Generic oncology sterile injectables are biologics that have the same active ingredients as that of the branded version, with only the inactive contents of the drugs being different. These generic oncology sterile injectables are used for the treatment of various types of cancer including breast, prostate, colorectal, melanoma, lung, and bronchus. These injectables are available at low prices and therefore, are widely preferred over branded drugs. Furthermore, R&D for generic drugs require less capital, which increases competition to enter into the generic oncology sterile injectable market as soon as the patent for branded injectable expires.
Asia Pacific Generic Oncology Sterile Injectable Market - Impact of the Coronavirus (COVID-19) Pandemic
The coronavirus (COVID-19) outbreak was first reported on December 31, 2019, in Wuhan, China. The World Health Organization declared COVID-19 as pandemic on March 11, 2020. According to the Coronavirus (COVID-19) Weekly Epidemiological Update by the World Health Organization, over 256.4 million cases and 5.1 million deaths due to coronavirus (COVID-19) were reported till November 21, 2021, across the globe.
Impact of COVID-19 on Demand and Supply of Generic Oncology Sterile Injectable:
The COVID-19 pandemic and lockdown in various countries across the globe have impacted the financial status of businesses across all sectors including private healthcare sector. The COVID-19 pandemic has impacted the entire supply chain of the healthcare industry mainly due to strict lockdown in several regions. The COVID-19 pandemic has affected the economy of various regions across the globe in three main ways; 1) by directly affecting the production and demand; 2) by creating disruptions in distribution channels; and 3) through its financial impact on companies and financial markets. Several countries such as Thailand, Indonesia, and Singapore are facing problems with regards to transportation and distribution of healthcare products.
Thus, impact of the coronavirus (COVID-19) pandemic is expected to limit the growth of the Asia Pacific generic oncology sterile injectable market during the forecast period, owing to decrease in supply of raw materials for manufacturing oncology drugs in Asia Pacific market.
The Asia Pacific generic oncology sterile injectable market is estimated to be valued at US$ 4,055.3 Mn in 2021 and is expected to exhibit a CAGR of 12.2% over the forecast period (2021-2028).
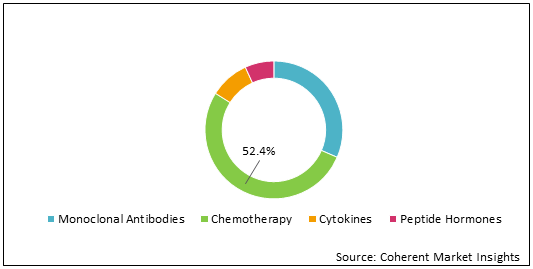
Figure 1: Asia Pacific Generic Oncology Sterile Injectable Market Share (%) Analysis, By DrugType, 2021
To learn more about this report, Request Free Sample
Patent expiry of biologics is a major factor that is expected to drive the market growth over the forecast period
Generic pharmaceuticals market is expected to witness significant traction, owing to large number of patent expiry of branded drugs. As a patent expires, various manufacturers anticipate to gain profit by selling the generic version of those drugs at significantly reduced prices. According to the IMS data in 2017, the patent expiry up to 2025 is likely to grow the generic injectable market from US$ 40 Bn in 2014 to US$ 70 Bn in 2020, of which the major share is contributed by the injectables. For instance, Cetuximab (Erbitux) is an epidermal growth factor receptor inhibitor that is indicated for the treatment of metastatic colorectal cancer and head and neck cancer. It is manufactured under the patent by Bristol-Myers Squibb, an American multinational pharmaceutical company and Eli Lily Company, a global pharmaceutical company. The patent of the drug expired in 2018 and thus, is expected to provide opportunity for a large number of market players to manufacture its generic version.
Rising incidence of cancer cases
Increasing incidence of cancer cases in Asia Pacific region is expected to augment the growth of Asia Pacific generic oncology sterile injectable market. According to Economic Intelligence Unit Limited — a Britain-based business within the Economist Group — in 2016, South Korea witnessed one of the world’s fastest increase in breast cancer incidence, with the age-standardized rates (ASR) roughly tripling over the past two decades.
Asia Pacific Generic Oncology Sterile Injectable Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 1,303.8 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 12.2% | 2028 Value Projection: | US$ 8,832.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Eli Lily and Company, Biocon Ltd., Sun Pharmaceutical Industries Ltd., Dr. Reddy’s Laboratories Ltd, Baxter International Inc., Hikma Pharmaceuticals, Mylan N.V., Sandoz International GmbH, Teva Pharmaceutical Industries Ltd., and Pfizer Inc |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Asia Pacific Generic Oncology Sterile Injectable Market – Restraints
Shortage of supply for various important generic drugs, especially the injectable form of drugs that are primarily used in cancer chemotherapy, is a major problem faced globally. Due to consistent shortage, patients are unable to gain access to basic life-saving drugs and this shortage is expected to severely affect the growth of the market.
The reasons for the shortage of drugs are increasing manufacturing issues such as introduction of particulate matter in the vials, equipment failures, and fungal or bacterial contaminations, delay in supply due to unavailability of raw material to start drug manufacture, and shortage of active pharmaceutical ingredients (API). Low availability of raw materials is one of the major issues, which is expected to adversely affect the growth of Asia Pacific oncology sterile injectable market.
Asia Pacific Generic Oncology Sterile Injectable Market – Country Analysis
On the basis of country, the Asia Pacific generic oncology sterile injectable market is segmented into China, India, Japan, Australia, South Korea, ASEAN, and Rest of Asia Pacific.
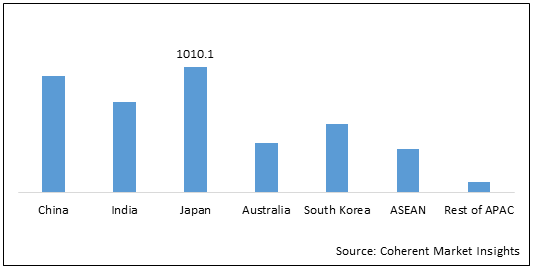
Japan is expected to hold dominant position in the Asia Pacific generic oncology sterile injectable market growth over the forecast period, owing to increasing number of mergers and acquisitions by market players. For instance, in 2016, Nichi-Iko Pharmaceutical Co., Ltd, a Japan based company which manufacture and sale of generic pharmaceutical products and Sagent Pharmaceuticals Inc. entered into a definitive merger agreement. The merger brought together two leading companies in the generics market with a broad product portfolio.
Figure 2: Asia Pacific Generic Oncology Sterile Injectable Market Value (US$ Mn), by Country, 2021
To learn more about this report, Request Free Sample
Asia Pacific Generic Oncology Sterile Injectable Market – Competitive Landscape
Major players operating in the Asia Pacific generic oncology sterile injectable market include Eli Lily and Company, Biocon Ltd., Sun Pharmaceutical Industries Ltd., Dr. Reddy’s Laboratories Ltd, Baxter International Inc., Hikma Pharmaceuticals, Mylan N.V., Sandoz International GmbH, Teva Pharmaceutical Industries Ltd., and Pfizer Inc.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients