-up of sequencing infrastructure globally.
(Source: World Health Organization)
Furthermore, as compared to other methods such as serology and antigen-based tests, molecular diagnostic tests are more accurate and reliable, thus reinforcing the use of these tests as a gold standard in diagnosis owing to the increased need for rapid, accurate, and efficient diagnostic tools in the management and control of infectious diseases.
The PCR-Based Testing Segment Dominates the Global Nipah Virus Infection Market
The PCR-based testing segment is projected to hold a market share of 72.0% in 2026, attributed to the fact that this testing method has been identified as the gold standard in the detection of viral pathogens. The use of the technology of PCR has been critical in the detection of viruses because it has helped in the amplification of the viruses and thus enabled detection even when the concentration of the viruses is extremely low, which becomes critical in the early detection and effective management of the disease of Nipah virus infection.
Moreover, the specificity of the technology ensures that the chances of cross-reactivity with other viruses are low, making it the most preferred technology in the detection of the disease of Nipah virus infection. The capability of the technology in adapting itself to the newer strains of the virus has proved to be crucial in the effective detection of the viruses despite the genetic differences.
The advantage of PCR-based testing is further highlighted by the advancements in technology in real-time quantitative PCR and multiplex PCR, which enables the simultaneous testing of several targets and determination of viral loads. For instance, a January 2026 study, indexed by the National Library of Medicine, showed that multiplex PCR systems using microfluidic chips could provide results in less than two hours. These systems boasted a sensitivity and specificity of roughly 95–98%, representing a substantial leap in diagnostic speed and accuracy.
(Source: National Library of Medicine)
These capabilities go beyond disease detection to disease monitoring and epidemiological analysis. In addition, increased global investment in PCR technology, accompanied by innovations in user-friendly technology, has extended its reach beyond traditional laboratory settings to remote and resource-poor areas. Its high accuracy, scalability, and accessibility have continued to make PCR technology the leading technology platform for Nipah virus infection.
Pipeline & Key Company Landscape – Nipah Virus Infection Market
|
Company / Organization |
Product / Candidate |
Type |
Mechanism / Platform |
Current Status |
Funding / Collaboration |
Market Relevance |
|
University of Oxford |
ChAdOx1 Nipah Vaccine |
Vaccine |
Viral vector (ChAdOx1 platform) |
Phase I clinical trials ongoing/recently initiated |
Supported by Coalition for Epidemic Preparedness Innovations (CEPI) |
Among the most advanced vaccine candidates; key for outbreak prevention |
|
National Institutes of Health (NIH) |
m102.4 monoclonal antibody |
Biologic |
Neutralizing antibody targeting Nipah virus |
Early clinical / compassionate use studies |
Government-funded research |
Leading therapeutic candidate for post-exposure intervention |
|
Coalition for Epidemic Preparedness Innovations (CEPI) |
Funded Nipah Vaccine Candidates (Portfolio Programs) |
Vaccine |
Viral vector, mRNA, and subunit platforms |
Preclinical to early clinical stages |
Global partnerships (multi-country programs) |
Central driver of global pipeline and funding ecosystem |
|
Indian Council of Medical Research (ICMR) |
RT-PCR diagnostic capability |
Diagnostics |
Molecular (PCR-based detection) |
Actively deployed during outbreaks |
Government of India |
Critical for confirmed diagnosis and outbreak control |
|
University of Queensland |
Molecular clamp vaccine research |
Vaccine |
Protein subunit (molecular clamp technology) |
Preclinical / early-stage research |
CEPI-supported programs |
Expands diversity of vaccine platforms under development |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Regional Insights
To learn more about this report, Request Free Sample
North America Nipah Virus Infection Market Analysis and Trends
The North America region is projected to lead the market with a 32.0% share in 2026, attributed to the high standard of healthcare infrastructure, the high allocation of funds to emerging infectious diseases, and the supportive regulatory environment to enable the swift development and approval of new products. Additionally, the U.S. government’s initiatives to address the biodefense and emerging infectious diseases, such as those by the National Institutes of Health (NIH) and the Biomedical Advanced Research and Development Authority (BARDA), speed up the development process.
Furthermore, the region is home to pharmaceutical and biotechnology firms like GSK, Moderna, and Regeneron Pharmaceuticals. These companies leverage cutting-edge technologies, including mRNA platforms and monoclonal antibodies, to combat new infectious diseases, including Nipah virus.
For instance, in July 2022, Moderna revealed that it had begun dosing the initial participant in a Phase 1 clinical trial for their Nipah virus mRNA vaccine candidate, mRNA-1215. This project, a collaboration with the U.S. National Institutes of Health, underscores the potential of cutting-edge mRNA technology to address new and evolving infectious diseases.
(Source: Moderna)
Moreover, the existence of trade agreements in the region facilitates the trade of research materials. For instance, the existence of the United States-Mexico-Canada Agreement (USMCA) facilitates the easy exchange of information in the protection of intellectual property in the region. Therefore, the existence of the agreement facilitates the easy exchange of biomedical research materials and even technology in the region. This helps in the promotion of collaboration in the region between pharmaceutical companies and health centers. This collaboration helps in the promotion of the speedy production of diagnostics, vaccines, and even treatments for infectious diseases.
(Source: Office of the United States Trade Representative)
Asia Pacific Nipah Virus Infection Market Analysis and Trends
Asia Pacific is anticipated to witness the fastest growth, holding a market share of 28.0% in 2026, primarily driven by ongoing outbreaks in India and Bangladesh. This is evident from the repeated outbreaks in the Indian state of Kerala in the years 2018, 2021, and 2023. Government efforts are further fueling the market for Nipah virus infection.
For example, the Indian Council of Medical Research (ICMR) and the Department of Biotechnology (DBT) have increased their investment in research related to Nipah infections. This is seen through the latest government initiatives like ICMR’s initiative on seeking industry partnerships for the development of monoclonal antibodies and vaccines, as well as the availability of high-end BSL-3 and BSL-4 facilities through institutes like the National Institute of Virology for conducting preclinical and clinical work.
(Source: Indian Council of Medical Research (ICMR))
Furthermore, the presence of local firms like Bharat Biotech, Panacea Biotec, and the Serum Institute of India adds considerable weight to the region’s capabilities. These entities are all deeply engaged in developing vaccines and therapies for Nipah virus infection. For example, partnerships facilitated by the Coalition for Epidemic Preparedness Innovations (CEPI) have helped regional players participate in global Nipah vaccine development initiatives. Furthermore, cooperation between India and Bangladesh in surveillance and containment has helped in the effective control of the disease, thus cementing Asia Pacific’s position in the market.
(Source: Coalition for Epidemic Preparedness Innovations (CEPI))
Nipah Virus Infection Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Nipah Virus Infection Market?
The U.S. leads in the R&D of the Nipah virus due to its high investment in biodefense and emerging infectious diseases. For instance, Federal biodefense spending is estimated to be around USD 27 billion in FY2026, with cumulative investments of almost USD 50 billion in biodefense programs, aided by organizations like the National Institutes of Health and the Centers for Disease Control and Prevention (CDC).
(Source: The Council on Strategic Risks)
Furthermore, the country has renowned biotech companies such as Moderna and Regeneron, which focus on the development of new and innovative technologies in therapy. The U.S. has provided the Nipah virus R&D with the right conditions to hasten the clinical trials and thus become an advanced R&D center.
How is India Helping in the Growth of the Nipah Virus Infection Market?
The Nipah virus infection market in India is greatly affected by the history of the country concerning Nipah virus infections (with approximately 99 total cases reported in India since 2001–2023, of which around 28% were from Kerala, and about 21 deaths recorded in the state). Government agencies have focused their attention on developing and establishing surveillance systems for fighting infections caused by the Nipah virus. This has led to research collaborations between academia and industry, thereby establishing India as a significant player in fighting Nipah virus infections in the region.
For instance, the government of Kerala, in association with the Ministry of Health and Family Welfare, has undertaken rapid response activities during the recent outbreak of Nipah virus infection, which included large-scale contact tracing operations and the utilization of mobile diagnostic labs. Following the recent outbreak, a mobile BSL-3 lab has been instrumental in tracing and testing over 190 contacts.
(Source: UN Office for the Coordination of Humanitarian Affairs (OCHA))
Key Drivers for the Growth of the Australia Nipah Virus Infection Market
The Nipah virus infection market in Australia has been affected by the country’s proximity to endemic areas in South Asia and Southeast Asia, leading to heightened investments in biosecurity preparedness, surveillance systems, and advanced diagnostic capabilities to enable early detection and rapid response to potential imported cases. This has made the country take a proactive approach to surveillance and preparedness. The country has also invested in the infrastructure and research partnership in countries such as India. For instance, Australia committed USD 410 million (AUD 620.47 million) under its “Partnerships for a Healthy Region” program for 2023–2027 to strengthen health systems and collaborative research across the Indo-Pacific, including India, and an additional USD 2.5 million (AUD 3.8 million) was allocated in 2024 under the Australia–India Strategic Research Fund to support joint scientific projects, including infectious disease research.
Further, companies such as CSL Limited have been able to participate in collaborative projects, thus enhancing the country’s contribution to the fight against Nipah virus infection.
Singapore Nipah Virus Infection Market Trends
Singapore has a comparative advantage in the Nipah virus infection market owing to its biomedical hub with government support in the fight against infectious diseases. This focus on public-private partnerships ensures the presence of various international firms and research centers (such as Duke-NUS Medical School, A*STAR, National Centre for Infectious Diseases, as well as multinational companies like Pfizer and GlaxoSmithKline) carrying out Nipah virus research and developing diagnostics for the virus. For instance, researchers at Duke-NUS Medical School have led studies on Nipah virus transmission mechanisms and antibody-based therapeutic targets, contributing to the development of potential antiviral strategies and diagnostic approaches.
Further, Singapore's streamlined regulatory framework promises swift product approvals. This efficiency will, in turn, foster innovation in drug development. Consequently, the nation is poised to become a significant force in combating infectious diseases throughout the region.
Regulatory Framework, Compliance Requirements, and Guidelines Governing the Nipah Virus Infection Market
|
Organization |
Regulation / Guideline |
Scope |
Market Impact |
|
World Health Organization (WHO) |
R&D Blueprint for Priority Pathogens |
Identifies Nipah virus as a priority disease for research and outbreak preparedness |
Drives global funding, accelerates diagnostic and vaccine development |
|
World Health Organization (WHO) |
Laboratory Biosafety Guidelines |
Defines BSL-3/BSL-4 requirements for handling high-risk pathogens |
Limits testing to specialized labs, increasing demand for high-containment infrastructure |
|
U.S. Food and Drug Administration (FDA) |
Emergency Use Authorization (EUA) Pathways |
Enables rapid approval of diagnostics during outbreaks |
Supports faster commercialization of diagnostic kits in emergency scenarios |
|
European Medicines Agency (EMA) |
PRIME (Priority Medicines) Scheme |
Facilitates accelerated assessment for unmet medical needs |
Encourages development of vaccines and therapeutics for rare infectious diseases |
|
Ministry of Health and Family Welfare (MoHFW) |
National Guidelines for Nipah Virus Management |
Outbreak response, testing protocols, and containment strategies |
Drives regional demand for diagnostics and strengthens surveillance systems |
|
Indian Council of Medical Research (ICMR) |
Diagnostic Testing Guidelines (RT-PCR protocols) |
Standardizes laboratory testing and confirmation methods |
Ensures dominance of molecular diagnostics in the market |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In December 2025, the University of Oxford initiated first Phase II clinical trial globally for its ChAdOx1 NipahB vaccine. This trial was being conducted by the university's Pandemic Sciences Institute. The vaccine's primary goal is to evaluate its safety and the immune responses it generates in regions prone to repeated outbreaks. This represents a major advancement in the development of a vaccine for the Nipah virus. This will improve the clinical pipeline and, in turn, expand the market beyond diagnostics.
- In November 2025, Indian Council of Medical Research invited pharmaceutical and biotechnology companies for collaboration in the development and manufacturing of monoclonal antibodies for the Nipah virus. This has come on the heels of the successful preclinical (animal) tests of indigenous antibody candidates. This is a positive sign of the pharmaceutical industry’s participation in the fight against Nipah virus infection.
- In October 2025, Serum Institute of India, in collaboration with the University of Oxford and CEPI, announced the establishment of the world’s largest reserve of the investigational Nipah virus vaccine. The program's work has included creating as many as 100,000 doses of the ChAdOx1 NipahB vaccine, which are intended for use in Phase II clinical trials. This development represents a significant step forward in the fight against the Nipah virus, suggesting a growing commitment within the industry to develop vaccines for this disease.
Top Strategies Followed by Global Nipah Virus Infection Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established market leaders focus on rigorous R&D to create advanced diagnostics tools, vaccines, and therapeutics. Furthermore, large-scale clinical trials focus on expanding the global footprint through strategic partnerships with governments, OEMs, and healthcare institutions utilizing existing supply chains for global distribution. |
In March 2024, Johnson & Johnson announced expansion of its infectious disease R&D pipeline through collaborations targeting emerging viral threats, strengthening vaccine platform capabilities for epidemic preparedness. |
|
Mid-Level Players |
Mid-level players focus on cost-effective and scalable solutions to serve the markets in the developing world. They encourage collaboration with contract manufacturers and technology suppliers to improve efficiency in the production process. |
In September 2023, Biological E Limited expanded manufacturing partnerships to scale up vaccine production capabilities for infectious diseases, focusing on affordable solutions for low- and middle-income countries. |
|
Small-Scale Players |
Small-scale players adopt niche strategies such as the development of innovative, portable, and user-friendly diagnostic technologies with specific focus on technologies such as microfluidics, biosensors, and AI. Furthermore, they opt for regional collaborations to penetrate the market. |
In June 2024, E25Bio advanced development of rapid diagnostic tests for infectious diseases using paper-based and point-of-care technologies aimed at improving detection in resource-limited settings. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Nipah Virus Infection Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 54.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 12.32% | 2033 Value Projection: | USD 123.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
BioPerfectus, Molbio Diagnostics, Huwel Lifesciences, BGI Genomics, ELK Biotechnology, Creative Diagnostics, RayBiotech, Abbexa, AntibodySystem, and Alpha Diagnostic Intl |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Nipah Virus Infection Market Dynamics
To learn more about this report, Request Free Sample
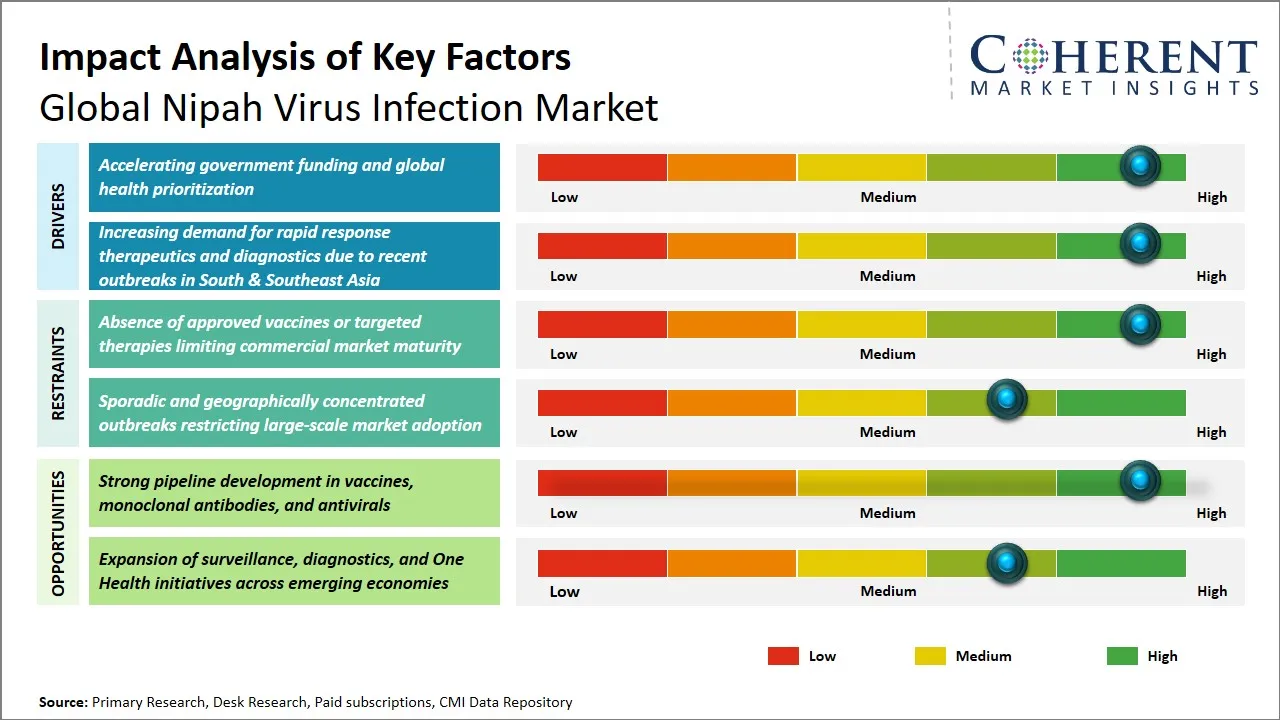
Global Nipah Virus Infection Market Driver – Accelerating Government Funding and Global Health Prioritization
The high mortality rate, which is estimated to be around 40% to 75% , has been a major factor in government funding and prioritization of the Nipah virus infection. Governments and agencies around the world have increased research to combat the severe clinical manifestations and epidemic potential of the Nipah virus. For example, the National Institutes of Health (NIH) has undertaken clinical research to investigate monoclonal antibodies to combat Nipah virus infection. This is an example of increased government investment and prioritization of the disease. At the same time, the European Medicines Agency has developed several regulatory pathways to aid the development of treatments to combat high-risk pathogens, including Nipah virus.
(Source: World Health Organization)
Moreover, the threat of Nipah virus-related outbreaks has resulted in several strategies to improve public health. One such country is Malaysia, where Nipah virus was first detected. The country has significantly improved its systems for monitoring zoonotic diseases. The country has incorporated animal and human health monitoring systems to prevent Nipah virus from re-emerging.
Furthermore, Singapore has also adopted strict biosecurity and infectious disease preparedness strategies to combat the risk of Nipah virus, as well as other emerging zoonotic diseases. As can be inferred from these strategies, it is evident that the high mortality rate and risk of Nipah virus-related outbreaks continue to influence several strategies to improve public health.
Global Nipah Virus Infection Market Opportunity - Robust Pipeline Development in Vaccines, Monoclonal Antibodies, and Antivirals
The global Nipah virus infection market presents a lucrative opportunity in view of the large pipeline of vaccines, monoclonal antibodies, and antiviral agents currently in the pipeline for the prevention and treatment of Nipah virus infection. High mortality potential and epidemic possibility of Nipah virus infection make it imperative to speed up research and development of vaccines in the biotech/pharmaceutical sector. For instance, in November 2022, the University of Queensland unveiled a Nipah Virus vaccine candidate built on a molecular clamp platform. Profectus BioSciences, following its acquisition by Emergent BioSolutions, also made strides in this area, creating Vesicular Stomatitis Virus (VSV) based vaccine platforms aimed at Nipah Virus infection. These efforts represent a diversification of vaccine technologies, encompassing approaches such as Protein Subunit and Viral Vector Technologies.
(Source: University of Queensland, Emergent BioSolutions)
Meanwhile, monoclonal antibodies and anti-viral drugs are also gaining momentum in the development of potential treatments for Nipah virus infections. For instance, the potential of monoclonal antibodies in the prevention of Nipah virus infections has been shown in preclinical studies by researchers at Rocky Mountain Laboratories. In addition, anti-viral drugs like ribavirin have also been used in outbreaks as potential candidates in the reduction of the severity of Nipah virus infections. This growing list of potential treatments for Nipah virus infections are being made possible by the increased funding and collaboration between governments, research institutions, and private players worldwide, thus providing a great platform for potential innovations in addressing the gaps in Nipah virus treatments.
Analyst Opinion (Expert Opinion)
- Since response time is a critical factor in outbreak management, there is a considerable opportunity for companies to develop point-of-care technologies for outbreak management. Companies can look to differentiate themselves on ultra-rapid technologies for point-of-care management using multiplex technology and emergency use validation.
- The high mortality rate associated with Nipah virus infections creates a considerable opportunity for vaccine and antibody development. Companies can use advanced technologies such as mRNA and viral vectors and partner with international health organizations to speed up vaccine and antibody development and approvals.
- The fact that the market is based on public health infrastructure creates considerable opportunities for partnerships in high-risk countries such as South and Southeast Asia. Companies can look to differentiate themselves in these countries by providing integrated solutions.
- There exists considerable potential for companies to integrate diagnostic technologies with digital surveillance technologies. Companies can invest in technologies such as AI for digital surveillance and data sharing in line with national disease surveillance.
Market Segmentation
- Product Type Insights (Revenue, USD Mn, 2021 - 2033)
- Assay kits
- Instruments/Analyzers
- Reagents and Consumables
- Test Type Insights (Revenue, USD Mn, 2021 - 2033)
- Molecular Tests
- Serology Tests
- Antigen-Detection Tests
- Confirmatory/Specialized Tests
- Technology Platform Insights (Revenue, USD Mn, 2021 - 2033)
- PCR-Based Testing
- ELISA
- Lateral Flow
- Next-Generation Sequencing
- Others
- Sample Type Insights (Revenue, USD Mn, 2021 - 2033)
- Blood/ Serum
- Urine
- Cerebrospinal Fluid (CSF)
- Throat Swab
- Nasal Swab
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Hospitals and Infectious Disease Centers
- Public Health Agencies
- Reference Laboratories
- Others
- Regional Insights (Revenue, USD Mn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- BioPerfectus
- Molbio Diagnostics
- Huwel Lifesciences
- BGI Genomics
- ELK Biotechnology
- Creative Diagnostics
- RayBiotech
- Abbexa
- AntibodySystem
- Alpha Diagnostic Intl
Sources
Primary Research Interviews
Industry Stakeholders
- Molecular Diagnostic Kit Manufacturers (RT-PCR, ELISA)
- Infectious Disease Diagnostic Companies
- Public Health Laboratory Experts (BSL-3/BSL-4 labs)
- Regulatory and Biosafety Authorities
- Government Procurement Agencies (outbreak response)
- Epidemiologists and Virology Researchers
End Users
- Public Health Laboratories
- Infectious Disease Hospitals
- National Disease Control Centers
- Research Institutes & Virology Labs
Government and International Databases
- World Health Organization – Nipah epidemiology, outbreak reports, R&D Blueprint
- Centers for Disease Control and Prevention – Clinical guidance and surveillance
- National Institutes of Health – Clinical and virology research
- Indian Council of Medical Research – Outbreak data and diagnostic protocols
- ClinicalTrials.gov – Pipeline and clinical trial tracking
Trade Publications
- Pharmaceutical Technology
- BioPharma Dive
- Fierce Biotech
- Genetic Engineering & Biotechnology News (GEN)
- Rapid Microbiology
Academic Journals
- The Lancet Infectious Diseases
- Journal of Clinical Virology
- Emerging Infectious Diseases (CDC)
- Viruses (MDPI)
- Frontiers in Microbiology
Reputable Newspapers
- Financial Times – Global healthcare policy, funding trends, and pharmaceutical industry insights
- The New York Times – Public health developments and global disease outbreak reporting
Industry Associations
- Coalition for Epidemic Preparedness Innovations (CEPI)
- International Society for Infectious Diseases
- American Society for Microbiology
Public Domain Resources
- NIH PubMed Central (PMC) – Open-access studies
- WHO Global Health Observatory – Disease burden
- CEPI Reports – Vaccine pipeline and funding
- Indian Council of Medical Research (ICMR) – Outbreak data, surveillance reports, and diagnostic guidelines
- United Nations Office for the Coordination of Humanitarian Affairs (UNOCHA) – Emergency response updates and outbreak situation reports
- World Organisation for Animal Health – Zoonotic disease surveillance and animal host data
- Food and Agriculture Organization – Animal-human transmission insights and risk assessments
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 10 Years
Share
Share
About Author
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients