North America Hydrocortisone Market Size and Forecast – 2026 To 2033
The North America hydrocortisone market is expected to grow from USD 82,925.8 Mn in 2026 to USD 162,787.1 Mn by 2033, registering a compound annual growth rate (CAGR) of 11.7%. The North America hydrocortisone market is poised for significant expansion owing to the increasing cases of adrenal insufficiency which need a lifetime of hormone replacement therapy.
According to the National Institutes of Health, primary adrenal insufficiency affects around 100–140 people per million, necessitating continuous hydrocortisone use as standard treatment. (Source: National Institutes of Health)
Key Takeaways of the North America Hydrocortisone Market
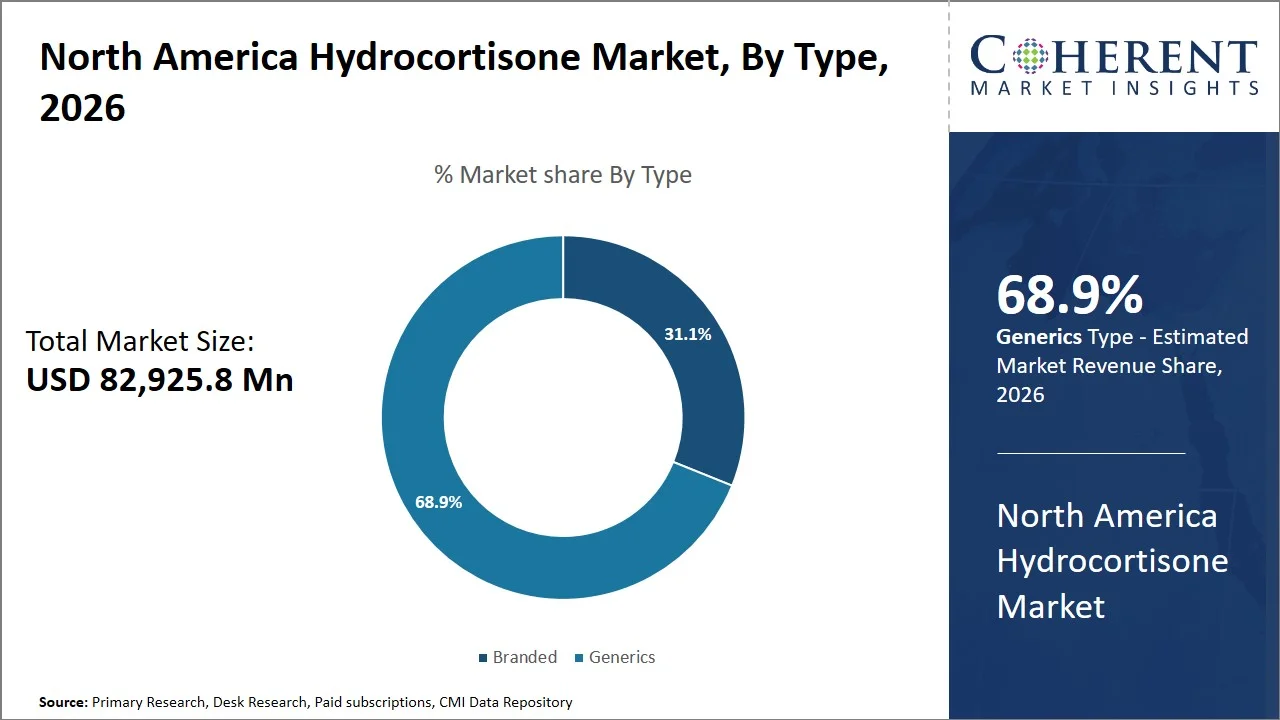
- Generics are projected to hold a market share of 68.9% of the North America hydrocortisone market share in 2026, making it the dominant type segment, owing to the high degree of price sensitivity, coupled with the impact of regulatory structures (e.g., U.S. Food and Drug Administration generic drug approval pathways, Hatch-Waxman Act, and Medicare Part D reimbursement policies), which, in turn, fosters the broad integration of unpatented hydrocortisone pharmaceuticals into reimbursement policies and purchasing procedures. Consequently, this has facilitated consistent sales expansion of generics across both retail and hospital settings.
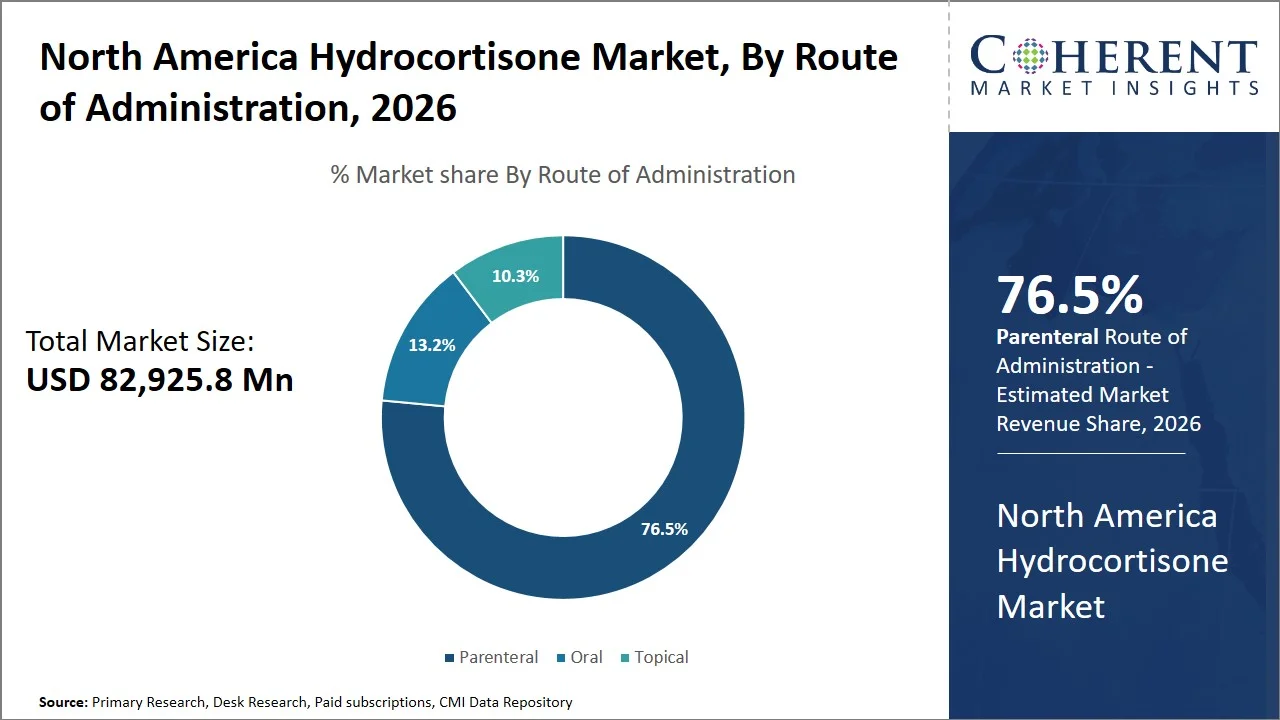
- Parenteral is projected to hold 76.5% of the North America hydrocortisone market share in 2026, making it the dominant route of administration segment. The frequent use of parenteral administration highlights the drug's importance in acute and emergency medicine, particularly in intensive care units where immediate delivery is essential for stabilizing patients through drug treatment.
- The Sepsis and septic shock segment is projected to hold 5% of the North America hydrocortisone market share in 2026, owing to the medicine's significance in critical treatment protocols, where hydrocortisone is frequently employed as an adjunctive therapy for the management of systemic inflammation.
- Expanding Use in Post-Procedural and Hospital-Based Inflammation Management: The rising application of hydrocortisone in post-operative and hospital settings is attributed to its anti-inflammatory properties, which also help in counteracting allergic reactions and adrenal insufficiency among severely sick patients. Rising surgeries and hospitalizations in North America contribute significantly to rising demand for injectable hydrocortisone.
- Rising Demand in Pediatric and Geriatric Dermatology Segments: Increasing cases of skin diseases in children and aged people are leading to increased utilization of mild steroids such as hydrocortisone. There is an increasing preference among healthcare professionals for milder steroids due to the sensitivity of the skin in these patient segments.
Why Do Generics Dominate the North America Hydrocortisone Market?
Generics are projected to hold a market share of 68.9% in 2026, bolstered by robust regulatory backing and policies favoring economical medications (such as, Hatch-Waxman Act, Medicare Part D, Medicaid Drug Rebate Program).
The Association for Accessible Medicines (AAM) indicates that generic medications account for about 90% of all prescriptions dispensed in the U.S. (Source: Association for Accessible Medicines (AAM). This statistic highlights the extensive acceptance of generics, primarily because they're more affordable and readily available. Public healthcare programs, including Medicare and Medicaid (e.g., Medicare Part D, Medicaid Drug Rebate Program, 340B Drug Pricing Program), encourage the use of generics to help control rising healthcare costs. Since hydrocortisone is a corticosteroid, whose patent has lapsed, and it's available by prescription and over-the-counter, it aligns well with these goals.
Why is Parenteral the Most Preferred Route of Administration in North America Hydrocortisone Market?
To learn more about this report, Request Free Sample
Parenteral is projected to hold 76.5% of the market share in 2026, as it is the most preferred route of administration in emergency and hospital settings. The National Library of Medicine recommends that adrenal crisis is a critical medical situation where hydrocortisone should be administered intravenously or intramuscularly immediately to provide quick effects. (Source: National Library of Medicine)
National Library of Medicine suggested that the administration of the drug should begin with an injection of 100 mg hydrocortisone and then continue over 24 hours. (Source: National Library of Medicine). Moreover, as per a study by National Library of Medicine published in August 2024, hydrocortisone is required for saving patients' lives during adrenal crisis and it can be administered in the emergency department or intensive care unit. (Source: National Library of Medicine)
Sepsis and Septic Shock Segment Dominates the North America Hydrocortisone Market
The Sepsis and septic shock segment is expected to hold 35.5% of the North America hydrocortisone market share in 2026, because of its extremely high prevalence rate and the urgent requirement for rapid treatment with corticosteroids in severe cases. As per statistics from the Centers for Disease Control and Prevention, more than 1.7 million adults get diagnosed with sepsis each year in the U.S., resulting in around 350,000 deaths, highlighting sepsis as a leading cause of death in hospitals, claiming almost one out of three deaths occurring there (Source: Centers for Disease Control and Prevention). Furthermore, the severity of the disease drives demand for hydrocortisone, as the mortality rate associated with septic shock ranges up to about 34.3%, much higher than non-severe sepsis. (Source: National Library of Medicine)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Regulatory & Industry Shift: Expansion of Value-Based Care Models by Centers for Medicare and Medicaid Services |
|
|
Industry & Policy Shift: Rapid Growth of Accountable Care Organizations (ACOs) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
(Source: Centers for Medicare & Medicaid Services, American Hospital Association)
North America Hydrocortisone Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
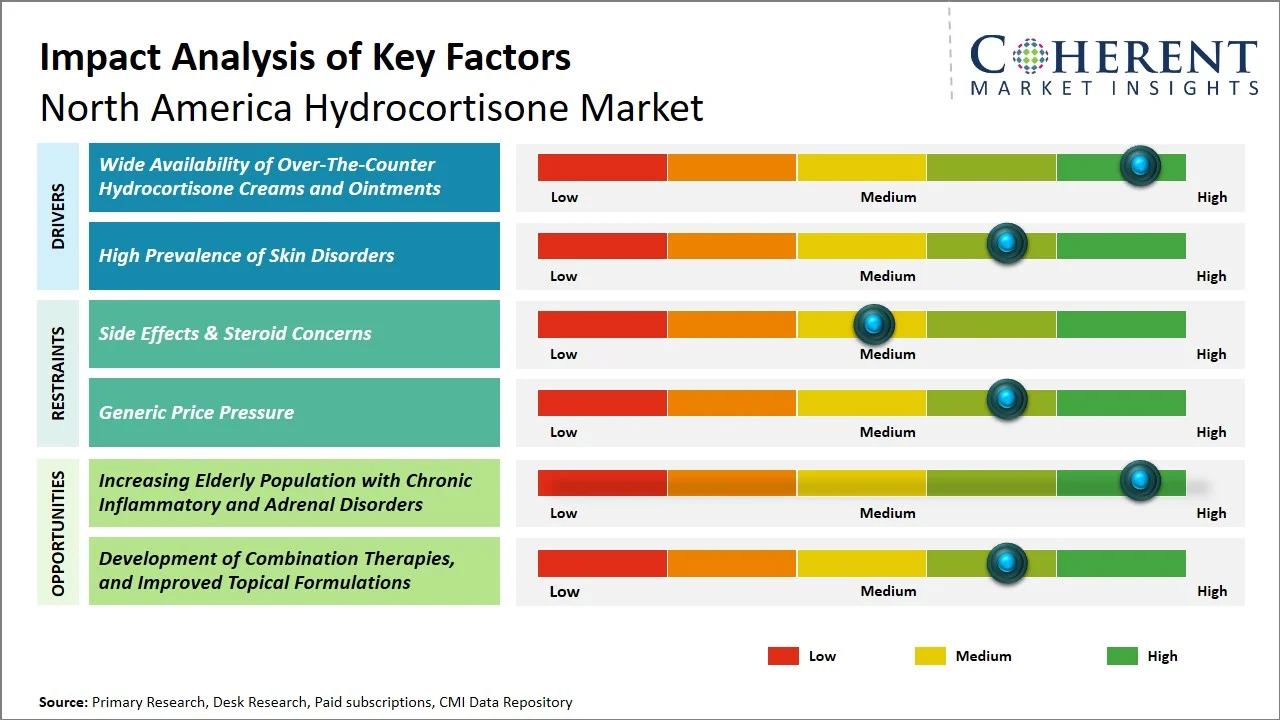
- Wide Availability of Over-The-Counter Hydrocortisone Creams and Ointments: This enables direct consumer access without prescription barriers, significantly increasing treatment adoption for mild to moderate skin conditions. In the U.S., Food and Drug Administration (FDA) permit the sale of low-strength topical corticosteroids (e.g., 0.5%–1% hydrocortisone) as OTC drugs, making them easily accessible through retail pharmacies and e-commerce channels. This accessibility aligns with rising self-care trends, where a large share of patients prefers managing minor dermatological conditions independently, thereby driving high-volume consumption and repeat purchases of hydrocortisone-based products. (Source: U.S. Food and Drug Administration)
- High Prevalence of Skin Disorders: The high and growing prevalence of skin and allergic conditions in North America is significantly expanding the addressable patient pool for hydrocortisone treatments. According to the Centers for Disease Control and Prevention (CDC), 31.7% of U.S. adults have at least one allergic condition (including eczema), with 7.7% specifically affected by eczema, while 12.7% of children are diagnosed with eczema. (Source: Centers for Disease Control and Prevention) Additionally, the National Institute of Allergy and Infectious Diseases reports that eczema affects 2–10% of adults and up to 30% of children in the U.S. (Source: National Institute of Allergy and Infectious Diseases) Conditions associated with symptoms like irritation, itching, and inflammation are usually managed with the use of hydrocortisone. The drug is among those that are usually prescribed as the first line of treatment; hence, its high demand.
Emerging Trends
- Shift Toward Low-Dose and Safer Formulations: Manufacturers are putting more emphasis on creating hydrocortisone formulations that use lower doses and target specific areas, aiming to reduce problems like skin thinning and the potential for long-term reliance. The U.S. Food and Drug Administration's regulatory framework is also pushing for safer over-the-counter options, which in turn fuels innovation in gentler, dermatologically refined products.
- Rising Adoption of Combination and Multi-Action Therapies: There's a rising interest in hydrocortisone combination products. These formulations combine antifungal, antibacterial, or moisturizing agents to improve treatment outcomes. This trend reflects a growing demand for more effective solutions to complex skin conditions, which subsequently increases patient compliance and helps products differentiate themselves in a competitive landscape.
Regulatory Framework Governing the North America Hydrocortisone Market
|
Regulatory Body |
Description |
Impact on Market |
|
U.S. Food and Drug Administration (FDA) |
Primary authority overseeing drug approval, labeling, and safety in the U.S. |
Regulates hydrocortisone formulations (Rx & OTC), ensuring safety, efficacy, and proper labeling |
|
Health Canada |
Governs pharmaceutical approvals and compliance in Canada |
Ensures product quality, dosage regulations, and market authorization for hydrocortisone products |
|
Federal Food Drug and Cosmetic Act (FD&C Act) |
Core legislation regulating drugs in the U.S. |
Establishes requirements for approval, manufacturing, and marketing of corticosteroids |
|
United States Pharmacopeia (USP) |
Sets quality and purity standards for pharmaceutical ingredients |
Defines standards for hydrocortisone API and formulations to ensure consistency and safety |
|
Good Manufacturing Practice (GMP) |
Quality assurance guidelines for drug manufacturing |
Ensures consistent production quality and compliance across manufacturers |
|
Drug Enforcement Administration (DEA) |
Regulates controlled substances in the U.S. |
Limited impact, but ensures compliance in manufacturing and distribution practices where applicable |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is the increasing elderly population with chronic inflammatory and adrenal disorders creating new growth opportunities in the North America hydrocortisone market?
The growing number of senior citizens in North America will offer substantial growth prospects for the hydrocortisone market since most elderly people suffer from chronic inflammatory and endocrine disorders that require the regular administration of corticosteroids. As per the U.S. Center for Disease Control and Prevention, the U.S. population is aging, with almost a quarter of the population expected to be 65 or older by 2060. (Source: U.S. Center for Disease Control and Prevention)
Additionally, the U.S. Center for Disease Control and Prevention report on “Older People Health” states that nearly 47.8% of adults aged 65 years and above suffer from diagnosed arthritis, an inflammatory disease that requires treatment through corticosteroids such as hydrocortisone (Source: U.S. Center for Disease Control and Prevention). Further, the National Institute on Aging further emphasizes that elderly people are more prone to developing chronic skin disorders, delayed healing of wounds, and inflammation, which makes them more dependent on corticosteroid drugs such as hydrocortisone. (Source: National Institute on Aging)
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- In December 2025, Cosette Pharmaceuticals revealed the U.S. FDA's green light for the first generic version of CIPRO HC. This version, an otic suspension combining ciprofloxacin 0.2% HCl and hydrocortisone 1%, also came with 180 days of Competitive Generic Therapy exclusivity. The introduction of this generic version is expected to make hydrocortisone-based combination therapies more affordable and easier to get, which should encourage their wider use in treating ear infections.
- In May 2025, NKHINDIVI (hydrocortisone) oral solution was granted regulatory approval by the U.S. Food and Drug Administration for Eton Pharmaceuticals. This medicine is the only commercially available liquid form of hydrocortisone indicated for children with adrenocortical insufficiency. The pre-measured 1 mg/mL dose ensures proper administration without the need for pill splitting.
- In February 2024, Eton Pharmaceuticals stated that the U.S. Patent and Trademark Office (USPTO) awarded a patent for its unique oral liquid hydrocortisone formulation. This patent ensures market exclusivity and solidifies the company's reputation in the realm of hydrocortisone medicines, especially for the pediatric patient population.
Competitive Landscape
The North America hydrocortisone market is highly competitive with established pharmaceutical companies and robust over-the-counter brand competition battling for dominance in both retail outlets and online platforms. To gain an edge, businesses are prioritizing product differentiation, exploring new formulations, and crafting consumer-focused branding strategies. Key focus areas include:
- Advanced OTC Formulations & Combination Products
- Strong Retail & E-commerce Distribution Strategies
- Regulatory Compliance & Dermatology-backed Positioning
Market Report Scope
North America Hydrocortisone Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 82,925.8 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 11.7% | 2033 Value Projection: | USD 162,787.1 Mn |
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Johnson & Johnson, Bayer AG, GlaxoSmithKline plc, Viatris Inc., Teva Pharmaceutical Industries Ltd., Sanofi S.A., Sun Pharmaceutical Industries Ltd., Hikma Pharmaceuticals PLC, and Novartis AG |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- North America hydrocortisone market is gradually transitioning to become customer-focused with factors such as accessibility, convenience, and self-medications playing critical roles in driving the market. There is going to be an increase in OTC developments, innovation in products used topically for dermatological conditions and customization for skin care products driven by awareness and preventive measures for skin health. Gradually, this trend is reducing dependence on prescriptions and enabling constant interaction with customers through both the retail sales network and online health portals.
- The most promising opportunity expected in the next few years is in the form of OTC topical products based on hydrocortisone drugs for eczema and dermatitis cases in the U.S. hydrocortisone market because of the high frequency of occurrence and penetration through the retail networks. In addition, the increasing need for specialized formulations for sensitive skins, children and anti-itch formulations among others provides an opportunity in volume. Lastly, innovation in consumer healthcare in the form of sprays and gels is growing in popularity.
- In order to gain an edge, firms need to concentrate on product differentiation based on formulation and consumer-driven brand messaging. They will also need to increase their presence within the pharmacy channel, online commerce, and even through consumer-focused channels. Efforts made towards clinically validated and dermatologically positioned formulations will prove to be crucial within such a competitive OTC environment. In addition, partnerships with retail partners as well as healthcare professionals and even entry into adjacent markets will allow for greater market exposure.
Market Segmentation
- Type Insights (Revenue, USD Mn, 2021 - 2033)
- Branded
- Generics
- Route of Administration Insights (Revenue, USD Mn, 2021 - 2033)
- Parenteral
- Oral
- Topical
- Acute Care Indication Insights (Revenue, USD Mn, 2021 - 2033)
- Sepsis and Septic Shock
- Adrenal Crisis and Peri-operative Stress-dose Steroids.
- Acute Asthma/COPD Exacerbations
- Severe allergic reactions and anaphylaxis
- Neuro/Trauma
- Others
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Acute Care Hospitals
- Hospital-based outpatient clinics and Infusion Centers
- Community Clinics and Urgent Care Centers
- Retail / community care
- Others
- Distribution Channel Insights (Revenue, USD Mn, 2021 - 2033)
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Key Players Insights
- Pfizer Inc.
- Johnson & Johnson
- Bayer AG
- GlaxoSmithKline plc
- Viatris Inc.
- Teva Pharmaceutical Industries Ltd.
- Sanofi S.A.
- Sun Pharmaceutical Industries Ltd.
- Hikma Pharmaceuticals PLC
- Novartis AG
Sources
Primary Research Interviews
- Dermatologists / Clinical Specialists (U.S., Canada)
- Pharmaceutical Product Managers – Topical Corticosteroids
- Retail Pharmacy Chain Executives (OTC segment)
- Regulatory Affairs Experts – Dermatology & OTC Drugs
- Hospital Pharmacists & Procurement Heads
Databases
- Centers for Medicare & Medicaid Services (CMS) – Prescription utilization, reimbursement trends
- National Health Service (for benchmarking) – Dermatology treatment trends
- StatCan – Healthcare expenditure, population health data
Magazines
- Dermatology Times – Skin disease trends, treatment innovations
- Pharmaceutical Technology – Drug formulation and OTC market insights
- Drug Store News – Retail pharmacy and OTC trends
- Managed Healthcare Executive – Healthcare access and treatment patterns
Journals
- Journal of the American Academy of Dermatology – Clinical insights on skin disorders
- British Journal of Dermatology – Treatment efficacy and dermatology research
- JAMA Dermatology – Epidemiology and therapy trends
Newspapers
- The Wall Street Journal – OTC drug market trends, pharmaceutical company strategies
- The New York Times – Consumer healthcare trends and dermatology awareness
- Financial Times – Global pharmaceutical industry developments and market expansion
- The Washington Post – U.S. healthcare access, retail pharmacy, and OTC adoption trends
- USA Today – Consumer health behavior and over-the-counter medication usage
Associations
- American Academy of Dermatology – Clinical guidelines and prevalence data
- Canadian Dermatology Association – Treatment practices and awareness
- Consumer Healthcare Products Association – OTC drug usage trends
Public Domain Sources
- U.S. Food and Drug Administration (FDA) – Drug approvals, OTC regulations
- Health Canada – Drug safety and regulatory framework
- Centers for Disease Control and Prevention (CDC) – Skin condition prevalence data
- National Institutes of Health (NIH) – Dermatology research and clinical studies
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients