The Generic Oncology Sterile Injectables Market is estimated to be valued at 35.8 Bn in 2026 and expected to reach USD 68.9 Bn by 2033, witnessing a CAGR of 9.8% over the forecast period (2026-2033). This growth is driven by rising prevalence of cardiovascular diseases and growing demand for continuous ambulatory cardiac monitoring solutions.
The market for generic oncology sterile injection drugs is defined as injectable cancer drugs that are manufactured under sterile conditions and used in chemotherapy, targeted therapy, and supportive care for cancer patients and are therefore critical to the practice of oncology due to their rapid systemic delivery and high efficacy in the treatment of aggressive cancers. The generic oncology sterile injectable market exists within the larger pharmaceutical and oncology environment and serves hospitals, cancer treatment centers, and specialty clinics, with applications across many different types of cancers including lung cancer, breast cancer, and hematologic cancer.
The key drivers of growth in this market are the increasing number of oncology drugs that are coming off patent, the rising pressure on healthcare costs, and the overall strong regulatory support for generic drug approvals. Technological advancements in sterile manufacturing processes and supply chain optimization are further enhancing the ability to develop products on the larger scale. The generic oncology sterile injection Market plays an important role in increasing the accessibility and affordability of cancer treatments globally, particularly in emerging markets where the costs of branded therapies create barriers to access.
|
Current Event |
Description and its Impact |
|
U. S. FDA’s Increased Approvals of Generic Drug Products |
|
|
Global Oncology Frug Shortage crisis |
|
|
Expansion of India’s PLI pharmaceuticals manufacturing subsidies. |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The chemotherapy drug category will be leading the market at a share of approximately 46.2% in 2026 because they are still used as primary line of treatment for many different types of cancers. Although the increase in targeted therapies has been significant, these drugs are still vital to treating many cancers due to their ability to treat a broad range of cancers effectively in their generic formulations.
The segment of lung cancer is expected to be the largest in terms of market share in 2026. It will command nearly 29.8% of the entire market with a significant share being attributed to global burden of lung cancer and the widespread adoption of injectable chemotherapy regimens for the treatment of this disease. The increasing rates of tobacco usage by individuals as well as environmental factors are increasingly being identified as contributors to the rising rates of incident cases of lung cancer.
In February 2025, Dr. Reddy’s Laboratories introduced a generic injectable for lung cancer treatment in Europe, targeting increased accessibility.
As of 2026, hospital pharmacies constitute approximately 52.5% of the total share of market activity. Oncology injectables are administered by a physician only under strict supervision, making them one of the only types of cancer drugs to require administration under safe prescriptive guidelines. The hospital setting continues to be the primary location where patients receive treatment for their cancer and have access to requisite support from the properly trained healthcare practitioners.
To learn more about this report, Request Free Sample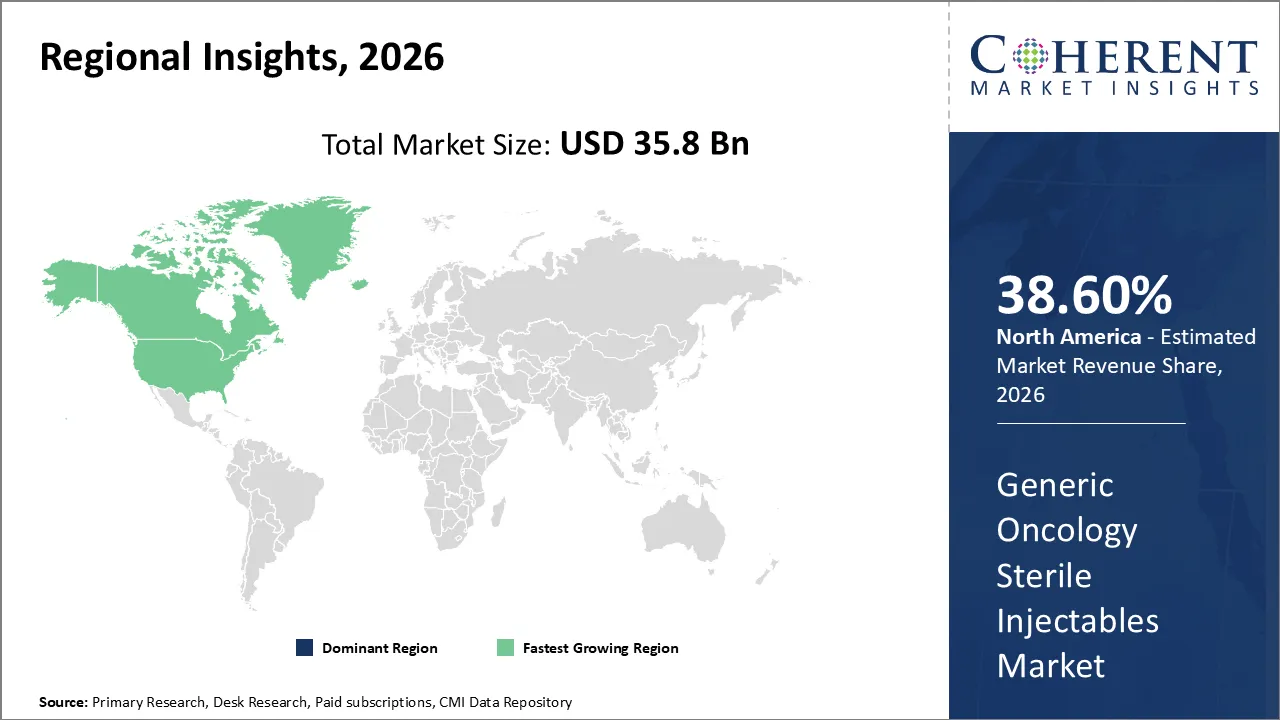
North America is the major region of the generic oncology sterile injectables market, accounting for 38.60% of market share. High incidence of cancer, advanced healthcare infrastructure and strong regulations are the contributing factors to North America being the most dominant region.
Asia Pacific is the fastest-growing region of the generic oncology sterile injectables market due to the expansion of healthcare infrastructure and increased awareness of cancer.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 35.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.8% | 2033 Value Projection: | USD 68.9 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Eli Lilly & Company., Biocon Ltd, Baxter International Inc., Hikma Pharmaceuticals PLC, Mylan N.V., Sandoz International GmbH, Teva Pharmaceutical Industries Ltd, Pfizer Inc. |
||
| Growth Drivers: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
As a result of the growing incidence of cancer affecting the entire globe, oncology services continue to experience strong demand due to their being highly sought after and relatively inexpensive for many patients. As countries deal with an aging population and increasing cancer cases related to lifestyle choices, access to affordable methods of treating cancer become a major focus of health systems and a number of other entities are positively impacting the oncology market.
As a large group of blockbuster oncology patents are scheduled to expire in the coming years this will allow generic companies to be able to enter the oncology market and providing competition, lower drug prices, and improving patient access to cancer treatments across the entire globe.
With the rising incidence of cancer and a global focus on decreasing cost for healthcare, the growth potential for the generic oncology sterile injectables market remains very high. The market is expected to experience rapid change in its competitive landscape due to continued technological advancements and other similar products, despite the continued regulatory hurdles and challenges associated with making those products. Companies that concentrate their efforts on developing new product/services, ensuring compliance and having resilient supply chain will have the greatest amount of competitive advantage within this market over time.
In August 2025, Amneal Pharmaceuticals launched a new oncology sterile injectable in North America, focusing on improving supply reliability and reducing drug shortages.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients