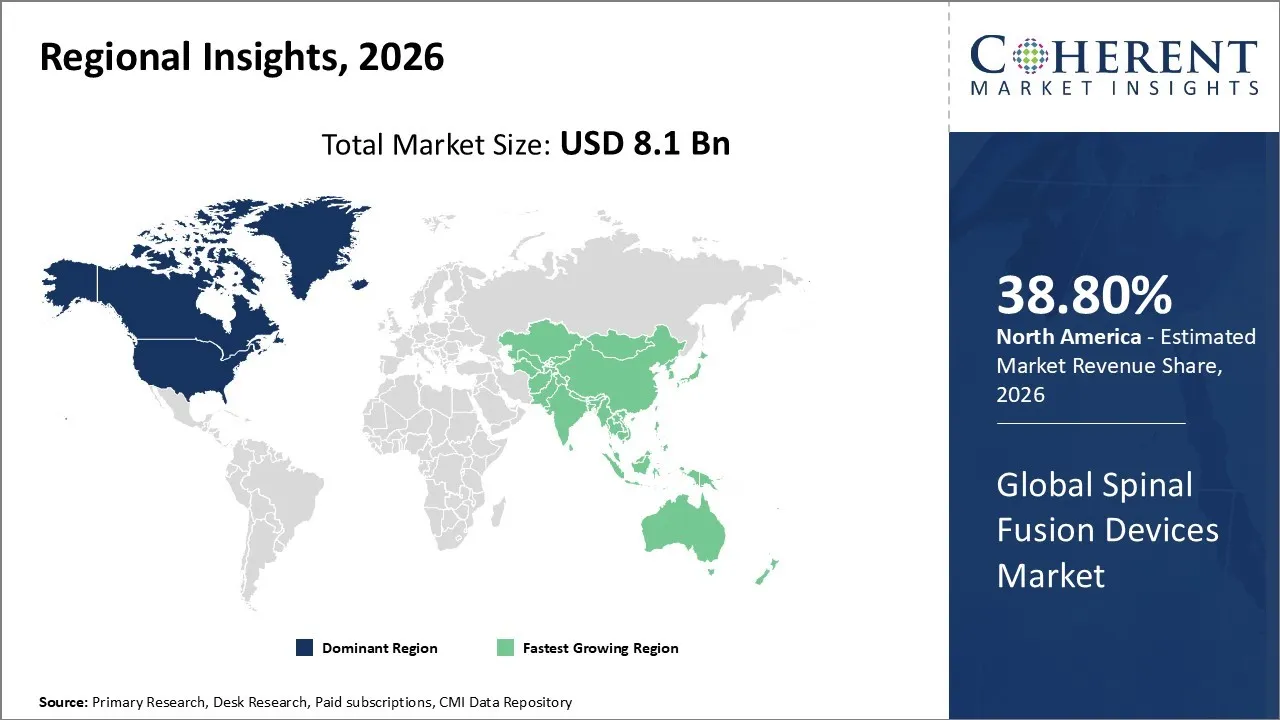
The Spinal Fusion Devices Market is estimated to be valued at USD 8.1 Bn in 2026 and is expected to reach USD 10.54 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.9% from 2026 to 2033.
The Spinal Fusion Devices Market delivers medical devices that stabilize and fuse vertebrae in patients with spinal disorders, including degenerative conditions, trauma, deformities, and herniated discs. It includes implants such as spinal plates, interbody cages, screws, and rods, along with supporting surgical instruments. Rising spinal conditions, innovations in minimally invasive and robotic-assisted surgeries, growing patient awareness, and expanding healthcare infrastructure drive market growth. Hospitals and specialized spine centers actively utilize these devices as primary end-users globally.
|
Current Events |
Description and its impact |
|
Macroeconomic Events |
|
|
Technological Advancements |
|
|
Geopolitical Developments |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Reimbursement plays a critical role in the adoption and commercialization of spinal fusion devices, particularly advanced and personalized implant technologies. Favorable reimbursement policies directly influence hospital purchasing decisions, surgeon adoption, and patient access to innovative treatments.
A strong example of this impact is Carlsmed’s Aprevo personalized interbody implants, which received New Technology Add-On Payment (NTAP) approval from the Centers for Medicare & Medicaid Services (CMS) in August 2025. The reimbursement was granted for cervical fusion procedures using Aprevo implants under CMS’s Hospital Inpatient Prospective Payment System (IPPS) Final Rule for FY 2026.
NTAP is designed to support hospitals adopting new medical technologies during their early market introduction by providing additional reimbursement beyond standard DRG payments. Beginning October 1, hospitals performing cervical fusion procedures with Aprevo devices are eligible for up to $21,125 in additional reimbursement per qualifying procedure from CMS and private payors. This reduces financial barriers for hospitals and encourages the use of innovative, patient-specific technologies.
Carlsmed’s Aprevo system leverages an AI-powered surgical planning platform and digital manufacturing process to create implants tailored to each patient’s anatomy and spinal alignment. The company’s technology has received FDA Breakthrough Device designation for both lumbar and cervical interbody fusion devices, with FDA clearance already in place for lumbar fusion procedures across anterior, lateral, and transforaminal approaches.
Cervical Spine expected to hold the largest market share of 45.6% in 2026. The cervical spine segment in the Spinal Fusion Devices Market grows as rising numbers of patients experience neck disorders, including cervical spondylosis, herniated discs, and spinal stenosis, necessitating surgical treatment. Surgeons enhance outcomes and speed recovery by employing minimally invasive and precision-guided techniques, which drives broader adoption. Hospitals and specialized spine centers increasingly perform cervical fusion procedures, while higher patient awareness and expanded healthcare infrastructure actively support the widespread use of advanced cervical spine solutions worldwide. For instance, in December 2025, Carlsmed unveiled Aprevo platform for cervical fusion surgeries, expanding its personalized spine solutions with AI-driven planning and 3D-printed, patient-specific implants.
Spinal Plating Systems hold the largest market share of 43.3% in 2026. The Spinal Fusion Devices Market drives demand for spinal plating systems as more patients experience spinal injuries, degenerative disorders, and deformities that need surgical stabilization. Surgeons improve fusion success and outcomes using innovative implant materials and designs, including titanium and biocompatible alloys. Minimally invasive techniques and advanced navigation technologies allow precise and safer plate placement. Hospitals and specialized spine centers expand their use of these systems, while increasing patient awareness actively encourages broader adoption of spinal plating solutions globally. For instance, in June 2025, Johnson & Johnson MedTech launched the VOLT™ Distal Radius and VOLT™ Proximal Humerus 3.5 Plating Systems to improve treatment for two common fractures.
Hospitals acquired the prominent market share of 44.8% in 2026. Hospitals actively fuel the Spinal Fusion Devices Market by performing complex spinal surgeries that rely on advanced infrastructure, skilled surgical teams, and comprehensive postoperative care. They respond to the growing number of spinal disorders and trauma cases by adopting modern fusion devices and minimally invasive technologies. By expanding specialized spine units, utilizing state-of-the-art surgical tools, and engaging increasingly aware patients, hospitals drive the widespread implementation and use of spinal fusion systems across healthcare settings worldwide. For instance, in October 2025, Hospital for Special Surgery collaborated with General Atlantic to launch a national network of independently operated ambulatory surgery centers providing advanced orthopedic and spine outpatient care.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 38.80% in 2026. The North America Spinal Fusion Devices Market grows as more patients seek advanced spinal treatments and experience degenerative spinal disorders. Hospitals and specialized spine centers actively implement minimally invasive and robotic-assisted surgical technologies to enhance outcomes and shorten recovery times. Surgeons use innovative implant materials, such as titanium and biocompatible alloys, to improve surgical success. Expanding healthcare infrastructure, rising patient awareness, and strong reimbursement systems drive broader adoption of spinal fusion devices, establishing North America as a leading, technologically advanced market worldwide. For instance, in December 2025, Spinal Simplicity received FDA 510(k) clearance for the Minuteman G6 MIS Fusion Device, a next-generation platform that enhances procedural efficiency, tissue-sparing access, and segmental stabilization.
The Asia Pacific Spinal Fusion Devices Market grows as more patients experience spinal disorders, trauma, and degenerative conditions that require surgical treatment. Hospitals and emerging spine centers actively adopt modern fusion technologies, including minimally invasive systems and advanced implant designs, to improve patient outcomes. Expanding healthcare infrastructure, a larger pool of skilled surgeons, and increasing patient awareness drive market adoption. Governments and medical tourism initiatives further promote spinal care, establishing Asia Pacific as a rapidly developing and high-potential market.
The United States Spinal Fusion Devices Market grows as more patients experience spinal disorders, including degenerative conditions and trauma-related injuries. Hospitals and specialized spine centers actively implement advanced surgical technologies, such as minimally invasive procedures, robotic-assisted systems, and image-guided navigation, to improve precision and patient outcomes. Surgeons enhance fusion success with innovations in implant materials and designs, while robust healthcare infrastructure, widespread insurance coverage, and rising patient awareness drive broader adoption, establishing the U.S. as a leading and highly competitive market worldwide. For instance, in September 2025, Aurora Spine launched its DEXA-L™ Anterior Lumbar Interbody Fusion Device, part of its patented DEXA™ Technology Platform featuring the world’s first bone density–matched implants.
The India Spinal Fusion Devices Market grows as more patients experience spinal disorders, trauma, and degenerative conditions that require surgical intervention. Hospitals and emerging spine centers actively implement modern fusion technologies, including minimally invasive systems and advanced implant designs, to improve patient outcomes. Expanding healthcare infrastructure, a larger pool of skilled surgeons, and rising patient awareness drive broader adoption. Government initiatives, the growth of medical tourism, and increasing demand for quality spinal care establish India as a rapidly emerging and high-potential market in the region.
The market increasingly favors minimally invasive spinal fusion procedures, driven by patient demand for reduced pain, smaller incisions, and faster recovery. Hospitals and specialized spine centers adopt MIS-compatible implants and advanced surgical tools, while surgeons leverage navigation and imaging technologies for precision. This trend encourages device manufacturers to innovate low-profile plates, modular screws, and expandable cages, enhancing procedural efficiency and improving outcomes, positioning minimally invasive techniques as a central focus in current spinal fusion practices.
Innovations in materials, design, and customization are shaping the market. Surgeons now use titanium, PEEK, and biocompatible alloy implants with porous coatings for better fusion and bone integration. Customizable and patient-specific implants, often 3D-printed, address anatomical variability and complex spinal cases. These advancements improve surgical accuracy, reduce complications, and expand the range of treatable conditions, driving demand for cutting-edge fusion systems that integrate seamlessly with modern surgical workflows.
The rising preference for minimally invasive spinal fusion and outpatient surgeries creates significant opportunities for device manufacturers. Hospitals seek implants compatible with smaller incisions and shorter operative times to improve patient satisfaction. Companies can develop low-profile plates, modular screws, and expandable cages tailored to these procedures, while providing training and support for surgeons. This trend allows healthcare providers to offer faster recovery options, reduce hospital stays, and expand the adoption of advanced spinal fusion systems.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8.1 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.9% | 2033 Value Projection: | USD 10.54 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Zimmer Biomet Holdings, Inc., Stryker Corporation, Inc., Exactech, Inc., Orthofix International N.V., Globus Medical, Inc., NuVasive, Inc., Medtronic Plc., Depuy Synthes, B. Braun ,Melsungen AG, Wenzel Spine, Inc., K2M Group Holdings, Inc., Centinel Spine, Inc., and Spineart Geneva SA. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients