Global Zoladex Market Size and Forecast – 2026-2033
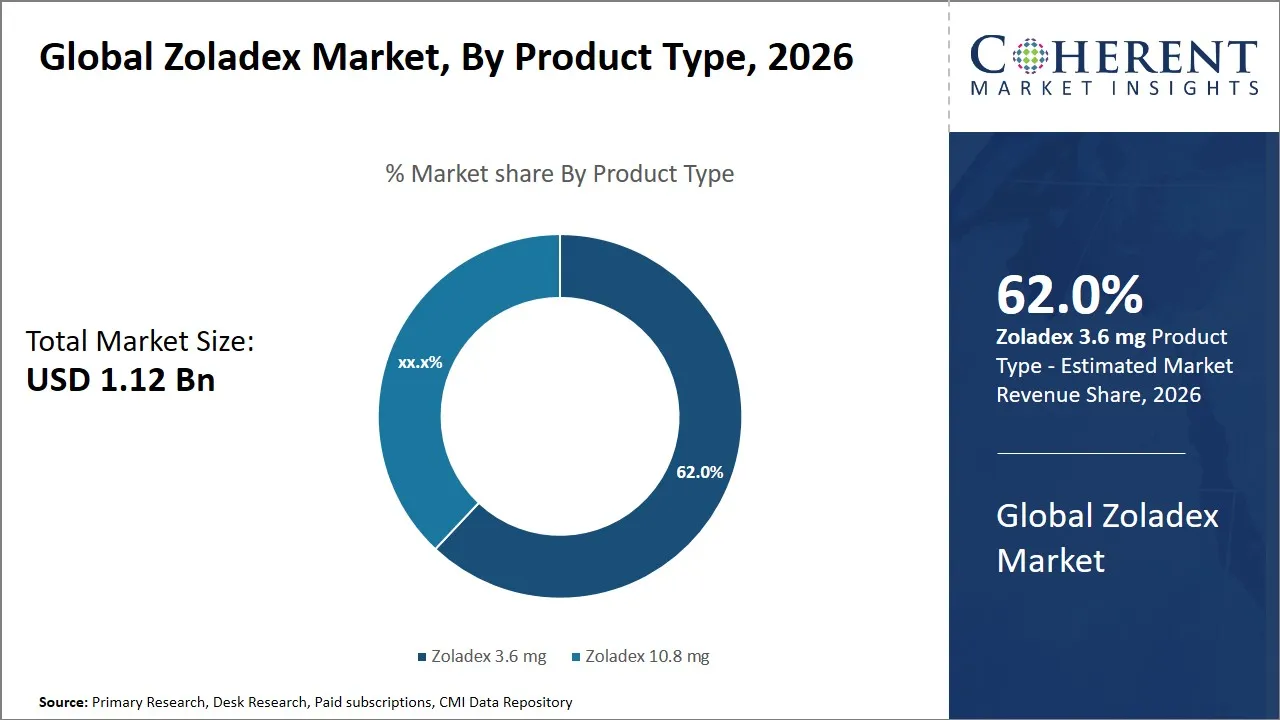
The global Zoladex market is estimated to be valued at USD 1.12 Bn in 2026 and is expected to reach USD 1.31 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 2.3% from 2026 to 2033. This steady growth reflects increasing demand for hormone therapy treatments and the expanding patient base for hormone-dependent cancers. The market growth is supported by advancements in drug delivery technologies and rising awareness among healthcare providers and patients.
Key Takeaways of the Global Zoladex Market
- Zoladex 3.6 mg segment is expected to lead the Zoladex market, capturing 62% share in 2026.
- Prostate cancer segment is estimated to represent 42% of the Zoladex market share in 2026.
- Hospitals segment is projected to dominate with 55% of the global Zoladex market share in 2026.
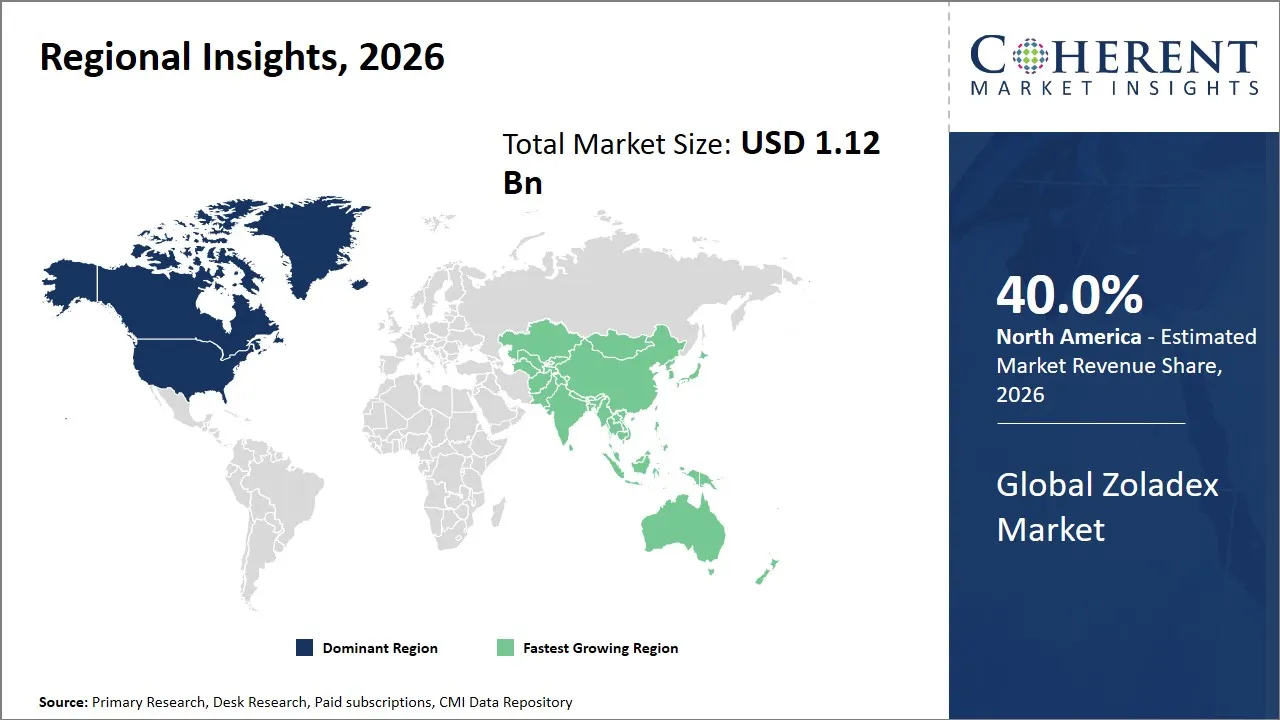
- North America is expected to lead the market, holding a share of 40% in 2026. Asia Pacific is anticipated to be the fastest-growing region, with 28% share in 2026.
Market Overview
- Zoladex (goserelin acetate) is widely used in hormone‑dependent cancers, especially prostate and breast cancer, and is increasingly leveraged in gynecological disorders (e.g., endometriosis, uterine fibroids). Its role beyond oncology supports incremental market expansion.
- Rising global incidence of prostate and breast cancers continues to be the primary growth engine for Zoladex, as it remains a standard hormone therapy choice in oncology care pathways across geographies. Early detection and screening improvements are expanding patient pools requiring long‑term endocrine management.
- Beyond oncology, broader clinical adoption for conditions such as endometriosis, uterine fibroids, and fertility preservation are adding new demand vectors, reducing reliance on core cancer applications and widening total addressable market.
- Trend toward longer‑acting depot injections (3‑month/extended release) and innovations in drug delivery systems are improving treatment adherence and convenience, which supports higher uptake and retention among patients and prescribers.
Currents Events and their Impact
|
Current Events |
Description and its Impact |
|
Patent Expiry & Generic Competition |
|
|
Regulatory & Reimbursement Policy Shifts |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Why Does the Zoladex 3.6 mg Segment Dominate the Global Zoladex Market in 2026?
The Zoladex 3.6 mg segment is expected to hold the largest share of 62% in 2026, owing to its balanced therapeutic efficacy and patient compliance. The dosage formulation is a good blend of efficacy, safety and dosing convenience and hence greatly improves patient compliance and overall treatment results. This is because one of the key factors that established Zoladex 3.6 mg as a dominant drug is the fact that it is a well-fit administration once in month and this fits the treatment guidelines of maintaining consistent hormone inhibition without placing too much burden on the treatment of patients. It is a popular monthly dosing routine among clinicians and patients and it makes treatment regimens less cumbersome and it reduces the need to visit hospitals frequently hence increasing compliance particularly in long-term therapy.
For instance, the National Comprehensive Cancer Network based in the U.S. updated its Breast Cancer Clinical Practice Guidelines (Version 1.2024), adding Zoladex 3.6 mg every 4 weeks and 10.8 mg every 12 weeks as recommended options for ovarian function suppression in premenopausal hormone‑receptor‑positive breast cancer treatment.
(Source- https://www.targetedonc.com/view/nccn-updates-breast-cancer-guidelines-to-include-goserelin)
Why Does the Prostate Cancer Segment Dominate the Global Zoladex Market in 2026?
The prostate cancer segment is expected to hold 42% share in 2026, driven by rising incidence rates and improved diagnostic technologies. The increased incidences of prostate cancer especially in the male population in most parts of the world as they age has increased pressure on effective hormone therapies that can help control the progression of disease and improve the survival chances of affected patients. The mechanism of action of Zoladex as a luteinizing hormone-releasing hormone (LHRH) agonist predisposes it as the cornerstone of androgen deprivation therapy (ADT) the standard mode of treatment of hormone-deprivate prostate cancer.
For instance, according to the American Cancer Society, prostate cancer is the second most frequently diagnosed cancer in men globally, trailing only lung cancer, with approximately 1.5 million new cases and 400,000 deaths annually as of 2022. It is the most common male cancer in 118 of 185 countries. While often indolent, it is the fifth leading cause of cancer death among men.
The Hospitals Segment Dominates the Global Zoladex Market
The hospitals segment is expected to hold the largest share of 55% in 2026, due to their extensive healthcare infrastructure and multidisciplinary treatment capabilities. These facilities act as the initial care provider in diagnosis, initiation of treatment and follow up of complex diseases, including prostate cancer, breast cancer, and other hormone sensitive diseases. Hospitals are a natural avenue of delivering Zoladex because of their ability to offer integrated care, such as oncology services, hormonal therapy and monitoring of the patient.
Regional Reimbursement and Payer Coverage Policies for Zoladex
- North America & Europe: Zoladex is generally covered by both the public and personal health insurance systems such as Medicare, Medicaid, and the national health systems. Usually oncology indications (prostate, breast cancer) and selected gynecological disorders are all covered, but co-pay assistance programs and formulary restrictions can affect access by the patient.
- Asia Pacific & Emerging Markets: The coverage is less uniform and also may be restricted to big hospitals or government cancer programs. National reimbursement of hormone therapy is provided in some countries (Japan, South Korea, Australia), other countries do this using out-of-pocket money or partial insurance, which has an impact on adoption and prices.
Regional Insights
To learn more about this report, Request Free Sample
North America Zoladex Market Analysis and Trends
The North America region is projected to lead the market with a 40% share in 2026, driven by a well-established healthcare infrastructure, early adoption of advanced therapies, and strong presence of pharmaceutical giants. The region enjoys a strong government coverage of cancer and hormones related disease treatment, high levels of awareness of patients and access to healthcare.
The main stakeholders, including the initial developer of Zoladex, that is AstraZeneca, Parker and AbbVie, are engaged in research, development, and marketing. This ecosystem helps to be innovative, conduct broad clinical trials, and fast regulatory approvals, so North America is not leaving the leading position in the Zoladex market.
For instance, the U.S. FDA revised the Zoladex prescribing information in late 2025, updating warnings and precautions related to cardiovascular risk, severe cutaneous reactions, and other safety considerations. This update reflects the agency’s ongoing post‑market safety monitoring for GnRH agonists including Zoladex 10.8 mg.
(Source- https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/019726s074,020515s011lbl.pdf)
Asia Pacific Zoladex Market Analysis and Trends
The Asia Pacific region is expected to exhibit the fastest growth in the Zoladex market contributing 28% share in 2026, propelled by increasing healthcare expenditure, rising incidence of hormone-dependent cancers, and expanding patient pool. The increasing demand is due to the improvement of healthcare infrastructure, increase in affordability due to government insurance plans and the increased awareness of specific therapies.
The presence of local and international pharmaceutical firms like AstraZeneca, Cipla which is based in India, and Hanmi Pharmaceutical based in South Korea is further fueling the market by establishing their presence with the help of regional and localized manufacturing as well as participation.
For instance, China’s National Medical Products Administration (NMPA) granted approval for Baituowei (Zoladex microspheres for injection) to be used in the treatment of breast cancer in premenopausal and perimenopausal women eligible for hormone therapy. This marks the first approved long‑acting microsphere formulation of goserelin in the world.
(Source- https://www.luye.cn/lvye_en/view.php?id=2204)
Zoladex Market Outlook for Key Countries
How is the U.S. Helping in the Growth of the Zoladex Market?
Zoladex is still a strategic market in the U.S. because of the high level of healthcare provision and wide range of specialized oncology centers. Zoladex has a long track record of innovation and the generation of clinical evidence, which is backed by the long-standing presence of AstraZeneca. Moreover, the extensive patient access is supported by the high health insurance cover and the government reimbursement policies. Its distribution channels are strong that guarantee the presence in the hospitals and clinics, keeping the country on the top level in North America.
How is Germany Helping in the Growth of the Zoladex Market?
The Zoladex market in Germany is typified with an extensive healthcare system and a high regulatory environment that guarantees high levels of drug efficacy and safety. High contributions of pharmaceutical giants like the European business of AstraZeneca and local giants like the Bayer, which act complementary in the distribution and promotion of hormone therapy drugs are made in this country. The strong reimbursement systems and emphasis on individualized medicine in Germany has facilitated the consistent adoption of Zoladex.
Key Drivers for the Growth of the Japan Zoladex Market
Japan is a country that is still leading in the Asia Pacific region and has already an aging population that requires hormone related treatment. The presence of well-established pharmaceutical firms including Takeshda and Daiichi Sankyo to join up with international companies in manufacture and marketing of Zoladex gives the Zoladex market strength. The Japanese government is highly supportive of innovation with extensive policies on health and significant investments on the research and development that lead to faster regulatory channels that are conducive to the market dynamics.
India Zoladex Market Trends
The Zoladex market in India is an ever-changing market due to the growing reporting of healthcare insurance and awareness regarding the use of hormone therapies in cancer therapy. The role of local companies such as Cipla and Sun Pharma to make accessibility more effective through generic drugs production and innovator products has played a significant role. The government strategies to simplify the drug approvals and promote foreign direct investments have led to increase in competitiveness in the market and improvement of the supply chain which has led to the growth of the market.
Impact of Regional Clinical Guidelines on Zoladex Use
|
Guideline |
Impact on Zoladex Use |
|
National Comprehensive Cancer Network (NCCN) |
Inclusion of goserelin 3.6 mg monthly and 10.8 mg 3‑monthly for ovarian function suppression (OFS) in premenopausal hormone receptor‑positive breast cancer (Category 2A). |
|
American Society of Clinical Oncology (ASCO) |
ASCO practice surveys and guidelines reflect that GnRH agonists (including goserelin) remain commonly used for OFS in high‑risk premenopausal hormone receptor‑positive breast cancer, with variable initiation and monitoring practices among clinicians. |
|
European Society for Medical Oncology (ESMO) |
ESMO breast cancer guidelines include OFS as part of adjuvant endocrine therapy strategies for premenopausal patients, influencing European clinical standards for goserelin use (e.g., integrated with tamoxifen/aromatase inhibitors). |
|
Local Protocols (Hospital/Research Settings) |
Local clinical trial protocols often require adherence to ASCO/CAP receptor testing standards and may use goserelin for ovarian suppression or hormone management based on guideline criteria. |
|
National/Regional Guidelines (e.g., NICE) |
National bodies (like NICE in the U.K.) recommend OFS as part of endocrine treatment for hormone receptor‑positive breast cancer when clinically appropriate, supporting goserelin use where indicated by evidence of added survival benefit. |
|
Consensus & Expert Panels (e.g., ESO‑ESMO/International Consensus) |
International consensus recommendations underpin guideline committees’ positions, emphasizing ovarian suppression (with LHRH agonists) as part of multimodal therapy in appropriate premenopausal patients. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
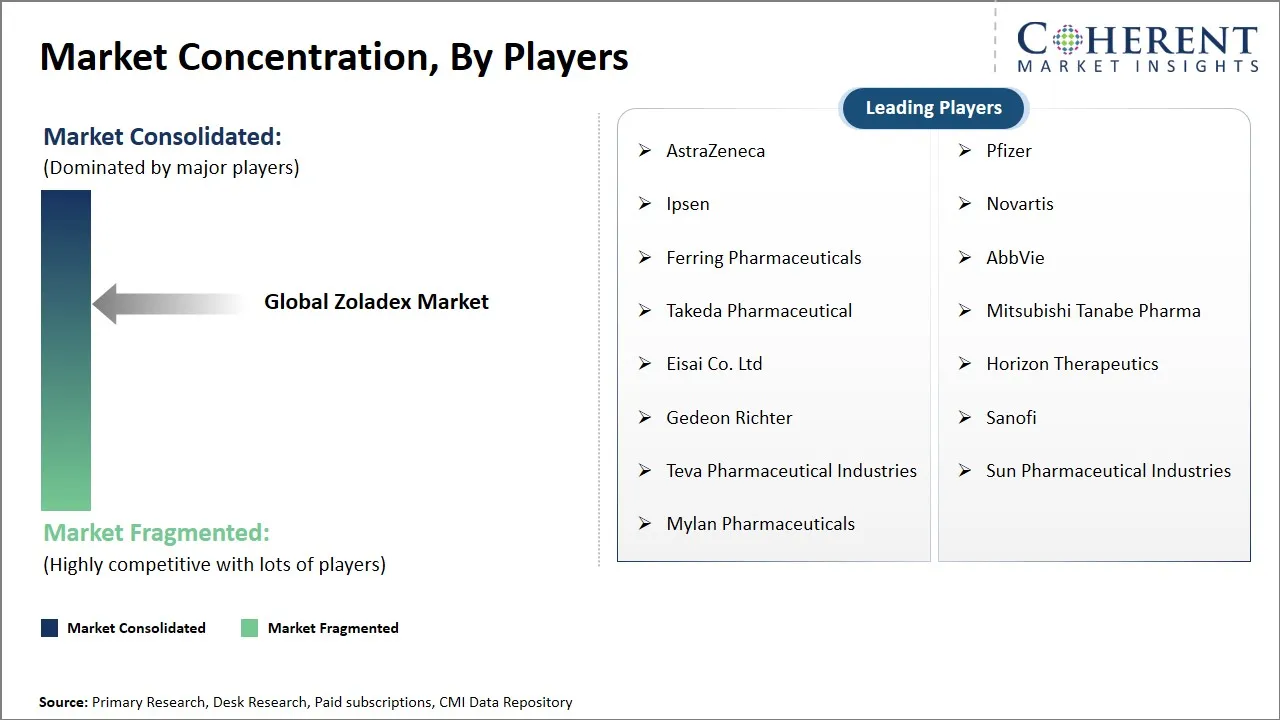
Market Players, Key Developments, and Competitive Intelligence
To learn more about this report, Request Free Sample
Key Developments
- On January 30, 2026, AstraZeneca announced a strategic collaboration with CSPC Pharmaceuticals that gives AstraZeneca exclusive global rights outside China to CSPC’s long‑acting injectable portfolio and preclinical programmes, supporting broader oncology pipeline development that complements hormonal therapies such as Zoladex.
- On August 25, 2025, GlaxoSmithKline Pharmaceuticals India entered oncology market with the launch of Jemperli and Zejula for advanced endometrial and ovarian cancer, further illustrating sector momentum in hormone‑responsive cancer treatments that influence market dynamics relevant to Zoladex.
- In June 2024, TerSera Canada Inc. received regulatory approval from Health Canada for a 12‑week dosing schedule (10.8 mg) of Zoladex LA (goserelin acetate) for high‑risk ER‑positive early and advanced breast cancer in pre‑ and perimenopausal women, enhancing convenience and adherence in oncology protocols.
Top Strategies Followed by Global Zoladex Market Players
|
Player Type |
Strategic Focus |
Examples |
|
Established Market Leaders |
Established Zoladex market leaders focus on R&D to improve formulations and drug delivery, form strategic partnerships with pharma companies and healthcare providers, and expand global distribution to capture emerging markets. |
AstraZeneca invests in developing long-acting Zoladex depots while partnering with oncology clinics worldwide to enhance market reach and patient access. |
|
Mid-Level Players |
Mid-level Zoladex players target price-sensitive markets by offering affordable yet reliable therapies, often in developing regions. They partner with CMOs and tech providers to boost production and access advanced formulations without heavy R&D costs, and expand reach through regional distribution agreements. |
Mylan Pharmaceuticals offers cost-effective Zoladex formulations in emerging markets through local manufacturing and distribution collaborations. |
|
Small-Scale Players |
Small-scale Zoladex players focus on niche, innovative formulations using advanced delivery technologies and bioengineering, often targeting specific patient groups or emerging indications. They expand reach through local partnerships with startups or regional manufacturers. |
SciGen Pharmaceuticals, an Indian biotech firm, develops specialized goserelin products for premenopausal breast and prostate cancer patients, collaborating with regional distributors and smaller pharma companies across Asia Pacific and Africa to expand market reach, share expertise, and increase visibility in targeted segments. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Report Scope
Zoladex Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1.12 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 2.3% | 2033 Value Projection: | USD 1.31 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AstraZeneca, Pfizer, Ipsen, Novartis, Ferring Pharmaceuticals, AbbVie, Takeda Pharmaceutical, Mitsubishi Tanabe Pharma, Eisai Co. Ltd, Horizon Therapeutics, Gedeon Richter, Sanofi, Teva Pharmaceutical Industries, Sun Pharmaceutical Industries, and Mylan Pharmaceuticals |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Zoladex Market Dynamics
To learn more about this report, Request Free Sample
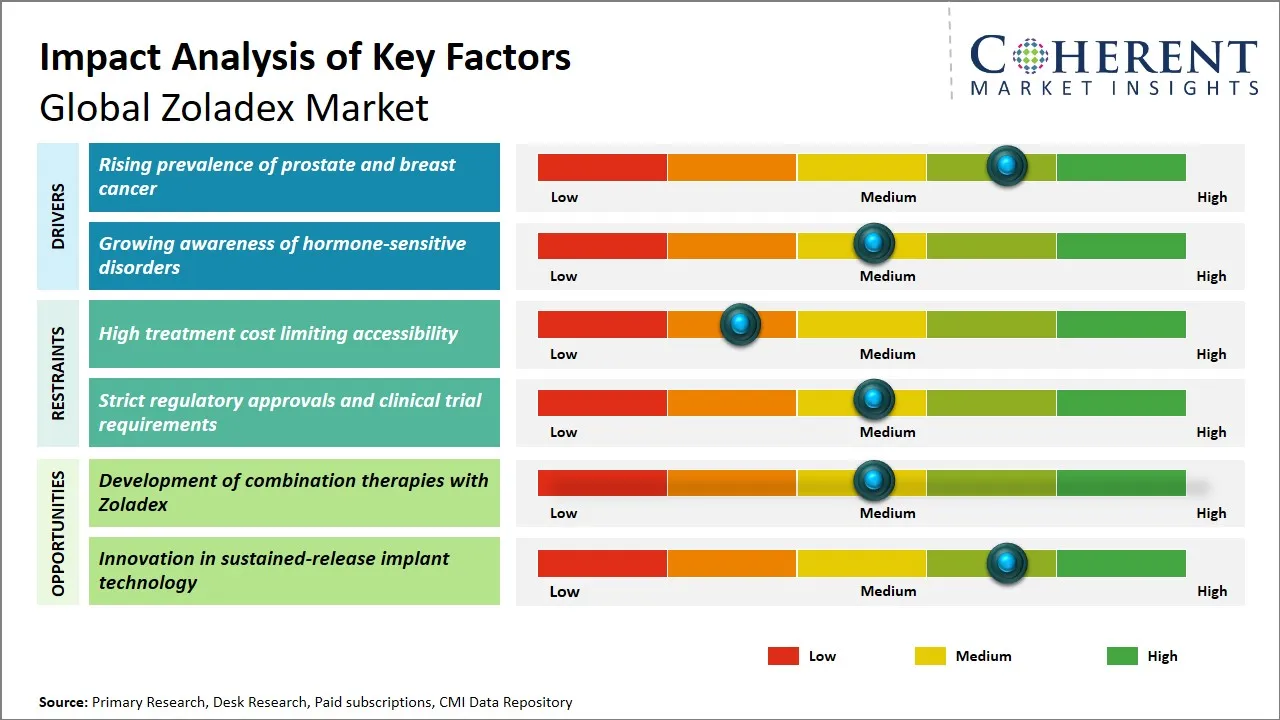
Zoladex Market Driver - Rising Prevalence of Prostate and Breast Cancer
The rising cases of prostate and breast cancer in the global world is a very strong force which is driving the demand of Zoladex. Being one of the most common ways of treating cancers that are sensitive to hormones, Zoladex is of great importance in the aspect of treatment of the same since it prevents the production of hormones that stimulate the growth of the tumors. The ageing population of the world and the increased ability to screen and detect diseases is creating more reported cases earlier on, especially in developed countries and in developing countries.
In addition to this, lifestyle and environmental factors are increasing the occurrence of these cancers and this is further enhancing the demand of good therapeutic alternatives such as Zoladex. The fact that the drug can be administered together with other therapies like chemotherapy or radiations further entrenches its role in the management protocols of cancer treatment.
For instance, in 2022, according to WHO, prostate cancer was the 4th most commonly diagnosed cancer overall and the 2nd most common in men worldwide, with approximately 1.47 million new cases and ~397,000 deaths. It is the leading cancer in 118 of 185 countries, with higher incidence rates in more developed regions and a projected rise to 2.4 million cases by 2040.
Zoladex Market Opportunity - Development of Combination Therapies with Zoladex
The world market of Zoladex offers a great growth opportunity due to the introduction of combination therapies that will incorporate Zoladex with other modes of treatment. Synergistic treatment regimens based on Zoladex (goserelin acetate), which is mainly utilized in hormone-responsive tumors like prostate and breast cancer, may provide an effective foundation of therapy that has the potential to increase the therapeutic efficacy and patient outcome.
New clinical studies indicate that Zoladex is likely to have improved disease control in providing a combination of Zoladex with novel targeted therapies, immunotherapies or chemotherapeutic agents to address multiple oncogenic pathways. This strategy is especially pertinent in the light of the increasing number of hormone-responsive cancers as well as the increased interest towards personalized medicine.
For instance, in controlled clinical study data, administering Zoladex (3.6 mg) with flutamide before and during radiation therapy significantly reduced local failure rates (16% vs. 33% at 4 years) and showed a trend toward fewer distant metastases compared with radiation alone.
(Source- https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/019726s074,020515s011lbl.pdf)
Analyst Opinion (Expert Opinion)
- Currently, the market of Zoladex is expanding as a result of increasing incidences of hormone sensitive cancer, the broader use of ovarian suppression treatment, as well as the development of drug delivery. Combination therapy, longer acting preparations and new markets have opportunities and challenges are pricing, variability of reimbursement and continued innovation is required.
- Medical meetings such as ASCO, ESMO, and the World Cancer Congress have contributed to the exchange of the knowledge and the emphasis on the new treatment strategies. Practical programs, including access programs of AstraZeneca and regional partnerships of Sciences Gen Pharmaceuticals in Asia Pacific and Africa, as well as new information provided by NCCN, ASCO, and ESMO guidelines, are still influencing clinical application and market development.
Market Segmentation
- Product Type Insights (Revenue, USD Bn, 2021 - 2033)
- Zoladex 3.6 mg
- Zoladex 10.8 mg
- Application / Indication Insights (Revenue, USD Bn, 2021 - 2033)
- Prostate Cancer
- Breast Cancer
- Endometriosis
- Precocious Puberty
- Other Hormone-Sensitive Disorders
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals
- Oncology Clinics
- Specialty Treatment Centers
- Research and Academic Institutes
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- AstraZeneca
- Pfizer
- Ipsen
- Novartis
- Ferring Pharmaceuticals
- AbbVie
- Takeda Pharmaceutical
- Mitsubishi Tanabe Pharma
- Eisai Co. Ltd
- Horizon Therapeutics
- Gedeon Richter
- Sanofi
- Teva Pharmaceutical Industries
- Sun Pharmaceutical Industries
- Mylan Pharmaceuticals
Sources
Primary Research Interviews
- Industry Stakeholders
- Oncology pharmacists
- Hospital formulary managers
- Pharmaceutical R&D directors
- Clinical trial investigators
- Biotech executives
- Regulatory affairs specialists
- End Users
- Oncologists
- Gynecologists
- Oncology nurses
- Endocrinologists
- Patients’ advocacy group representatives
- Clinical trial participants
Government and International Databases
- World Health Organization (WHO) – Global Health Observatory
- U.S. Food and Drug Administration (FDA) – Drugs@FDA
- European Medicines Agency (EMA) – Clinical Data & Reports
- National Cancer Institute (NCI) – SEER Database
- International Agency for Research on Cancer (IARC) – GLOBOCAN
- Centers for Disease Control and Prevention (CDC) – Cancer Statistics
Trade Publications
- The Lancet Oncology News
- Journal of Clinical Oncology News Updates
- Cancer Therapy Advisor
- Oncology Times
- MedPage Today – Oncology Section
- Targeted Oncology
Academic Journals
- The Lancet Oncology
- Journal of Clinical Oncology
- Cancer Research
- Breast Cancer Research and Treatment
- Prostate
- Endocrine-Related Cancer
Reputable Newspapers
- The New York Times – Health Section
- The Guardian – Science & Health
- The Washington Post – Health
- Financial Times – Healthcare
- The Wall Street Journal – Health & Medicine
Industry Associations
- American Society of Clinical Oncology (ASCO)
- European Society for Medical Oncology (ESMO)
- International Menopause Society (IMS)
- Prostate Cancer Foundation (PCF)
- Breast Cancer Research Foundation (BCRF)
- Endocrine Society
Public Domain Resources
- ClinicalTrials.gov
- WHO Reports and Fact Sheets
- National Institutes of Health (NIH) Publications
- OpenFDA Drug Labeling Data
- HealthData.gov
Proprietary Elements
- CMI Data Analytics Tool: Proprietary analytics tool to analyze real-time market trends, consumer behavior, and technology adoption in market
- Proprietary CMI Existing Repository of Information for Last 8 Years
Share
Share
About Author
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients