North America Pharmaceutical Stability and Storage Services Market Size and Forecast – 2026-2033
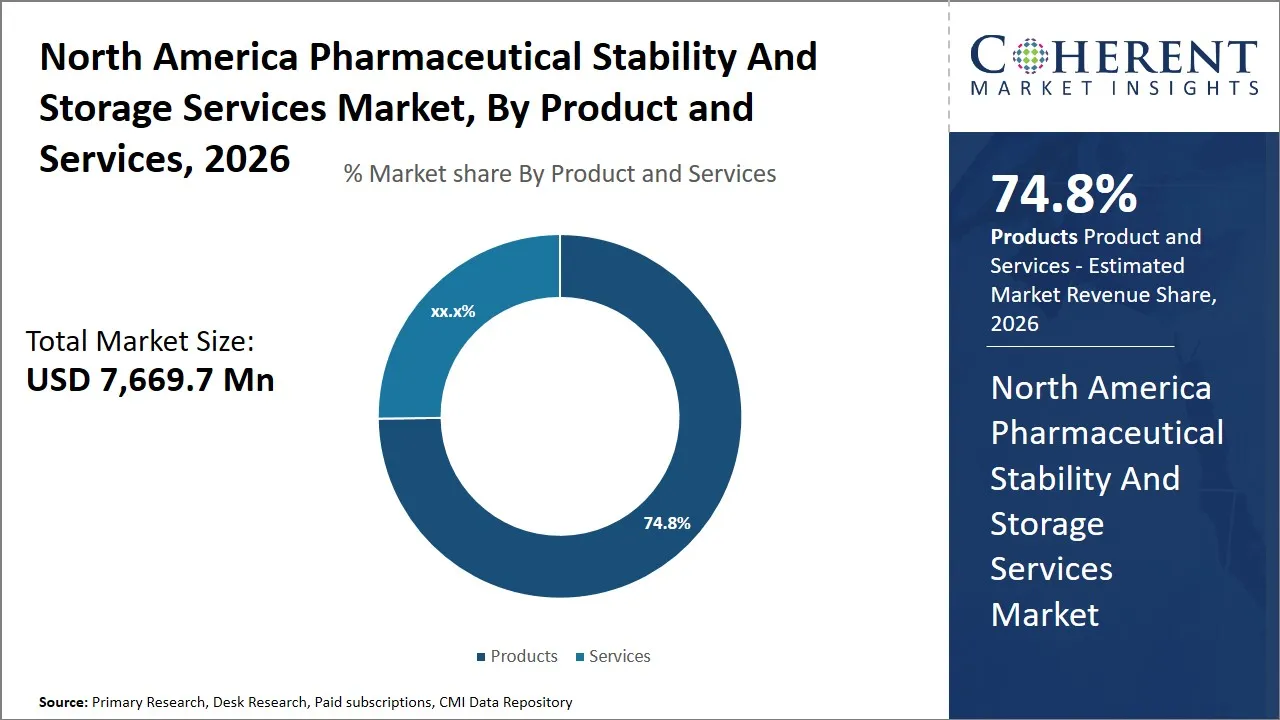
The North America pharmaceutical stability and storage services market is estimated to be valued at USD 7,669.7 Mn in 2026 and is expected to reach USD 10,611.9 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.7% from 2026 to 2033. This steady growth reflects increasing demand for controlled storage solutions that ensure the integrity and efficacy of pharmaceutical products throughout their lifecycle across the region.
Key Takeaways of the North America Pharmaceutical Stability and Storage Services Market
- Products segment is expected to lead the North America pharmaceutical stability and storage services market, capturing 74.8% share in 2026.
- Finished formulations segment is estimated to represent 42.3% of the pharmaceutical stability and storage services market share in 2026.
- Pharmaceutical and biotechnology companies segment is projected to dominate with 45.3% of the North America pharmaceutical stability and storage services market share in 2026.
Market Overview
- North America is a leading market for pharmaceutical stability and storage services due to its strong pharmaceutical and biotechnology industry base.
- The North America pharmaceutical stability and storage services market is growing steadily with rising demand for outsourced stability testing, controlled storage, and long-term sample management services.
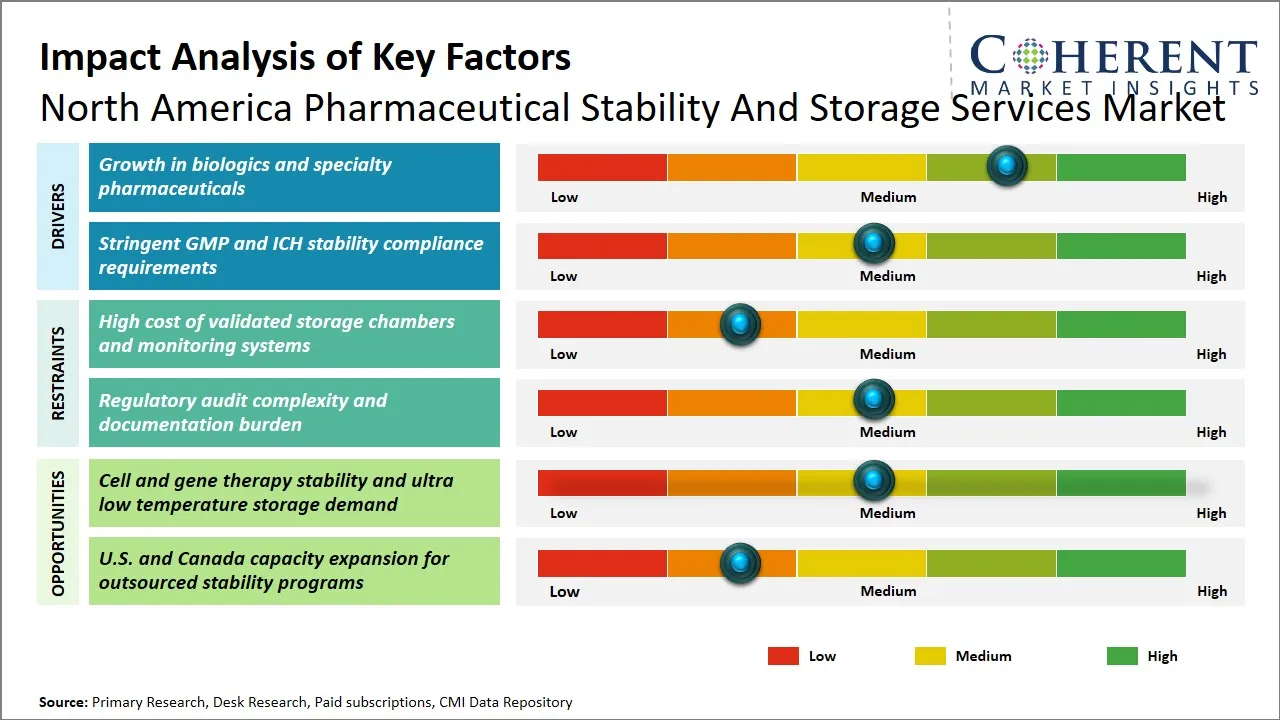
- Growth is being driven by increasing biologics, specialty drugs, and complex formulations that require validated temperature-controlled environments.
- Stringent U.S. Food and Drug Administration (FDA) and International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) compliance requirements are pushing pharmaceutical companies to rely on specialized third-party service providers.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Expansion of stability storage capacity across North American biopharma hubs |
|
|
Growth in integrated storage and analytical service models |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Products Segment Dominate the North America Pharmaceutical Stability and Storage Services Market in 2026?
The products segment is expected to hold the largest market share of 74.8% in 2026, owing to the critical role these solutions play in maintaining the integrity and efficacy of pharmaceutical compounds. The variety of products such as refrigerators, storage systems, stability chambers, containers, drums, racking, and shelving systems satisfy the high and diverse demands of pharmaceutical stability tests and long-term storage requirements. The increasing complexity of pharmaceutical preparations and biologics has increased the need to have complex storage facilities which provide temperature consistency, safety, and regulatory adherence.
Special storage facilities and fridges are developed to give controlled conditions that help in maintaining the drugs stability particularly those that are sensitive to temperature such as pharmaceutical drugs like vaccines and biologics. Stability chambers also recreate different conditions in terms of temperature and humidity enabling manufacturers to test the performance of their products in accelerated and extended storage conditions. Raw materials and finished products are physically fixed into containers and drums and offers limited chances of contamination. At the same time, novel racking and shelving solutions maximize the use of space in warehouses as there is growing demand on capacity with increasing volumes of production.
For instance, in May 2025, SK pharmteco announced the expansion of its stability storage capacity at its California facility to support increasing demand for small-molecule drug substance and drug product programs. The expansion includes additional current Good Manufacturing Practice-compliant stability chambers designed to meet International Council for Harmonisation stability conditions, strengthening the company’s ability to provide controlled storage and monitoring services for pharmaceutical clients across North America.
(Source- SK pharmteco)
Why Does the Finished Formulations Segment Dominate the North America Pharmaceutical Stability and Storage Services Market in 2026?
The finished formulations segment is expected to hold 42.3% of the market share in 2026, driven predominantly by the pharmaceutical industry's priority on final drug product quality and regulatory compliance. The finished formulations include tablets, capsules, injectables and other dosage forms which require thorough stability investigation to confirm to safety, efficacy and shelf-life standards prior to market release.
The focus on complete formulations can be explained by the acute necessity to observe the behavior of the ultimate drug product under different environmental conditions with time. To allow entry in the market and post-market monitoring, regulatory authorities require complete stability information on finished products to allow them entry into the market. This results in a long-term stability testing and storage services that are customized in these formulations.
For instance, in June 2025, the U.S. Food and Drug Administration released draft Q1 Stability Testing guidance covering stability data requirements for drug substances and finished drug products. The guidance emphasizes validated storage conditions and long-term stability studies needed to establish shelf life and labeling for marketed formulations, reinforcing strong demand for stability and storage services supporting finished pharmaceutical products in North America.
(Source- U.S. Food and Drug Administration)
The Pharmaceutical and Biotechnology Companies Segment Dominates the North America Pharmaceutical Stability and Storage Services Market
The pharmaceutical and biotechnology companies segment is expected to hold the largest market share of 45.3% in 2026, due to their expansive product pipelines and stringent regulatory commitments. These companies are required to manage complex stability and storage processes throughout the drug development lifecycle, from early research and clinical trials to commercialization, to ensure product safety, efficacy, and regulatory compliance.
The increase in the number of innovator drugs, biosimilars, and biologics is causing pharmaceutical and biotechnology companies to spend heavily on stability chambers, refrigerators, storage rooms, and various testing services. The necessity to certify credible long-term, medium, and fast stability information to match the international regulatory frameworks precipitates their demands of quality stability infrastructures and storage structures.
North America Drug Candidate Activity in Phase I to Phase III Clinical Trials
- North America has continued to be a prime location of drug candidates in Phase I to Phase III clinical trials, due to the high pharmaceutical R&D relationship, trial infrastructure, and biotech innovation concentration of the U.S. According to Pharmaceutical Research and Manufacturers of America, over 8,000 medicines are under a state of clinical development around the globe, and IQVIA 2025 R&D trends report has indicated that the core activity of clinical trial starts and pipeline remains robust, further supporting the centrality of North America in the global development picture.
- For a North America specific read, the latest market data indicates that Phase III trials accounted for 48.8% of the North America clinical trials market in 2025, reflecting a strong base of late-stage development activity. In parallel, ClinicalTrials.gov continues to show the United States as the dominant location globally for registered studies, underscoring North America’s leadership in the volume and progression of drug candidates through clinical development, particularly in advanced biologics and other high value therapeutic areas.
Market Players, Key Developments, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On January 27, 2026, Astoriom acquired Precision Stability Storage, expanding its pharmaceutical stability storage capabilities across the U.S. The acquisition adds multiple facilities in North Carolina, Florida, and Massachusetts, increasing Astoriom’s geographic coverage and capacity to serve biopharma and medical device companies requiring GMP-compliant storage services.
- In March 2025, DHL announced plans to acquire U.S.-based pharmaceutical logistics specialist, Cryopdp, which provides storage, packaging, and transportation services for pharmaceutical and biotech products. The deal strengthens DHL’s Life Sciences and Healthcare division and expands cold-chain and specialized pharmaceutical handling capabilities in North America.
- In July 2024, Cambrex announced the expansion of its stability storage business through Q1 Scientific with a new state-of-the-art stability storage facility in Durham, North Carolina. The facility provides advanced temperature-controlled storage environments and is located near Cambrex’s analytical testing operations, enabling integrated stability testing and storage services for pharmaceutical and biotechnology clients.
Growing Adoption of IoT-Enabled Stability Chambers and Remote Monitoring Systems in North America
- In the North America pharmaceutical stability and storage services market, the growing emphasis on tighter control over temperature, humidity, alarm systems, and data integrity is driving the adoption of IoT-enabled stability chambers and remote monitoring technologies among service providers and drug developers. This trend is being further strengthened by the tightness of expectations in the stability-data in the 2025 draft Q1 guidance by the U.S. Food and Drug Administration, which makes continuous monitoring, audit-ready records, and validated environmental control systems more valuable. North America too is the market leader in the regional market in terms of pharmaceutical stability and storage since it has a high biopharma base and compliance intensity.
- Practically, the IoT-powered chambers and remote monitoring platforms assist the operator to shift towards manual checks to real-time alerts, centralized management, and quicker reaction to excursions. The capabilities that vendors in this space point to include 24/7 remote alarming, secure electronic records, scalability to several chambers or locations, and automated reporting; all of which has a direct impact on GMP and GxP compliance requirements. In the case of North America, this is turning out to be a competitive differentiator since clients are gradually becoming more interested in storage companions who can provide interconnected infrastructure, lower the risk of compliance and business continuity assistance.
Top Strategies Followed by North America Pharmaceutical Stability and Storage Services Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Leading players strengthen their position through heavy R&D investment, strategic partnerships, and expansion of integrated service networks across key pharmaceutical hubs. This helps them deliver more compliant, high-performance stability and storage solutions while widening their reach in North America and other growth markets. |
In August 2025, Almac expanded its U.S. analytical capacity in Souderton, Pennsylvania with new GMP-qualified analytical stability chambers, reinforcing its integrated stability and analytical services network for pharmaceutical clients. This move reflects how established players use continuous investment and network expansion to improve service depth and widen their regional reach. |
|
Mid-Level Players |
Mid-level companies compete by offering cost-effective and flexible stability and storage solutions that balance compliance, quality, and affordability for pharmaceutical clients. They often strengthen their position through collaborations, technology integration, and scalable service models that help them serve a broader client base efficiently. |
Alcami expanded its advanced pharma storage services in North Carolina in 2024 to provide flexible storage, stability, and cold-chain support for a wide range of customer needs. |
|
Small-Scale Players |
Small-scale players compete by focusing on niche, specialized services tailored to specific client needs, often using technologies such as remote monitoring, modular storage setups, and customized sample management to stand out from larger rivals. Their strength lies in faster response, localized service, and closer relationships with startups and regional biopharma firms. |
Precision Stability Storage built its position through specialized off-site International Council for Harmonisation (ICH) stability storage and biological sample management services across multiple U.S. locations before being acquired by Astoriom in January 2026. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
North America Pharmaceutical Stability and Storage Services Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 7,669.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.7% | 2033 Value Projection: | USD 10,611.9 Mn |
| Segments covered: |
|
||
| Companies covered: |
Catalent, Almac Group, Charles River Laboratories, Eurofins Scientific, Intertek, SGS, Alcami Corporation, Element Materials Technology, BioLife Solutions SciSafe, Cambrex Q1 Scientific, Q Laboratories, Precision Stability Storage, Frontage Laboratories, Thermo Fisher Scientific Patheon, and Labcorp Drug Development |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
North America Pharmaceutical Stability and Storage Services Market Dynamics
To learn more about this report, Request Free Sample
North America Pharmaceutical Stability and Storage Services Market Driver - Growth in Biologics and Specialty Pharmaceuticals
The growth and uptake of biologics and specialty pharmaceuticals in North America is increasing at a very high rate thus necessitating advanced stability and storage services. These biologic molecules, such as monoclonal antibodies, gene therapies, and recombinant proteins, demand stringent temperature control and special handling to ensure their effectiveness and safety along the supply chain.
In contrast to more conventional small-molecule pharmaceuticals, biologics are more susceptible to environmental factors like temperature changes, humidity, and exposure to light, which requires sophisticated storage facilities as well as intense stability testing guidelines. Also, the influx of specialized medicine and customized medicines has resulted in an expanding number of specialty pharmaceuticals with short shelf life and require specialized storage facilities.
For instance, in February 2025, Cencora, a global pharmaceutical solutions company, announced to invest USD 1 billion through 2030 to bolster and expand its pharmaceutical distribution network in the U.S. The investments headlined by the opening of its second national distribution center in Ohio and an expanded presence in Alabama and California will increase Cencora’s capacity, improve efficiency and enhance the resilience of its national distribution network, strengthening the company’s ability to support its customers’ evolving needs.
(Source- Cencora)
North America Pharmaceutical Stability and Storage Services Market Opportunity - Rising Demand for Cell and Gene Therapy Stability and Ultra-Low Temperature Storage
A huge opportunity is being experienced in the pharmaceutical stability and storage services market in North America due to the accelerated development in cell and gene therapy. These state-of-the-art treatments demand a high level of stability testing, and special storage conditions, especially ultra-low temperature ones, to preserve the effectiveness and safety of these treatments within time limits.
Due to the growing regulatory approval of cell and gene therapies and commercial interest, there is an increasing need to find stable services that meet the needs of these biologics. Low temperature storage that is frequently between -80 and -196 C is essential to maintain the fragile biological constituents of the food, to forestall degradation, and to guarantee product integrity throughout the transportation and long term storage of the products. This has led pharmaceutical firms and contract development and manufacture organizations, (CDMOs) to consider finding sophisticated storage systems with the capability of intricate monitoring, security, and compliance capabilities.
For instance, in October 2024, Cryoport, Inc. entered a strategic partnership with VGXI, Inc., a GeneOne Life Science company, to provide specialized biostorage, logistics, and supply chain solutions for plasmid DNA products, supporting gene therapy and mRNA-based treatment programs. Beginning in October 2024, the partnership utilizes Cryoport's Houston, Texas facility to support VGXI's manufacturing sites in The Woodlands and Conroe, Texas.
(Source- Cryoport, Inc.)
Analyst Opinion (Expert Opinion)
- North America pharmaceutical stability and storage services market is also expanding with more drug developers outsourcing stability services to more biologics, specialty drugs and temperature sensitive products. Tighter regulatory expectations, such as the U.S. Food and Drug Administration draft Q1 stability guidance released in June 2025 and new capacity addition, such as Cambrex opening a facility in Durham in October 2024, are helping to support growth.
- The opportunity is also evident in those providers who offer a one-platform solution of storing, monitoring, and compliance support. Technology exchange within the pharmaceutical development chain is still facilitated through industry activities like INTERPHEX but the market is also becoming more demanding with an increased validation cost, increased quality demands and increased pressure to provide multi-site backup and integrated services.
Market Segmentation
- Product and Services Insights (Revenue, USD Mn, 2021 - 2033)
- Products
- Refrigerator
- Storage System
- Stability chamber
- Containers and Drums
- Racking and Shelving Systems
- Others (Bulk Bags, etc.)
- Services
- Stability Testing Services
- Long Term
- Intermediate
- Accelerated
- Storage
- Room temperature storage
- Cool and cold storage
- Freezer storage
- Stability Testing Services
- Products
- Sample Type Insights (Revenue, USD Mn, 2021 - 2033)
- Finished Formulations
- Small Molecules
- Large Molecules
- Bulk API
- Cell line and compound libraries
- Others (Reagents and Nucleic Acid etc.)
- Finished Formulations
- End User Insights (Revenue, USD Mn, 2021 - 2033)
- Pharmaceutical and Biotechnology Companies
- Hospitals and Clinics
- CDMO and CROs
- Others (Research and Academic Institutes etc.)
- Key Players Insights
- Catalent
- Almac Group
- Charles River Laboratories
- Eurofins Scientific
- Intertek
- SGS
- Alcami Corporation
- Element Materials Technology
- BioLife Solutions SciSafe
- Cambrex Q1 Scientific
- Q Laboratories
- Precision Stability Storage
- Frontage Laboratories
- Thermo Fisher Scientific Patheon
- Labcorp Drug Development
Sources
Primary Research Interviews
Industry Stakeholders list
- Stability study directors
- Quality assurance and compliance heads
- GMP warehouse and cold chain managers
- Analytical testing laboratory managers
- Regulatory affairs directors
- Clinical and commercial supply chain heads
End-users list
- Pharmaceutical manufacturing companies
- Biopharmaceutical companies
- Contract development and manufacturing organizations
- Clinical trial sponsors
- Hospital research pharmacies
- Biobanks and sample repository operators
Government and International Databases
- U.S. Food and Drug Administration
- Health Canada Drug Product Database
- ClinicalTrials.gov
- National Institutes of Health RePORTER
- International Council for Harmonisation guidelines database
Trade Publications
- Pharmaceutical Technology
- BioPharm International
- Contract Pharma
- Drug Development and Delivery
- Pharmaceutical Online
- Pharma Manufacturing
Academic Journals
- Journal of Pharmaceutical Sciences
- International Journal of Pharmaceutics
- AAPS Journal
- PDA Journal of Pharmaceutical Science and Technology
- European Journal of Pharmaceutics and Biopharmaceutics
- Pharmaceutics
Reputable Newspapers
- Financial Times
- The Wall Street Journal
- The New York Times
- The Washington Post
Industry Associations
- Parenteral Drug Association
- International Society for Pharmaceutical Engineering
- American Association of Pharmaceutical Scientists
- United States Pharmacopeia
- Drug Information Association
- INTERPHEX Conference Network
Public Domain Resources
- Drugs@FDA
- Federal Register
- USP General Chapter
- FDA Guidance Documents Database
- National Center for Biotechnology Information Bookshelf
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients