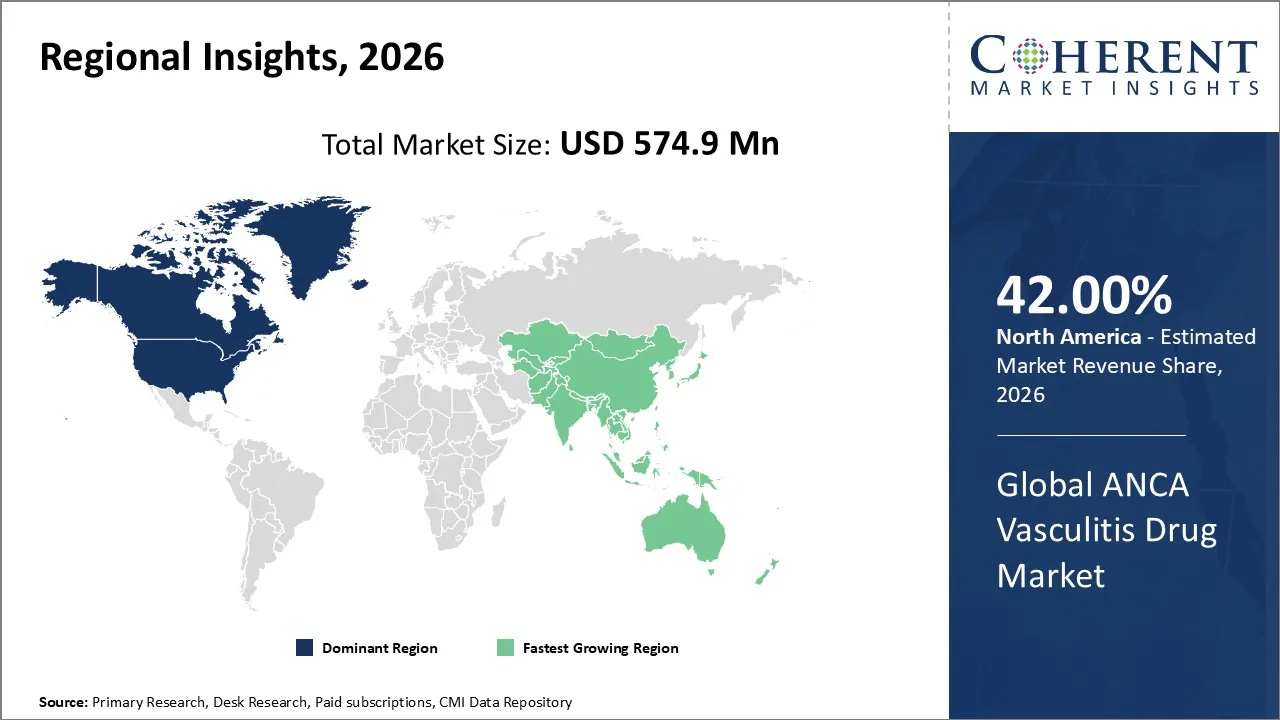
The ANCA vasculitis drug market is estimated to be valued at USD 574.9 Mn in 2026 and expected to reach USD 836.3 Mn by 2033, witnessing a CAGR of 5.5% over the forecast period (2026-2033). The ANCA vasculitis drug market in 2026 is growing due to rising prevalence, hospitals- based distribution, and more biologics are being used.
ANCA Vasculitis is a type of autoimmune disease that causes vasculitis, swelling or inflammation of blood vessels. ANCA stands for Anti-Neutrophilic Cytoplasmic Autoantibody. ANCA vasculitis is an autoimmune disease affecting small blood vessels in the body. Neutrophils are a particular subset of white blood cells that are the target of ANCAs, autoantibodies causing the disease. The ANCA vasculitis drug market is driven by rising prevalence of ANCA-associated vasculitis, increasing awareness, advancements in biologics, improved diagnostics, and growing demand for targeted therapies with better safety and efficacy profiles.
|
Current Event |
Description and its Impact |
|
Regulatory Framework Evolution and Drug Approval Processes |
|
|
Breakthrough Therapeutic Developments and Clinical Trial Advancements |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
|
Aspect |
Details (Numerical Data) |
Notes / Implications |
|
Incidence (Global) |
~20 cases per million annually worldwide |
Rare disease but a major contributor to kidney failure; guides trial site selection. |
|
Incidence (Europe) |
25–30 cases per million annually (highest reported in Northern Europe) |
Higher prevalence drives earlier drug launches and biosimilar adoption in Europe. |
|
Incidence (Asia) |
10–15 cases per million annually |
Lower prevalence but rising clinical trial activity in Japan, China, and South Korea. |
|
Prevalence (Global) |
~100–150 cases per million population |
Reflects chronic disease burden; important for healthcare resource allocation. |
|
Age Distribution |
Peak onset between 50–70 years; median age at diagnosis ~60 years |
Older patients have higher mortality and comorbidity; drug safety monitoring is critical. |
|
Gender Split (GPA) |
Male-to-female ratio ~1.2:1 |
Men more likely to present with renal involvement, influencing biologic therapy use. |
|
Gender Split (MPA) |
Nearly equal male-to-female ratio (1:1) |
Balanced distribution makes trial recruitment representative; symptom variability complicates endpoints. |
|
Clinical Impact |
Up to 30–40% of AAV patients develop kidney failure within 5 years if untreated |
Emphasizes need for effective therapies like rituximab and avacopan to reduce dialysis burden. |
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of drug type, the rituximab segment is expected to hold 37.8% share in 2026. It is widely regarded as the primary treatment for GPA and MPA, replacing cyclophosphamide in numerous treatment regimens. It is the most important part of treating ANCA vasculitis around the world due to its effectiveness has been approved by regulators, and is given through an infusion.
For instance, in June 2025, the new UK treatment guidelines for ANCA-associated vasculitis declare that Rituximab is better than cyclophosphamide, especially for relapsing cases. Doctors recommend rituximab for both induction and maintenance therapy, which usually lasts two to four years. This shows that Rituximab is still the best drug for ANCA vasculitis, especially for people with GPA and MPA.
In terms of type, the Granulomatosis with Polyangiitis (GPA) segment is expected to lead the market with 45% share in 2026, because it is increasingly prevalent than other types of ANCA vasculitis, there is a high demand for biologics like rituximab. Clinical guidelines put GPA treatment at the top of the list, which makes it easy for hospital-based infusion centers to adopt and keep making capital.
For instance, in February 2026, The FDA is conducting an investigation into Amgen's drug Tavneos (avacopan), which is approved for GPA and MPA, because of concerns about the safety and integrity of the trials. Amgen defends its benefit-risk profile.
In terms of distribution channel, the hospital pharmacies segment is projected to account for 52% share in 2026. Biologics like rituximab need to be given through an IV while a physician supervises, which keeps hospital sales strong. Centralized procurement, specialist oversight, and patient monitoring make this channel even stronger. Retail and online pharmacies are still not as important for oral therapies like glucocorticoids and avacopan.
To learn more about this report, Request Free Sample
North America is expected to dominate the ANCA vasculitis drug market with 42% share in 2026, owing to the advanced healthcare system, strong regulatory approvals, and widespread use of biologics. The region has the biggest market share because it has major pharmaceutical companies, ongoing clinical trials, and good reimbursement policies that make it easier for patients to get care.
In April 2025, the U.S. Patent Office granted approval for AZD1656, Conduit Pharmaceuticals' primary drug candidate aimed at autoimmune disorders such as ANCA vasculitis. This patent bolsters intellectual property safeguards, paving the way for clinical trials and potential collaborations. This achievement places Conduit in a favorable position to advance new therapies for autoimmune diseases, ultimately expanding treatment choices for patients and creating additional business prospects within the United States.
Asia Pacific is expected to exhibit the fastest growth, since greater resources is being invested in healthcare, diagnostic tools are getting better, and patients are becoming more aware. Countries like China, India, and Japan are using biologics and precision medicine, which is driving growth and making the region the fastest-growing market in the world.
For instance, in June 2025, Ono Pharmaceutical and Vertex worked together to make povetacicept, a BAFF/APRIL antagonist for
The ANCA vasculitis drug market in the US is highly competitive in 2026 because of the country's advanced healthcare system, strong FDA approvals, and widespread use of biologics. The US is a major market due to it has major pharmaceutical companies, ongoing clinical trials, and good reimbursement policies that make it easier for patients to get care.
For instance, in June 2025, Fate Therapeutics indicated that it had new clinical data for FT819, its ready-made CAR T-cell therapy. Lupus patients had long-lasting drug-free remissions and good safety outcomes. The FDA has let the program grow to include more autoimmune diseases, such as ANCA vasculitis. This shows how FT819 could change the way people in the US treat these diseases.
In 2026, the ANCA vasculitis drug market is exceptionally busy in China because the country's healthcare infrastructure is growing quickly, more money is being put into advanced therapies, and more individuals are growing awareness about autoimmune diseases. China is a key growth engine in Asia-Pacific due to its expanding diagnostic capabilities and the use of biologics.
For instance, in March 2025, Henlius received approval in China for a Phase 2 trial of HLX79 and HANLIKANG (rituximab) together in patients with active glomerulonephritis. The study encompasses secondary forms, including ANCA-associated vasculitis. HLX79 seeks to improve B-cell depletion, presenting a prospective therapeutic advancement for patients exhibiting insufficient response to rituximab monotherapy.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 574.9 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.5% | 2033 Value Projection: | USD 836.3 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
ChemoCentryx, Inc., Vifor Pharma, GlaxoSmithKline plc, Amgen, Genentech, Inc., Pfizer Inc., Biogen, InflaRx GmbH, and AstraZeneca |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Increasingly regulatory bodies around the world are granting approval to ANCA vasculitis drugs. This shows that people are becoming more confident in their safety and effectiveness. These approvals not only give patients more treatment options, but they also encourage drug companies to invest more money into this area of medicine. As more drugs become available, they become easier to obtain, which leads to more patients using them and doctors prescribing them. This trend greatly helps the ANCA vasculitis drug market growth because regulatory support speeds up the process of bringing new drugs to market and makes the market more competitive for new treatments.
Major pharmaceutical companies are investing more money into research and development to make better treatments for ANCA vasculitis. Researchers are seeking into new methods, such as biologics and targeted treatments, to help patients get better and have fewer side effects. These kinds of research and development projects add to the list of possible drugs, making sure that new ideas keep coming into the market. This increase in activity directly raises the ANCA Vasculitis drugs market demand, as both patients and healthcare providers are interested in better and more personalized ways to treat the disease.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients