Agitation In Delirium Management Market is estimated to be valued at USD 271.7 Mn in 2026 and is expected to reach USD 377.3 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 4.8% from 2026 to 2033.
Analysts’ Views on Global Agitation in Delirium Management Market:
Hyperactive delirium is characterized by agitation, increased psychomotor activity, and a heightened level of arousal. It is the most recognizable type but accounts for less than 10% of delirium observed in the emergency department (ED). Hypoactive delirium is the most common type, accounting for approximately 90%. Thus, it offers the opportunity to various manufacturers to offer various drugs for the management of agitation delirium.
Figure 1. Global Agitation in Delirium Management Market Share (%), By Drug Type, 2026
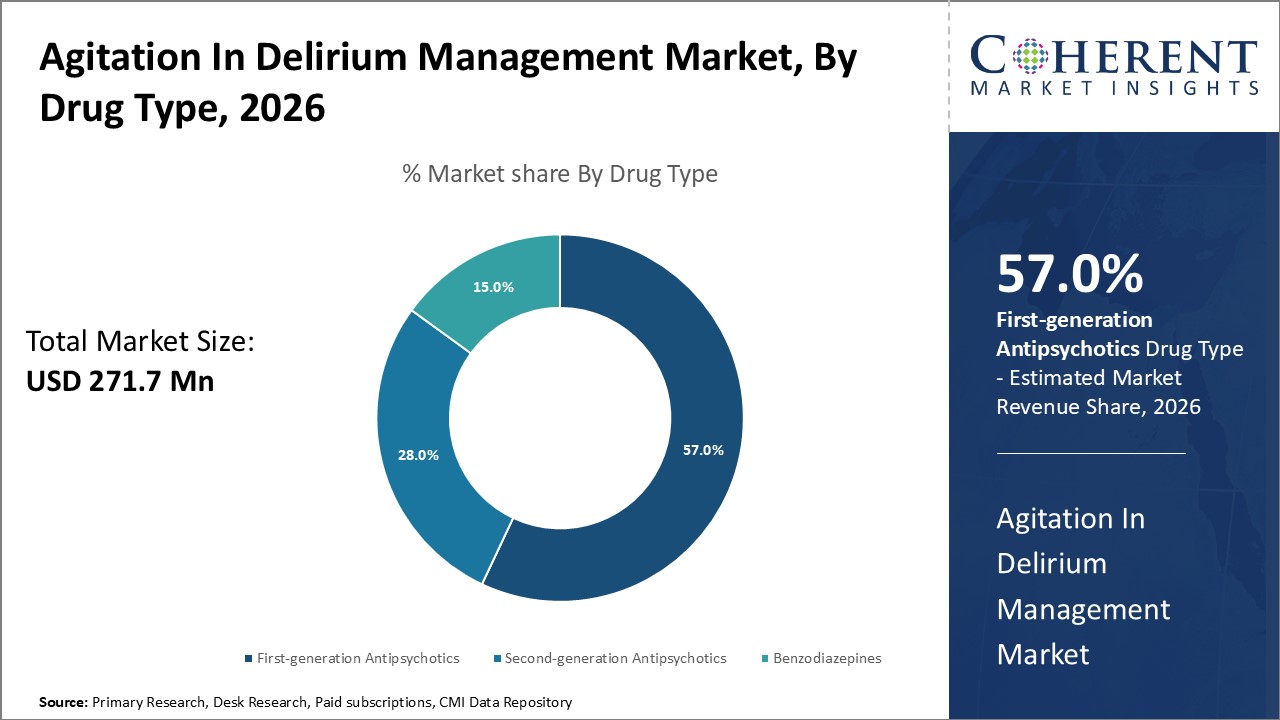
To learn more about this report, Request Free Sample
Global Agitation in Delirium Management Market– Drivers
Figure 2. Global Agitation in Delirium Management Market Share (%), By Region, 2026
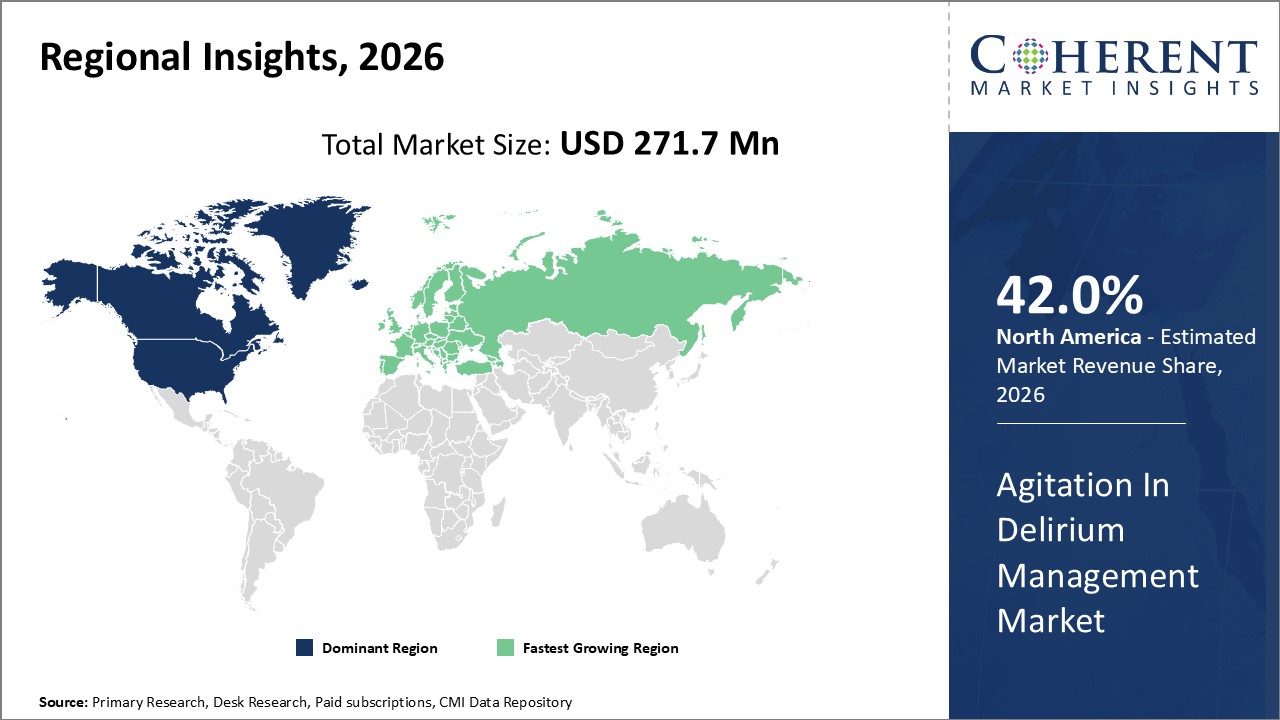
To learn more about this report, Request Free Sample
Global Agitation in Delirium Management Market - Regional Analysis
Among regions, North America is estimated to hold a dominant position in the global agitation in delirium management market over the forecast period. North America is estimated to hold 42.0% of the market share in 2026. North America agitation in delirium management market is expected to witness significant growth in the near future due to increasing healthcare expenditures in the region. The increasing prevalence of pulmonary embolism among patients is expected to drive the North America agitation in delirium management market growth over the forecast period. For instance, in U.S., the total health care expenditures associated with delirium are ranged from US$ 38 to US$152 billion each year, including costs associated with readmission, falls, and long-term care and the care costs of one patient with delirium increased their total care cost by about US$ 600 per day and US$ 18,000 over 30 days due to an increase in service use, including bed-related expenses, laboratory and diagnostic radiology costs, and pharmacy costs.
Global Agitation in Delirium Management Market - Segmentation
The global agitation in delirium management market is segmented into drug type, route of administration, distribution channel, and region.
Global Agitation in Delirium Management Market - Cross Sectional Analysis
Among drug type segments, first-generation antipsychotics are expected to drive the global agitation in delirium management market over the forecast period due to fewer extrapyramidal side effects. Greater awareness about delirium and availability of new formulations of antipsychotic drugs are boosting. For instance, several educational campaigns by medical associations and non-profit organizations in recent times have highlighted the need for prompt diagnosis and treatment of delirium. Additionally, the market has witnessed the launch of novel long-acting injectable and oral formulations of antipsychotics that claim better tolerability and ensure medication adherence in delirium patients. These advances in formulation and higher acceptability of pharmacological management indicate robust growth opportunities for players in this market over the coming decade.
Global Agitation in Delirium Management Market: Key Developments
In November 2021, British Geriatrics Society, a multidisciplinary professional organisation helping improve healthcare for older people. announced the launch of its Delirium Hub resource, which helps link users to useful, practical and high-quality information on all aspects of delirium in older people. The hub guides users through the topic of delirium, focusing on four key interlinked areas: an introduction to delirium presentation; screening and treatment of delirium in specific settings; education and training; and current research evidence.
Agitation In Delirium Management Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 271.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.8% | 2033 Value Projection: | USD 377.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Glenmark Pharmaceuticals Limited, Mylan N.V., Fresenius Kabi AG, Novartis AG, Akorn Incorporated, Zydus Cadila, Pfizer, Inc., BioXcel Therapeutics, Inc., and Takeda Pharmaceutical Company Limited |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Agitation in Delirium Management Market: Key Trends
Global Agitation in Delirium Management Market: Restraint
Global Agitation in Delirium Management Market - Key Players
Major players operating in the global agitation in delirium management market include Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Glenmark Pharmaceuticals Limited, Mylan N.V., Fresenius Kabi AG, Novartis AG, Akorn Incorporated, Zydus Cadila, Pfizer, Inc., BioXcel Therapeutics, Inc., and Takeda Pharmaceutical Company Limited
Global Agitation in Delirium Management Market
*Definition: Delirium refers to a state of mental confusion characterized by abrupt ups and downs in consciousness and attention. It is caused by underlying medical conditions such as infections, drug effects or withdrawal, and stress. Agitation often co-occurs with delirium and needs timely management to prevent self-harm and aid recovery. Growing awareness about delirium and agitation care protocols, along with favorable reimbursement for their management, is expected to drive the market during the forecast period.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients