Global Respiratory Monitoring Equipment Market Size and Forecast – 2026 To 2033
The global respiratory monitoring equipment market is expected to grow from USD 6.85 Bn in 2026 to USD 10.62 Bn by 2033, registering a compound annual growth rate (CAGR) of 6.5%. The market for respiratory monitoring equipment is poised for significant expansion, fueled by the soaring global burden of respiratory diseases.
According to the World Health Organization, chronic respiratory diseases (CRDs) are among the most common noncommunicable diseases (NCDs), and are the sixth leading cause of death in the WHO European Region, with almost 400,000 deaths every year, and nearly 80% of these deaths are caused by chronic obstructive pulmonary disease (COPD).
(Source: World Health Organization)
Key Takeaways of the Global Respiratory Monitoring Equipment Market
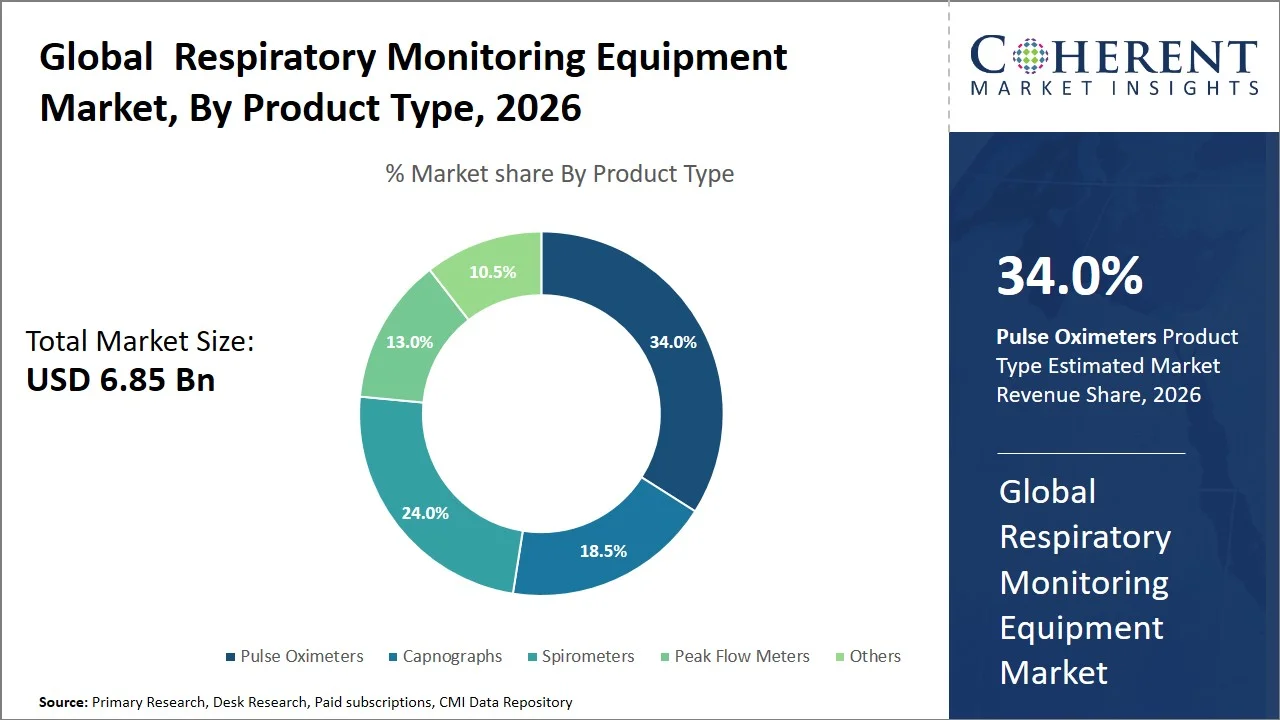
- Pulse oximeters are projected to hold 34.0% of the global respiratory monitoring equipment market share in 2026, making it the dominant product type segment, with North America leading adoption due to the widespread integration of pulse oximeters into hospital monitoring protocols and continuous patient surveillance systems. Pulse oximetry has been extensively employed as the standard for oxygen measurement that is non-invasive and instant in nature in both intensive care units and emergencies. For example, according to the U.S. Food and Drug Administration, pulse oximeters have been widely employed by clinicians for continuous monitoring of oxygen saturation. (Source: Food and Drug Administration)
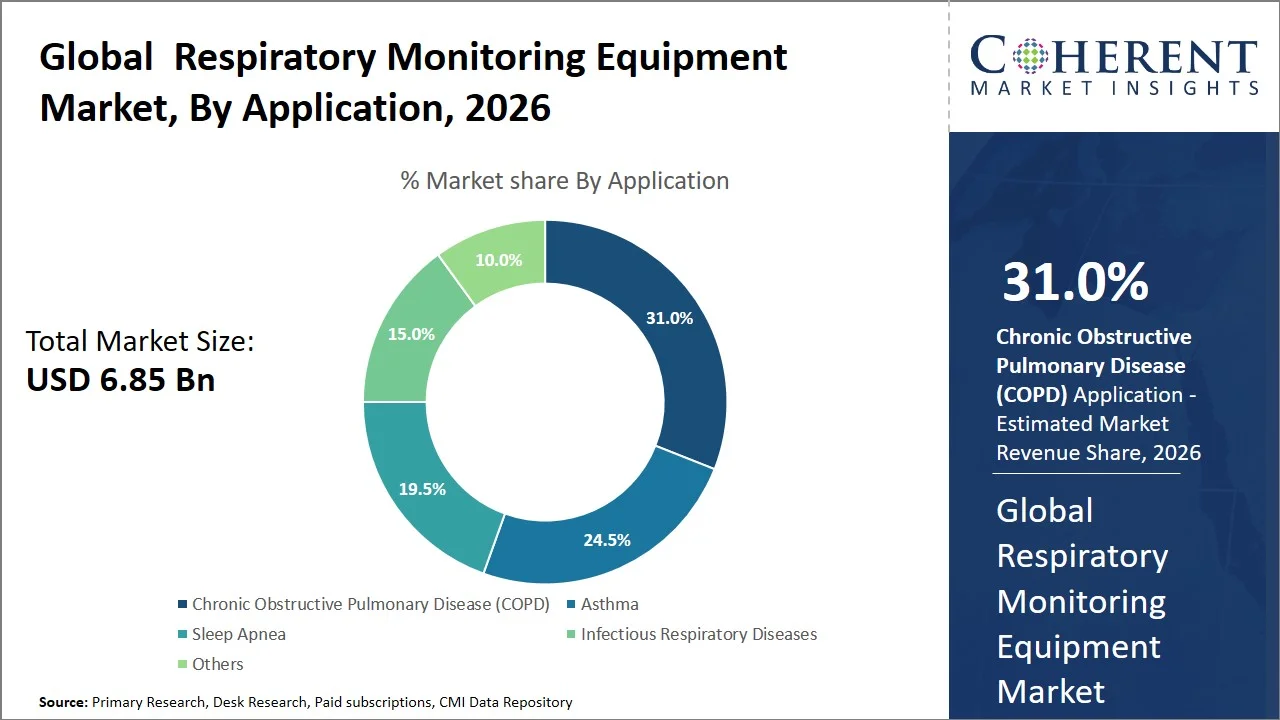
- Chronic Obstructive Pulmonary Disease (COPD) is projected to hold 31.0% of the global respiratory monitoring equipment market share in 2026, making it the dominant application segment, with Asia Pacific emerging as a key market owing to the increasing prevalence of Chronic Obstructive Pulmonary Disease (COPD) and the long-term management needs. For instance, according to the World Health Organization, respiratory diseases account for 12% of all deaths in the South-East Asia region, with 1.56 million deaths reported in 2021, including 1.24 million deaths from chronic obstructive pulmonary disease (COPD), and 37% of these deaths occurring before the age of 70. (Source: World Health Organization)
- Hospitals are projected to hold 42.0% of the global respiratory monitoring equipment market share in 2026, making it the dominant end user segment, with North America maintaining leadership owing to the availability of advanced critical care and patient monitoring infrastructure in the region. For instance, according to the American Hospital Association, there are more than 6,100 hospitals in the U.S., equipped with intensive care units and respiratory monitoring systems for managing acute and chronic conditions. The extensive hospital network and continuous need for real-time respiratory monitoring remain fundamental to the adoption of respiratory monitoring equipment across healthcare facilities. (Source: American Hospital Association)
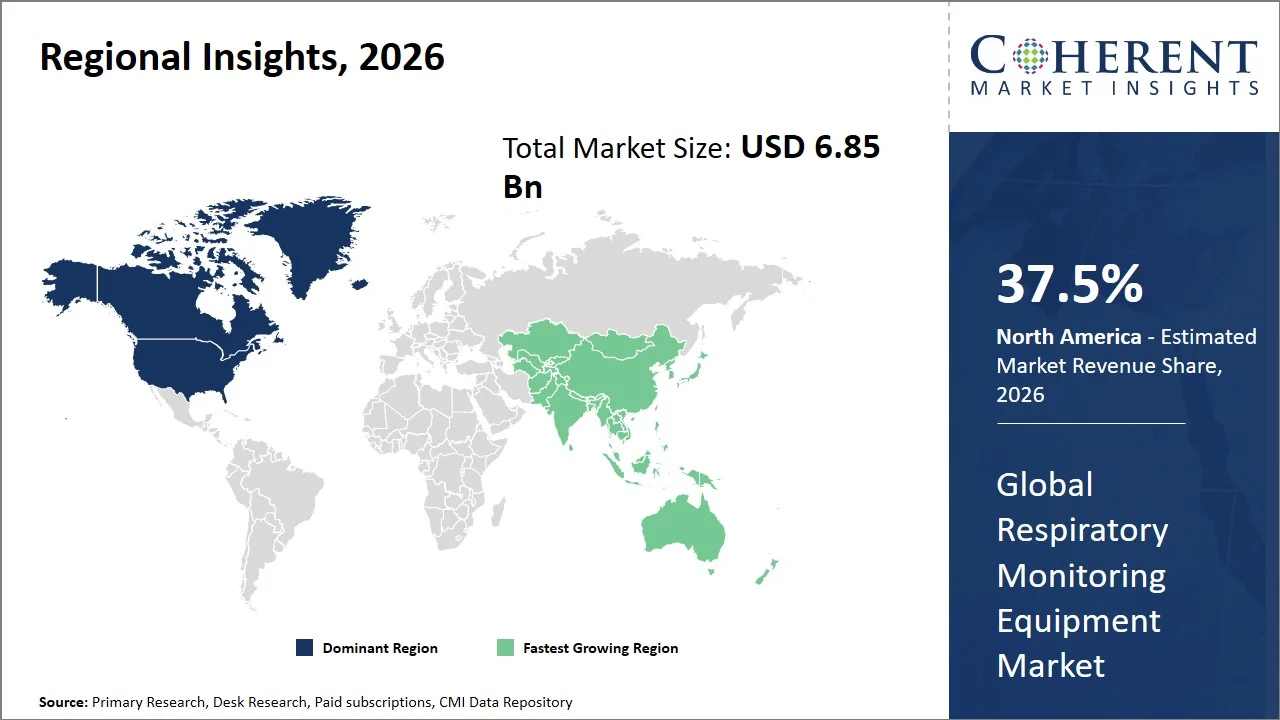
- The North America region maintains its dominance with an expected share of 37.5% in 2026, supported by robust clinical infrastructures and well-established standardized patient monitoring protocols. For instance, in the U.S., the expansion of remote patient monitoring reimbursement under Centers for Medicare & Medicaid Services (CMS) has significantly accelerated the adoption of continuous respiratory monitoring devices across homecare and post-acute settings, particularly for chronic respiratory conditions such as chronic obstructive pulmonary disease (COPD). (Source: Centers for Medicare & Medicaid Services)
- Asia Pacific is expected to exhibit the fastest growth with an estimated contribution of 31.5% share in 2026, attributed to the growing accessibility to healthcare and rapid infrastructure development in the region. For instance, in India, the government’s Ayushman Bharat program is enhancing secondary and tertiary healthcare capabilities, resulting in the higher usage of patient monitoring devices at public hospitals, while concurrent investments in China’s intensive care infrastructure (181,000 ICU beds in 2022, equivalent to 12.8 beds per 100,000 population, with national targets to reach 15 ICU beds per 100,000 by 2025) are promoting the uptake of sophisticated respiratory monitoring devices. (Source: National Health Authority, National Medical Products Administration)
- Increasing Use of Respiratory Monitoring in Post-Acute and ICU Care Settings: Continuous respiratory monitoring devices in ICUs and post-operational wards are gaining importance and there is an increasing need for advanced monitoring devices. Accurate and real time monitoring of vital information such as oxygen saturation, breathing rate, and ventilator status are very important as they help detect any possible complication at the earliest. Monitoring systems have gained popularity among hospitals to enhance their performance and minimize ICU time for patients.
- Integration of Wearables and Telehealth for Remote Patient Management: Wearable sensors for breathing monitoring and telemedicine tools are now being used to enable constant monitoring of patients from non-medical locations. Remote monitoring of important respiratory parameters allows doctors to intervene immediately in case of any decline. The move towards the decentralization of health care services is driving greater adoption of connected health systems. It also opens up a specialized market for cloud-connected breathing monitoring devices.
Why Do Pulse Oximeters Dominate the Global Respiratory Monitoring Equipment Market?
Pulse oximeters are projected to hold a market share of 34.0% in 2026, attributed to their non-invasive oxygen saturation measurement, affordability, and versatility in applications. Pulse oximeters are considered the first line of equipment used to monitor patients since unlike arterial blood gas tests, which require invasive procedures to obtain blood samples, they measure SpO₂ values instantly. The use of pulse oximeters increased even further due to the advent of the novel coronavirus, especially because of increased home and remote monitoring of patients that allowed early detection of “silent hypoxia”. (Source: National Library of Medicine)
Furthermore, advancements in technology such as wireless, wearable, and mobile-connected pulse oximeters have led to wider usage of these devices in telemedicine and the management of chronic diseases including Chronic Obstructive Pulmonary Disease (COPD). For instance, Masimo W1 medical watch by Masimo for continuous, real-time oxygen saturation monitoring across hospital, home, and remote care settings enables clinicians to track patients’ respiratory status outside traditional clinical environments (Source: Masimo). In terms of regulation, closer monitoring and guidance from regulatory bodies, such as the U.S. Food and Drug Administration, to ensure better accuracy and standardization of devices are further enhancing market adoption and credibility, thus sustaining growth within both mature and developing health care markets. (Source: Food and Drug Administration)
Why Does Chronic Obstructive Pulmonary Disease (COPD) Represent the Largest Application Segment in the Respiratory Monitoring Equipment Market?
To learn more about this report, Request Free Sample
Chronic Obstructive Pulmonary Disease (COPD) is projected to hold a market share of 31.0% in 2026, because of its progressive and chronic nature, requiring the need for constant monitoring of oxygen levels as well as identification of exacerbation. Unlike other respiratory disorders, chronic obstructive pulmonary disease (COPD) requires constant monitoring within both hospitals and home settings, making devices like pulse oximeters indispensable. According to the World Health Organization, about three million people around the world lost their lives in 2021 due to chronic obstructive pulmonary disease (COPD), approximately 5% of all global deaths, necessitating the need for constant monitoring to avoid death resulting from complications. (Source: World Health Organization)
From the industry perspective, organizations such as Philips have developed technology in connected care solutions by integrating pulse oximetry with digital health platforms for chronic obstructive pulmonary disease (COPD) disease management beyond hospitalization, reinforcing the fact that advancements in technology continue to solidify chronic obstructive pulmonary disease (COPD)’s position as the major application segment. For instance, Philips’ eCareCoordinator and eCareCompanion platforms enable clinicians to remotely monitor COPD patients by integrating vital parameters such as oxygen saturation (via pulse oximetry) with symptom tracking and care plans, supporting continuous disease management and early intervention in homecare settings. (Source: Philips - eCareCoordinator, eCareCompanion)
Hospitals Segment Dominates the Global Respiratory Monitoring Equipment Market
The hospitals segment is projected to hold a market share of 42.0% in 2026, as they serve as primary care settings for acute respiratory management, critical care, and perioperative monitoring, all of which require continuous use of multi-parameter respiratory monitoring equipment. The availability of ICUs, emergency wards, and advanced operating rooms has led to the heavy use of respiratory devices like pulse oximeters, capnography devices, and ventilated monitors. For instance, in the U.S., there exist 4,846 adult hospitals with nearly 79,876 adult critical care beds, indicating the magnitude of the inpatient facility that necessitates continuous respiratory monitoring. (Source: National Library of Medicine).
Furthermore, the presence of encouraging reimbursement structures from Centers for Medicare & Medicaid Services continue to encourage the adoption of continuous monitoring technologies within hospital settings, particularly through Remote Patient Monitoring (RPM) and Remote Therapeutic Monitoring (RTM) codes (CPT 99453, 99454, 99457, 99458; RTM: 98975, 98976, 98977, 98980, 98981), which incentivize continuous tracking of patient vitals including respiratory parameters. (Source: Centers for Medicare & Medicaid Services)
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
U.S. FDA Draft Guidance on Pulse Oximeters (January 2025) |
|
|
EU Medical Device Regulation (MDR 2017/745) – Transition Enforcement for Monitoring Devices (2024–2026) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Respiratory Monitoring Equipment Market Dynamics
To learn more about this report, Request Free Sample
Market Drivers
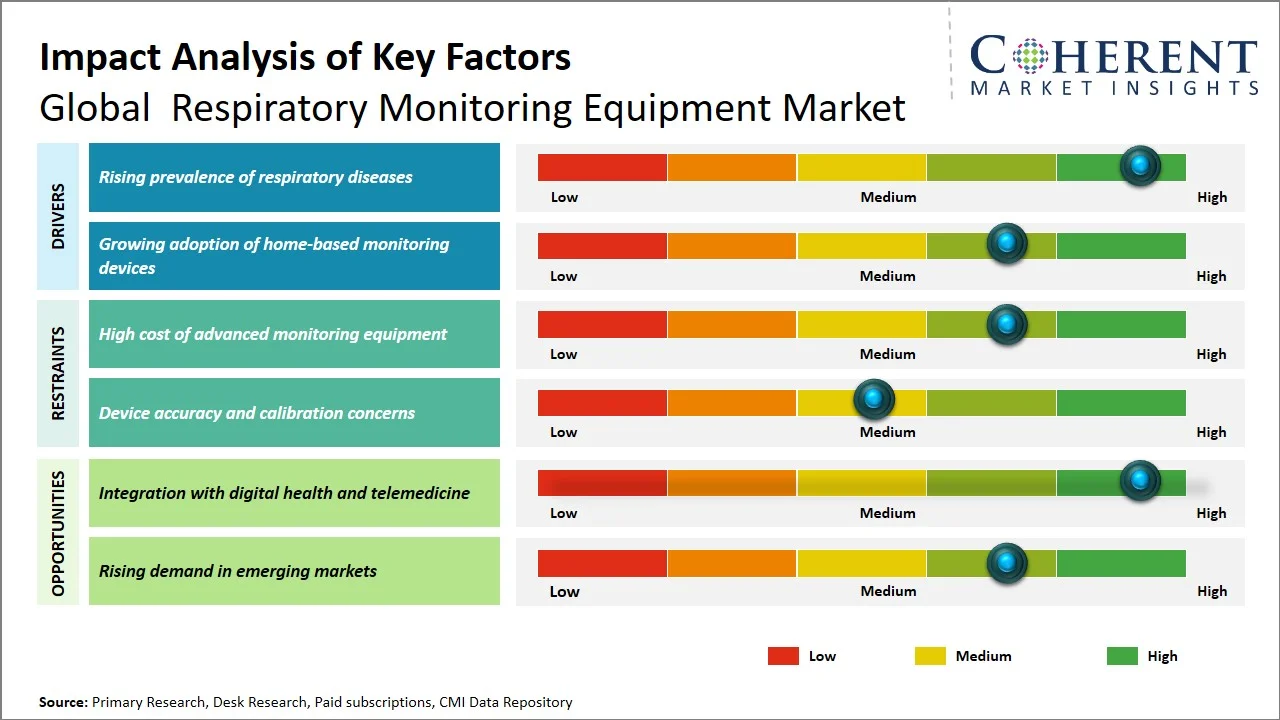
- Rising prevalence of respiratory diseases: The rising prevalence of chronic respiratory disorders (CRDs) plays a major role in fueling the demand for respiratory monitoring equipment, as these medical conditions necessitate the constant evaluation of oxygen saturation and pulmonary functions. Based on the statistics provided by the Institute for Health Metrics and Evaluation, the total number of prevalent cases associated with chronic respiratory disorders was estimated to be around 454.6 million people, which resulted in more than 4.0 million deaths, positioning this disease among the top three causes of death. Here, chronic obstructive pulmonary disease accounted for more than 212 million cases, along with causing the death of 3.3 million people, whereas asthma had impacted 262 million people (Source: Institute for Health Metrics and Evaluation). While there has been some improvement in age-standardized prevalence rate, the absolute burden of diseases is steadily rising, especially in developing nations attributed to pollution, tobacco use, and occupation-related exposures.
- Growing adoption of home-based monitoring devices: Growing preference for decentralized care has been increasing the need for respiratory monitors in homes, especially due to chronic ailments that demand continuous monitoring of vital signs, away from hospitals. For instance, U.K.’s National Health Service introduced a COVID Oximetry@home initiative, where patients were provided with pulse oximeters to monitor their SpO₂ levels regularly—showing decreased burden on hospitals and early detection of respiratory deterioration (Source: National Health Service England). Similarly, in India, with the introduction of the eSanjeevani telemedicine system, teleconsultations increased significantly through the deployment of basic medical instruments, such as pulse oximeters, for home-based quarantining and disease management (Source: Ministry of Health and Family Welfare). The rising acceptance of home-based monitoring devices by clinicians and their implementation in practical scenarios have been fueling the increasing use of portable respiratory monitors in developed and developing healthcare environments.
Emerging Trends
- Miniaturization and Integration of Multi-Parameter Monitoring Devices: The respiratory monitoring systems are evolving in such a way that they are becoming increasingly smaller and more capable of monitoring multiple parameters including oxygen levels, breaths per minute, and air flow all at once. These devices are gaining popularity because of their increasing ease-of-use and the fact that fewer devices are needed to get accurate results.
- Growing Use of Predictive Analytics for Early Respiratory Deterioration Detection: In recent years, there has been increased emphasis on the integration of predictive analytics in respiratory monitoring systems to detect any early warnings regarding the health status of the patients. Continuous analysis of the data streams enables such systems to trigger alarms before the event occurs. This development is especially applicable in the ward and home environments.
Regional Insights
To learn more about this report, Request Free Sample
Why is North America a Strong Market for Respiratory Monitoring Equipment?
North America leads the global respiratory monitoring equipment market, accounting for an estimated 37.5% share in 2026, due to the fast regulatory enablement of digital health solutions and their wide implementation within integrated care models. For example, the U.S. FDA had granted approvals for several digital health devices such as Apple Watch Blood Oxygen App, allowing for SpO₂ measurements in the field, thereby extending the applications of respiratory monitoring outside of clinical care. (Source: Apple)
Similarly, Kaiser Permanente has implemented remote monitoring processes whereby individuals who suffer from respiratory problems can be monitored from their homes by use of connected medical gadgets through electronic health records (Source: Kaiser Permanente). Besides, the high density of digitally-enabled hospitals and integrated delivery networks in the region ensures smooth adoption of connected respiratory monitoring solutions in inpatient and outpatient settings, strengthening North America’s position in the market.
Why Does Asia Pacific Respiratory Monitoring Equipment Market Exhibit High Growth?
The Asia Pacific respiratory monitoring equipment market is expected to exhibit the fastest growth with an estimated contribution of 31.5% share to the global market in 2026, attributed to the significant growth in public health care infrastructure, huge untapped patient base, and rising digitalization trends in emerging markets. For instance, India's Pradhan Mantri Ayushman Bharat Health Infrastructure Mission has been instrumental in developing critical care hospitals along with the integration of the public health lab system. This has resulted in a surge in the adoption of bed-side as well as portable respiratory monitoring devices in the second and third-tier locations. (Source: Ministry of Health & Family Welfare)
Furthermore, in China, the emphasis on the improvement of respiratory disease treatment and early diagnosis, as a part of Healthy China 2030 campaign, has resulted in greater acquisition of respiratory monitoring devices in primary and tertiary level hospitals (Source: ScienceDirect). In addition, the growing number of super-aged population in Japan (29.3% aged 65+ in 2024, and projected to reach 34.8% by 2040), who are facilitated through projects undertaken by the Ministry of Health, Labour and Welfare (such as the Community-based Integrated Care System and Data Health Plan), is contributing to the increased use of respiratory monitoring products for patients at home on a long-term basis. These regional and structure-related changes have contributed to the consistent high growth demand in Asia Pacific for respiratory monitoring products.
Why is the U.S. Leading Innovation and Adoption in the Respiratory Monitoring Equipment Market?
The U.S. is the leader of innovations and adoption in the market for respiratory monitoring equipment attributed to its regulatory flexibility, fast commercialization of digital health products, and large-scale implementation of solutions in real-world settings. The efforts of the U.S. Food and Drug Administration, wearable respiratory monitoring was significantly advanced, and innovations such as measuring blood oxygen saturation level and respiratory rate became available on consumer electronics, such as Apple Watch Series 9. (Source: Apple)
Moreover, regulatory processes such as 510(k) clearances for pulse oximeters ensure that there are no delays in introducing the advanced patient monitoring technologies into the market, speeding up the innovation cycle (Sources: Food and Drug Administration). Furthermore, the country gains advantages from an earlier integration of the connected health ecosystem, in which data on respiration collected from wearable devices is becoming more integrated with digital health solutions and clinical processes.
Is Japan a Favorable Market for Respiratory Monitoring Equipment?
Japan represents a prime target market for respiratory monitoring equipment owing to the super aging population within the country and effective integration into the healthcare system. As per official statistics, Japan has 36.2 million individuals above 65 years, representing 29.3% of the total population in Japan (Source: Statistics Bureau). This demographic distribution results in high incidences of respiratory diseases and requires constant monitoring of patients, which can be achieved using hospital and domiciliary monitoring systems. Additionally, government policies formulated by the Ministry of Health, Labour and Welfare, emphasize the provision of health management and surveillance at home through schemes such as the Community-based Integrated Care System that facilitates continuous healthcare and long-term care from within the community, and the Long-Term Care Insurance (LTCI) System that facilitates access to home care and surveillance. (Source: Community-based Integrated Care System, Long-Term Care Insurance (LTCI) System)
Is China Emerging as a Key Growth Hub for the Respiratory Monitoring Equipment Market?
China has emerged as one of the growth centers of the respiratory monitoring equipment market due to its large disease burden, increased surveillance programs, and rising health spending in the country. For instance, the prevalence of chronic obstructive pulmonary disease (COPD) in China is close to 100 million, contributing significantly to the global total and hence the need for continuous monitoring equipment. (Source: World Health Organization)
In addition, the China government is stepping up its efforts to monitor respiratory illnesses with a national surveillance and early warning system for pneumonia and novel infections (such as China Infectious Disease Surveillance System and the Pneumonia of Unknown Etiology Surveillance System). In addition, there is an increase in seasonal respiratory diseases in China (with human metapneumovirus accounting for 6.2% of positive respiratory tests and 5.4% of respiratory-related hospitalizations during the 2024–2025 seasonal surge) and hence the requirement for real-time monitoring devices in order to provide proper treatment is on rise. (Source: National Library of Medicine)
Why Does Germany Top the Europe Respiratory Monitoring Equipment Market?
Germany is the leader in the respiratory monitoring equipment market in Europe as a result of highly advanced hospital infrastructure, high ICU availability, and a technologically sophisticated healthcare sector. For instance, Germany accounts for the highest proportion of ICU beds among all countries (35.3 per 100,000 population), thus providing an opportunity for using continuous respiratory monitoring in critical care units (Source: MDPI). Additionally, Germany constitutes 26.5% of Europe’s medical devices market, accounting for roughly USD 44 billion (EUR 38 billion) in revenue annually, representing the largest segment with strong purchasing power and advanced technological adoption rate in the region (Source: International Trade Administration). These elements cumulatively contribute to Germany's status as a developed market with significant purchasing power, whereby robust infrastructure, high clinical application, and cutting-edge technological adoption consistently fuel the need for respiratory monitoring equipment.
Is the Respiratory Monitoring Equipment Market Developing in India?
The respiratory monitoring equipment market is rapidly growing in India, driven by a huge patient pool along with low diagnostic rate and lack of monitoring systems. The proportion of patients suffering from chronic respiratory diseases in India constitutes about 15.69% of the global patient population and more than 30.28% of related deaths, which implies over 55.23 million Chronic Obstructive Pulmonary Disease (COPD) patients in the country. (Source: The International Primary Care Respiratory) Thus, high investment in healthcare, increased awareness levels, and development of diagnostics can be viewed as important factors contributing to the adoption of respiratory monitoring equipment in hospitals and homes in India.
Regulatory Landscape Governing the Global Respiratory Monitoring Equipment Market
|
Region
|
Key Regulatory Authorities |
Description |
Impact on Market |
|
North America |
U.S. FDA (CDRH), Health Canada |
Respiratory monitoring devices are regulated as medical devices under a risk-based classification system (Class I–III). Requirements include premarket approval (510(k)/PMA), quality systems, labeling, and post-market surveillance. |
Strong regulatory oversight ensures device safety and clinical reliability, boosting adoption. However, stringent approval processes increase time-to-market and compliance costs. |
|
Europe |
EU MDR (Regulation 2017/745), Notified Bodies |
Devices fall under MDR with strict clinical evaluation, risk classification (Class I–III), and lifecycle monitoring requirements. Emphasis is on safety, traceability, and post-market surveillance. |
Enhances transparency and patient safety, supporting high-quality device adoption. However, increased documentation and certification requirements create entry barriers for smaller players. |
|
Asia Pacific |
China NMPA, Japan PMDA, India CDSCO (Medical Device Rules) |
Regulatory frameworks are evolving with risk-based classification, local clinical validation, and increasing focus on device quality and safety standards. Countries are strengthening approval pathways and compliance norms. |
Diverse regulatory systems create both growth opportunities and complexity. Faster approvals in emerging markets support adoption, while lack of harmonization challenges global scalability. |
|
Latin America |
Brazil ANVISA, Mexico COFEPRIS |
Devices are regulated under national medical device frameworks with classification systems, registration requirements, and local approvals prior to commercialization. Increasing alignment with global standards is observed. |
Gradual regulatory strengthening improves market confidence and adoption. However, approval timelines and fragmented regulations may delay product launches. |
|
Middle East & Africa |
National Health Authorities (e.g., Saudi FDA, UAE MOHAP) |
Regulations are developing, often aligned with international standards (FDA/EU). Focus areas include device registration, import controls, and basic compliance with safety standards. |
Emerging regulatory frameworks enable market entry and pilot adoption. However, limited standardization and infrastructure may restrict widespread commercialization. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
How is integration with digital health and telemedicine creating new growth opportunities in the respiratory monitoring equipment market?
The use of telemedicine has shifted the emphasis from episodic and facility-based care towards continuous care based on data collected from remote patients. Remote patient monitoring can support the real-time transfer of vital respiratory parameters such as oxygen saturation, respiratory rate, and lung functionality. This enables early identification of symptoms of deterioration and subsequent action. For instance, tele-monitoring interventions have been found to reduce admissions and emergencies and improve the quality of life and disease control in cases of chronic obstructive pulmonary disease (COPD). (Source: Dove Medical Press) Furthermore, telemonitoring and inclusion of mobile health apps and remote spirometry will increase access to healthcare services in underserved and rural areas. Incorporating artificial intelligence analytics in tele-monitoring systems is expected to create additional value through prediction of disease exacerbations and optimal care pathways.
Market Players, Key Development, and Competitive Intelligence

To learn more about this report, Request Free Sample
Key Developments
- On April 16, 2026, Strados Labs made an announcement about the purchase of the clinical trials division of NuvoAir Inc. Through the acquisition, Strados Labs will enhance its capabilities to integrate remote and clinic-based spirometry, airway inflammation measurement, and digital outcome measures. These will bolster the company's respiratory endpoint technology.
- In September 2025, Masimo Corporation expanded its strategic collaboration with Philips Healthcare to fast-track the implementation of cutting-edge monitoring technology. Masimo Corporation will integrate its pulse oximetry and sensors into Philips’ multichannel patient monitoring devices. Furthermore, the two companies have joined forces to develop future monitoring technology that is artificial intelligence enabled.
Competitive Landscape
The respiratory monitoring equipment market will likely stay highly competitive due to the increased number of chronic respiratory disease cases and rising demand for patient monitoring both clinically and at home. The rate of technological innovation of respiratory monitoring devices makes this market very competitive, especially among the device manufacturing companies. The advent of digital health platforms and remote monitoring is adding a new dimension to competitive positioning. Companies that excel in terms of technology and easy use are positioned to have an advantage over their competitors. Key focus areas include:
- Development of compact, portable, and wearable respiratory monitoring devices for home and ambulatory care
- Integration of digital health platforms, cloud connectivity, and real-time data analytics
- Enhancement of multi-parameter monitoring capabilities for comprehensive respiratory assessment
- Expansion of partnerships with hospitals, telehealth providers, and homecare service companies
Market Report Scope
Respiratory Monitoring Equipment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.85 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.5% | 2033 Value Projection: | USD 10.62 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic plc, Philips Healthcare, GE HealthCare, Masimo Corporation, Nihon Kohden Corporation, Drägerwerk AG, ResMed Inc., Schiller AG, Nonin Medical Inc., and Smiths Medical |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Analyst Opinion (Expert Opinion)
- In the coming years, the prospects of the respiratory monitoring equipment market is expected to experience a shift from the present-day approach to one where the emphasis will be on connectivity and predictive measures. Monitoring in this new setting will include not only the existing process but also monitoring through the application of sensors and software platforms in addition to other processes. The increasing number of chronic patients coupled with the provision of healthcare services outside hospitals will result in this trend.
- The largest opportunities are anticipated to be found in the home care and telemonitoring applications market, fueled by growing demands for constant monitoring outside of hospital premises. Among these, pulse oximeters and respiratory monitors are predicted to experience the greatest traction, especially within the Asia Pacific region including countries like India and China due to the huge number of patients, availability of advanced healthcare technology, and developing primary care systems.
- In order to establish a competitive advantage, companies must pay attention to the development of software-enabled clinical products that are able to integrate with telehealth and electronic medical record systems, and at the same time remain compliant with all evolving regulations. The following strategies need to be taken into consideration: the use of artificial intelligence to identify early-onset exacerbations; the creation of low-cost portable products aimed at the emerging markets; and cooperation with healthcare institutions to develop an end-to-end solution.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2021 - 2033)
- Pulse Oximeters
- Capnographs
- Spirometers
- Peak Flow Meters
- Others
- Application Insights (Revenue, USD Bn, 2021 - 2033)
- Chronic Obstructive Pulmonary Disease (COPD)
- Asthma
- Sleep Apnea
- Infectious Respiratory Diseases
- Others
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals
- Homecare Settings
- Ambulatory Care Centers
- Others
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
Sources
Primary Research Interviews
- Cryo-EM facility directors and technical specialists from national research centers (e.g., NIH-funded Cryo-EM facilities, UK national centers)
- Pharmaceutical and biotechnology companies leveraging Cryo-EM in biologics, vaccine, and structure-based drug design
- Academic researchers and structural biologists from leading institutes (e.g., IITs, EMBL, university microscopy centers)
- CRO executives and structural biology consultants involved in outsourced Cryo-EM services
Stakeholders
- Respiratory Monitoring Equipment Solution Providers: (e.g., medical device manufacturers, wearable health tech companies, patient monitoring system providers)
- End-use Sectors
- Hospitals & Clinics
- Home Healthcare Providers
- Ambulatory Surgical Centers
- Research & Academic Institutes
- Regulatory & Health Bodies: U.S. Food and Drug Administration (FDA) – device approval and safety standards, European Medicines Agency (EMA) – clinical and regulatory frameworks, National Institutes of Health (NIH) – respiratory research funding, World Health Organization (WHO) – global respiratory health initiatives
Databases
- ClinicalTrials.gov – clinical studies on respiratory monitoring devices and technologies
- World Health Organization (WHO) – global respiratory disease burden and health data
- Centers for Disease Control and Prevention (CDC) – respiratory disease statistics and surveillance data
- OECD Health Statistics – healthcare expenditure and medical device utilization data
Magazines
- Nature (News & Features section) – advancements in respiratory care technologies
- Science Magazine – innovations in medical devices and patient monitoring systems
- NIH Research Matters – research updates on respiratory diseases and monitoring tools
Journals
- American Journal of Respiratory and Critical Care Medicine
- Chest Journal
- Respiratory Medicine Journal
- European Respiratory Journal
Newspapers
- The New York Times (U.S.) – Covers healthcare technology adoption, hospital monitoring systems, and respiratory care trends
- The Wall Street Journal (U.S.) – Reports on medical device companies, FDA regulations, and market developments
- The Guardian (UK) – Covers public health issues such as COPD, COVID-19, and respiratory device usage
- Financial Times (UK) – Focuses on global healthcare investments, medtech innovation, and digital health integration
- The Hindu (India) – Reports on healthcare infrastructure, adoption of monitoring devices, and government health initiatives
- China Daily (China) – Covers healthcare reforms, hospital upgrades, and medical device adoption trends
- The Straits Times (Singapore) – Reports on healthcare innovation and respiratory care advancements in Asia
- The Sydney Morning Herald (Australia) – Covers hospital care, respiratory disease burden, and monitoring technologies
Associations
- American Thoracic Society (ATS)
- European Respiratory Society (ERS)
- American Association for Respiratory Care (AARC)
- International Society for Aerosols in Medicine (ISAM)
Public Domain Sources
- National Institutes of Health (NIH) – respiratory disease research and funding programs
- Centers for Disease Control and Prevention (CDC) – respiratory health data and surveillance
- European Commission – healthcare and medical device funding programs
- UK Research and Innovation (UKRI) – respiratory and digital health research initiatives
- World Health Organization (WHO) – global respiratory health policies and datasets
Proprietary Elements
- CMI Data Analytics Tool
- Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients