The Asia Pacific Endoscopy Devices Market is anticipated to grow at a CAGR of 7.3%, with an estimated market value of USD 6.55 Bn in 2026, and is expected to reach USD 10.75 Bn by 2033. UN ESCAP projects that by 2050, one in four people in Asia Pacific will be aged 60 years or above, equivalent to around 1.3 billion people, which is expected to significantly increase demand for endoscopy procedures used in the diagnosis and management of age-related gastrointestinal, oncological, and chronic conditions.
There is tremendous growth potential for visualization systems because all of the markets for diagnostic and therapeutic endoscopy rely on image processors, light sources, scopes, and monitors to find lesions and guide procedures. Japan alone has recorded 14.3 million gastrointestinal endoscopy cases that are insured, and the National Cancer Center Hospital in Japan has 15 endoscopy rooms; these numbers demonstrate the need for more sophisticated visualization platforms.
There are two key factors driving this segment's growth. The first is the rising number of endoscopic screenings and gastrointestinal cancers, which increases the need for brighter, sharper, and more accurate images used in diagnosis. The second is the replacement of old systems with new 4K capable, image-enhanced systems that help hospitals increase the speed and efficiency of workflows and improve therapeutic decision-making through a broader range of compatible diagnostic and interventional endoscopes used across multiple departments.
On April 23, 2025, FUJIFILM India introduced its ELUXEO 8000 Therapeutic Endoscopy Solution featuring a CMOS sensor and 4K output at the 22nd live endoscopy conference in Mumbai, while on April 23, 2020, Olympus launched its EVIS X1 advanced visualization systems for gastrointestinal disorders (stomach, colon, esophagus, and bronchus).
To learn more about this report, Request Free Sample
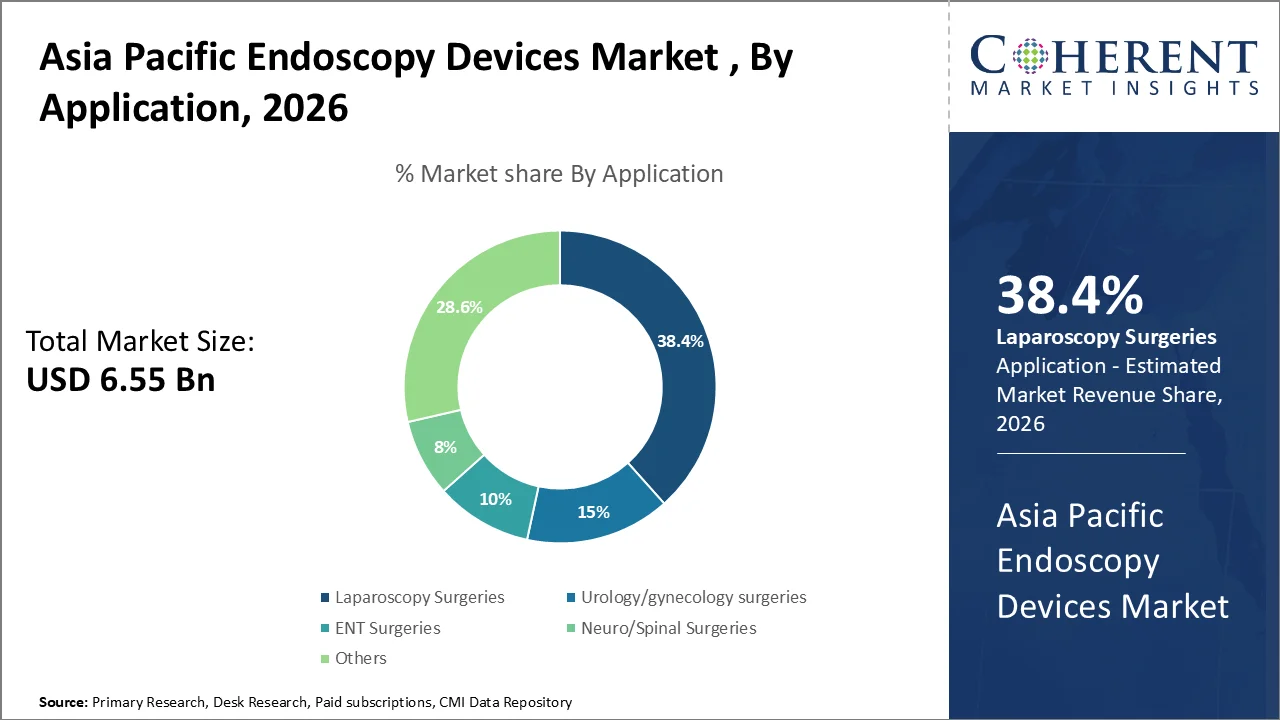
The Laparoscopy Surgeries dominates with 38.4% of market share in 2026, due to the increasing reliance on endoscopes for visualization, accuracy, and quicker recovery across the surgical specialties of minimally invasive abdominal, gynecologic, thoracic and urologic procedures. A 2025 national study from Japan reported 34,429 laparoscopic hysterectomies in 2023, accounting for 53.4% of all hysterectomies for benign diseases, while robot-assisted hysterectomies reached 8,088 cases, reflecting strong adoption of advanced laparoscopic surgery in the region.
Two key drivers for this segment's growth. First, laparoscopic surgery is preferred as it is associated with less pain, fewer wound infections, less blood loss, and shorter length of stay than open surgeries; second, strong registry coverage and structured training for surgeons in Japan is allowing surgeons to achieve standardized outcomes and to adopt minimally invasive surgery across many different specialized areas of practice.
On September 14th 2022 Olympus introduced their VISERA ELITE III surgical video imaging platform covering parts of Asia, Australia, and Japan, which was designed for laparoscopic diagnosis, treatment, and video observation; subsequent updates on March 11th 2026 will emphasize on the capabilities of the platform's 4K white-light/fluorescent-guided surgery features.
The endoscopy devices marketplace throughout the Asia Pacific region will see a significant transformation in 2026 with the advent of cloud-based, AI-assisted endoscopy ecosystems and the diminishing use of stand-alone hardware. Olympus recently confirmed via a press release dated February 2026 that its CADDIE CADe cloud-based solution demonstrated a 7.3% increase in adenoma detection rates, as well as a increase in the detection of sessile serrated lesions when utilized within the framework of the EAGLE randomized clinical trial, which evaluated CADDIE CADe at 8 sites, on 841 patients and over the course of participation by 22 endoscopists. Additionally, the world's first joint demonstration of endoscopy performed in the cloud has been completed between NTT and Olympus, thereby adding credibility to the emerging trend of real-time image processing in endoscopic procedures, rapid upgrade cycles for cloud-based systems and remote collaborative diagnostic capabilities.
|
Current Events |
Description and its Impact |
|
China tightens medical-device manufacturing compliance |
|
|
India strengthens and digitizes device regulation |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Endoscopy devices have the highest growth potential for the Asia-Pacific Region, because of a large number of diseases, more hospitals, and an increasing number of minimally invasive surgeries. In 2022, almost 49.2% of all new cancer cases occurred in the Asia-Pacific Region; and of the cancer deaths 56.1%. Japan, China and India are each increasing their surgical capacity, screening programs and medical device regulations. As such, the Region can be viewed as favorable for suppliers of visualization systems (endoscope), therapeutic endoscopic devices, and laparoscopic devices.
With its established infrastructures for both surgical procedures and clinical research (the Japan Gastrointestinal Endoscopy Database reports approximately 14.3 million GI endoscopic procedures each year), as well as several other factors, Japan has the highest level of maturity in the Asia Pacific region. Evidence from various studies shows an increase in the percentage of patients that undergo endoscopic resection for malignant gastric tumors, increased from 47% at the start of the study period to 57% by the end of the review period. This indicates that Japan continues to lead the region in terms of adoption of advanced endoscopic technologies.
On the other hand, China offers significant growth in the future due to the increase in hospital size, new hospitals opening and more generally because of the steep rise in the number of patients being diagnosed with gastrointestinal problems. According to national reviews of digestive endoscopic procedures, the number of procedures performed rose from 28.8 million in 2009 to 44.5 million in 2027, while the number of endoscopists rose from 26,203 to 39,638 in the same period. A more recent review has estimated that there will be 43.3 million procedures performed in China in the year 2023. The economies of scale of this growth make China a high priority market for both visualization systems and therapeutic endoscopy platforms.
Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.55 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.3% | 2033 Value Projection: | USD 10.75 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Accellent Inc., Aesculap Inc., Arthrex Inco, Boston Scientific Corporation, 3NT Medical, AnX Robotica Corporation, C.R. BARD INC, Conmed Corporation, Olympus Corporation, KARL STORZ Endoscopy-America, Inc., Cook Medical Incorporated, and Fujifilm Holding Corporation |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Major companies: Accellent Inc., Aesculap Inc., Arthrex Inco, Boston Scientific Corporation, 3NT Medical, AnX Robotica Corporation, C.R. BARD INC, Conmed Corporation, Olympus Corporation, KARL STORZ Endoscopy-America, Inc., Cook Medical Incorporated, and Fujifilm Holding Corporation
The Asia Pacific endoscopy devices market will continue to be driven by demographic pressure. According to the UN Economic and Social Commission for Asia and the Pacific (ESCAP), the projected amount of people aged 60 years or older in APAC will increase to one in four people in 2050, which is about 1.3 billion people, while the International Agency for Research on Cancer (IARC) states that the most common cancers causing people in Asia are lung cancer, liver cancer, and stomach cancer. The expected growth in demand for both upper GI and colorectal endoscopic procedures will lead to recurring orders of visualization systems, therapeutic scopes and service contracts associated with them.
Share
Share
Komal Dighe is a Management Consultant with over 8 years of experience in market research and consulting. She excels in managing and delivering high-quality insights and solutions in Health-tech Consulting reports. Her expertise encompasses conducting both primary and secondary research, effectively addressing client requirements, and excelling in market estimation and forecast. Her comprehensive approach ensures that clients receive thorough and accurate analyses, enabling them to make informed decisions and capitalize on market opportunities.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients