Global bacterial & viral specimen collection market is estimated to be valued at USD 25.62 Bn in 2026 and is expected to reach USD 62.93 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 13.7% from 2026 to 2033. Factors like increasing prevalence of infectious diseases, growing awareness regarding specimen collection techniques, and rapidly improving healthcare infrastructure and services across geographies can drive the market growth.
Discover market dynamics shaping the industry: Request Free Sample
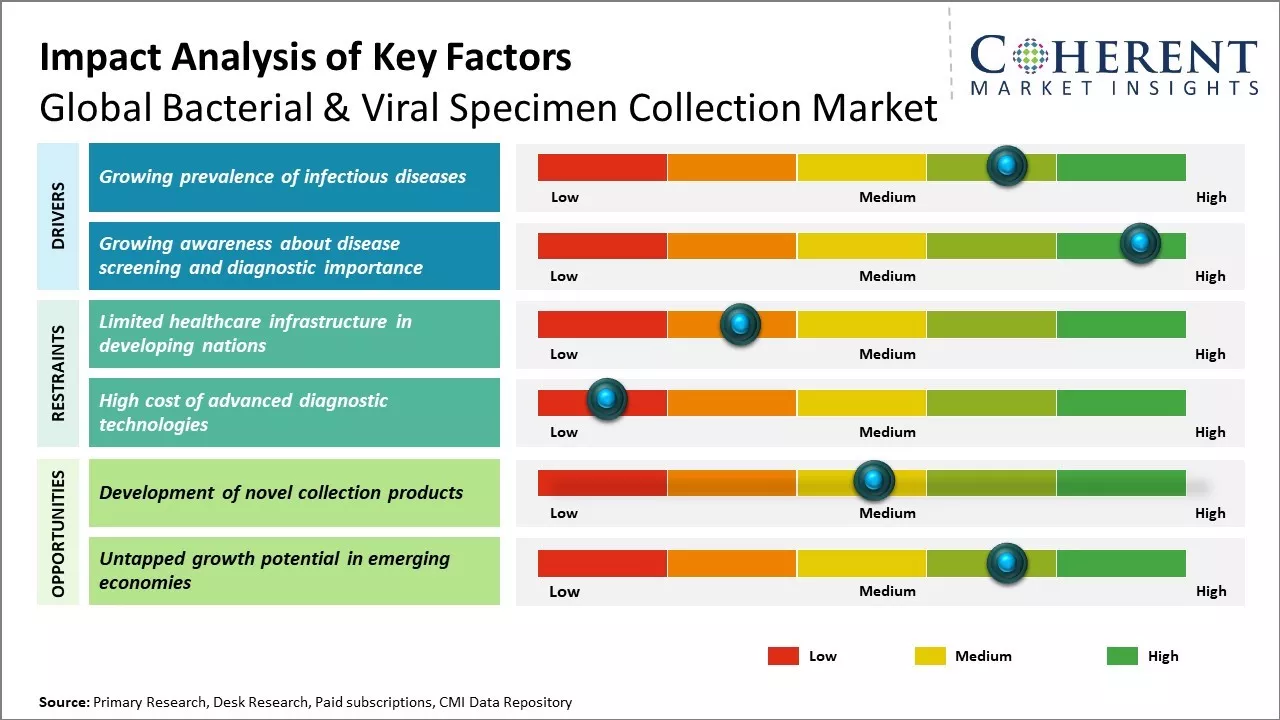
Growing prevalence of infectious diseases
Rising prevalence of infectious diseases can drive the market growth. Various factors such as climate change, increased global travel and connectivity, rising antimicrobial resistance, aging population, and environmental degradation contribute to prevalence of infectious disease. As per the WHO, infectious diseases account for nearly 15 million deaths each year worldwide. There has been increase in incidences of many viral and bacterial illnesses that can have serious health and economic implications such as tuberculosis, influenza, hepatitis, HIV/AIDS, pneumonia and several vector-borne diseases.
With growing international travel and migration, diseases are also spreading across borders much rapidly than before. Outbreaks that may have remained contained locally in the past can now turn into global epidemics within weeks. This presents serious public health challenges. Overuse and misuse of antimicrobial drugs has led to many microbes developing resistance to available treatments. Drug-resistant infectious pathogens have emerged as one of the biggest threats facing humanity. All these factors reinforce the need for better infectious diseases surveillance, early detection, and prompt diagnosis worldwide. Rising population susceptibility due to factors such as aging, greater prevalence of chronic diseases and immunosuppression has also contributed to the higher infectious diseases burden. The elderly people and people with compromised immunity are at an elevated risk of infections. This underscores the importance of diligent specimen collection, targeted screening, and timely administration of appropriate treatments.
Get actionable strategies to beat competition: Request Free Sample
Growing awareness about disease screening and diagnostic importance
In recent years, public understanding about the significance of regular health screening and early detection of illnesses has enhanced considerably. People are more informed now about how identifying infections early through appropriate tests can help receive prompt treatment and potentially avoid severe complications. There is growing realization that ignoring symptoms or delays in seeking medical attention can adversely impact health outcomes, especially for infectious diseases.
Various health awareness drives by governments and international agencies can drive the market growth. The advent of new diagnostic technologies and tests has boosted test accessibility and affordability in significant parts of the world. The COVID-19 pandemic, in particular, highlighted the value of diagnostic testing and highlighted how expanded testing capacity and infrastructure are vital for addressing public health emergencies. It underscored the need for continuous disease monitoring to quickly identify new outbreaks. Deadly infectious disease outbreaks in the past like Zika and Ebola outbreak accentuated the indispensable role of accurate and timely diagnosis. Rising health insurance penetration and availability of subsidized screening programs for sections of population have additionally boosted screening uptake. Growing recognition and emphasis on prevention and early detection of illnesses aid the expansion of bacterial & viral specimen collection market. With enhanced awareness, individuals are displaying greater proclivity for recommended medical checkups and tests.
Key Takeaways from Analyst:
Global bacterial & viral specimen collection market growth is driven by rising prevalence of infectious diseases. Increasing incidence of hospital-acquired infections and pandemic outbreaks can boost demand for specimen collection kits. Advancements in molecular diagnostic techniques requiring quality sample collection can also bost new product development.
North America currently dominates the market, owing to well-established healthcare infrastructure and stringent regulations regarding laboratory sample collection and shipping. However, Asia Pacific is expected to emerge as the fastest growing regional market due to rising healthcare spending, growing focus on modernizing healthcare facilities, and expansion of private sector hospitals in high potential countries like China and India.
Stringent safety regulations pertaining to shipping of biohazard samples can hamper the market growth. High costs associated with maintaining cold chain for temperature-sensitive specimens can also hamper the market growth. Lack of skilled technicians in underdeveloped regions hampers market growth.
New product launches catering to disease-specific sample collection and expanded manufacturing capacity in developing Asian nations can provide lucrative opportunities for manufacturers. Increased cooperation between international diagnostic companies and regional vendors can help bolster market access in emerging markets. Widespread adoption of viral specimen collection kits along with growing awareness regarding collection procedure optimization can drive the market growth.
Market Challenges: Limited healthcare infrastructure in developing nations
Global bacterial & viral specimen collection market growth can be hampered due to limited healthcare infrastructure in many developing nations. Many countries in Asia, Africa, and Latin America still lack proper medical facilities and trained healthcare professionals even in major cities. This becomes more problematic in remote and rural areas which have little to no access to basic diagnostic services. The lack of adequate laboratories, trained lab technicians, specimen transportation networks, and others severely hampers early testing and diagnosis of various bacterial and viral infections in these regions. This subsequently affects the demand for specimen collection products in such nations. Unless efforts are made by governments and international organizations to strengthen primary healthcare facilities, improved access to infectious disease diagnostics will remain limited especially in resource-poor settings.
Market Opportunities: Development of novel collection products
Global bacterial & viral specimen collection market has a significant opportunity in developing innovative collection product offerings that can address the specific needs of testing in remote locations. With advancements in molecular diagnostic technologies, there is a scope for startups and medical device companies to design novel self-collection kits, more stable transport medias and integrated diagnostic platforms that do not require sophisticated lab infrastructure. Development of products with longer shelf life, higher accuracy from small sample volumes, integrated pathogen preservation capabilities can expand the reach of infectious disease testing. Novel 3D printed cartridges for isothermal PCR instruments have potential to provide lab-quality diagnostics at the point of care. Companies investing in R&D around user-friendly, affordable and versatile collection solutions can tap into the huge untapped market, especially in developing world.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
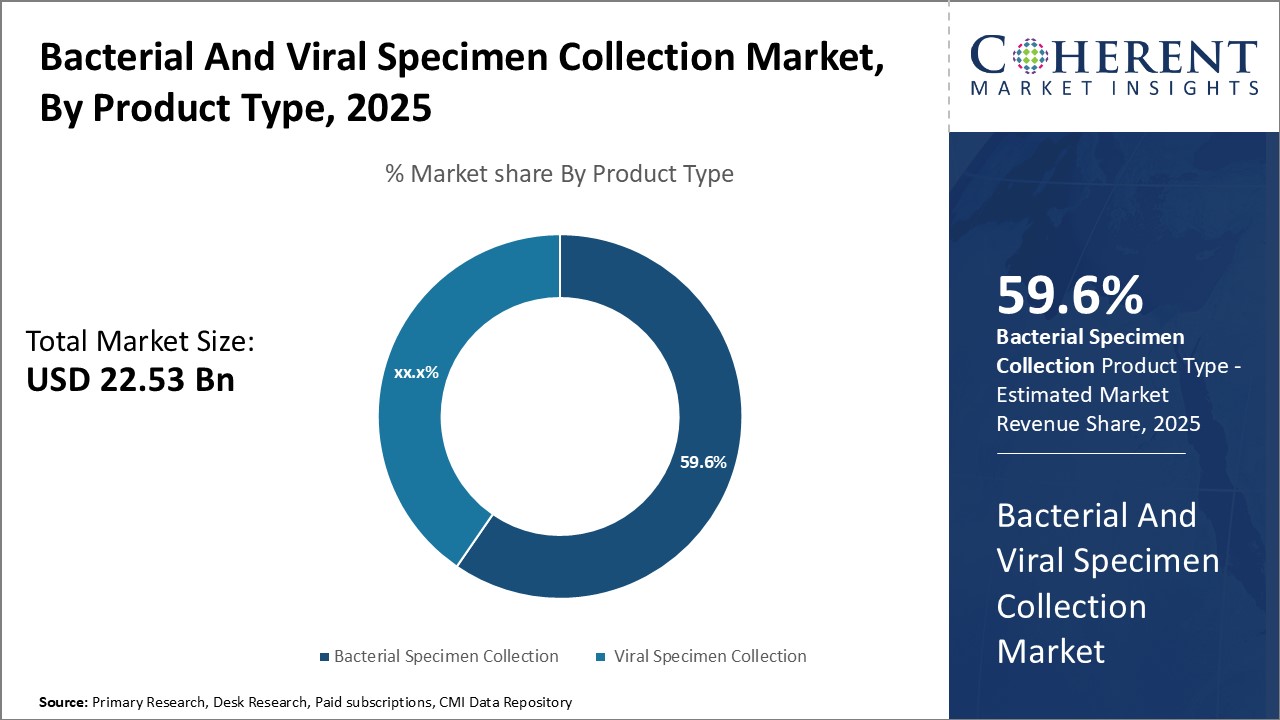
By Product Type- Bacterial specimen collection dominates due to growing diagnostics need
In terms of product type, bacterial specimen collection segment is estimated to contribute the highest market share of 60.6% in 2026, owing to growing need for fast and accurate diagnosis of bacterial infections. As bacterial infections remain a major public health concern worldwide, there has been huge demand for bacterial diagnostic tests. Specimen collection is the first critical step in bacterial diagnostics. Among bacterial specimen collection products, swabs continue to be the most widely used due to their ease of use and reliability in collecting diagnostic specimens without causing discomfort to patients. However, bacterial transport media plays an equally important role in maintaining the quality and integrity of specimens during transport and storage. Sample collection kits that integrate swabs and transport media are gaining popularity as these improve workflow efficiency for healthcare practitioners. Bacterial specimen collection segment growth is driven by increased diagnostic testing for bacterial diseases.
To learn more about this report, Request Free Sample
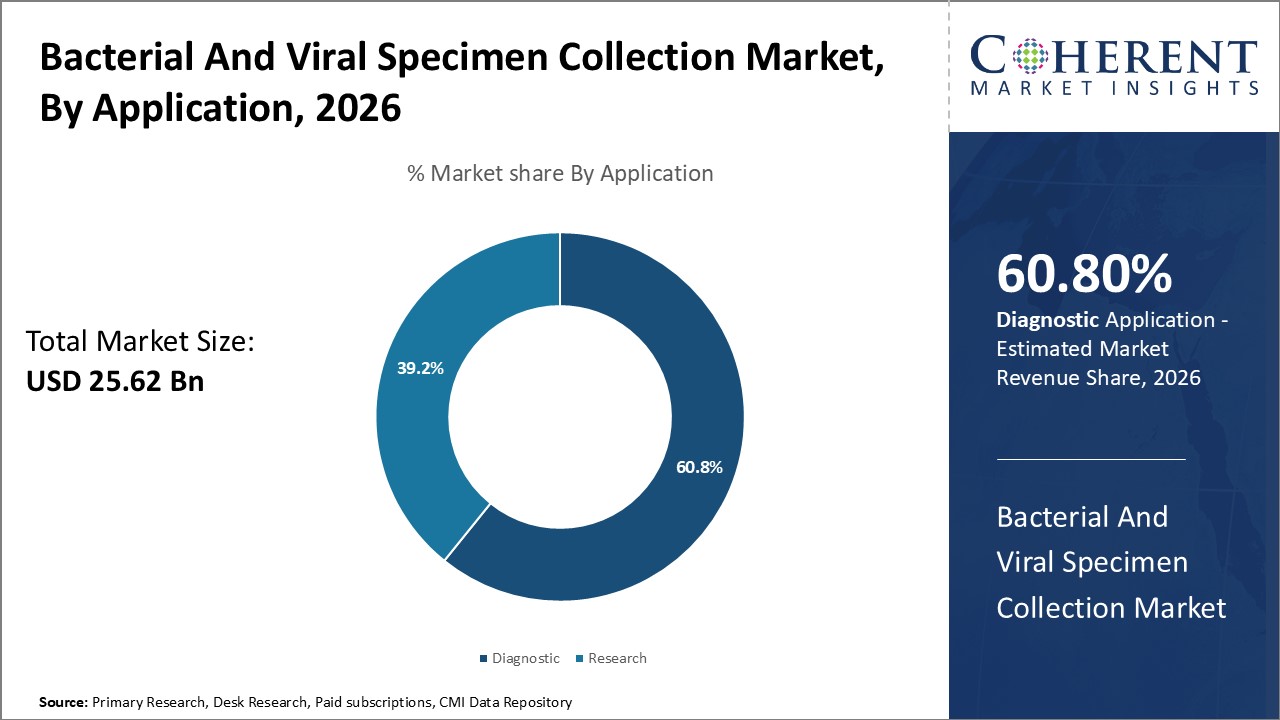
By Application- Rising diagnostic testing volumes boosts diagnostics segment growth
In terms of application, diagnostics segment is estimated to contribute the highest market share of 60.8% in 2026. Timely and accurate diagnosis is critical for both bacterial and viral infections. Advanced diagnostic technologies have significantly improved testing capabilities in recent years. This has enabled laboratories to process large volumes of tests on a daily basis to meet healthcare needs. As new and re-emerging infectious diseases pose challenges, there has been growing emphasis on making diagnostic testing more affordable and accessible. This boosts demand for bacterial and viral specimen collection products that have reliable performance, and are easy to use. Within diagnostics, microbiological testing remains a major application area. Moreover, point-of-care testing is gaining popularity due to advantages such as rapid turnaround times.
By End User- Rising emphasis on quality specimen collection boosts demand for hospital
In terms of end user, hospitals & clinics segment is estimated to contribute the highest market share of 43.5% in 2026, due to growing awareness about quality specimen collection. As frontline healthcare facilities, hospitals and clinics play a pivotal role in disease outbreak response and public health surveillance. Ensuring reliable initial diagnostic specimen collection is critical in infectious disease management. Lack of adequate infection control practices during specimen collection can affect diagnostic accuracy. Therefore, hospitals place strong emphasis on adopting standardized operating procedures and using certified collection methods/media. There is also greater focus on staff training in collection techniques. The complex requirements are best addressed through integrated solutions offered by major specimen collection vendors. Furthermore, point-of-care testing expansion allows hospitals to decentralize diagnostic capabilities. These factors boosts demand from hospitals and clinics. Other segments such as diagnostic laboratories and research institutions also contribute significantly as testing volumes rise globally.
Need a Different Region or Segment? Request Free Sample
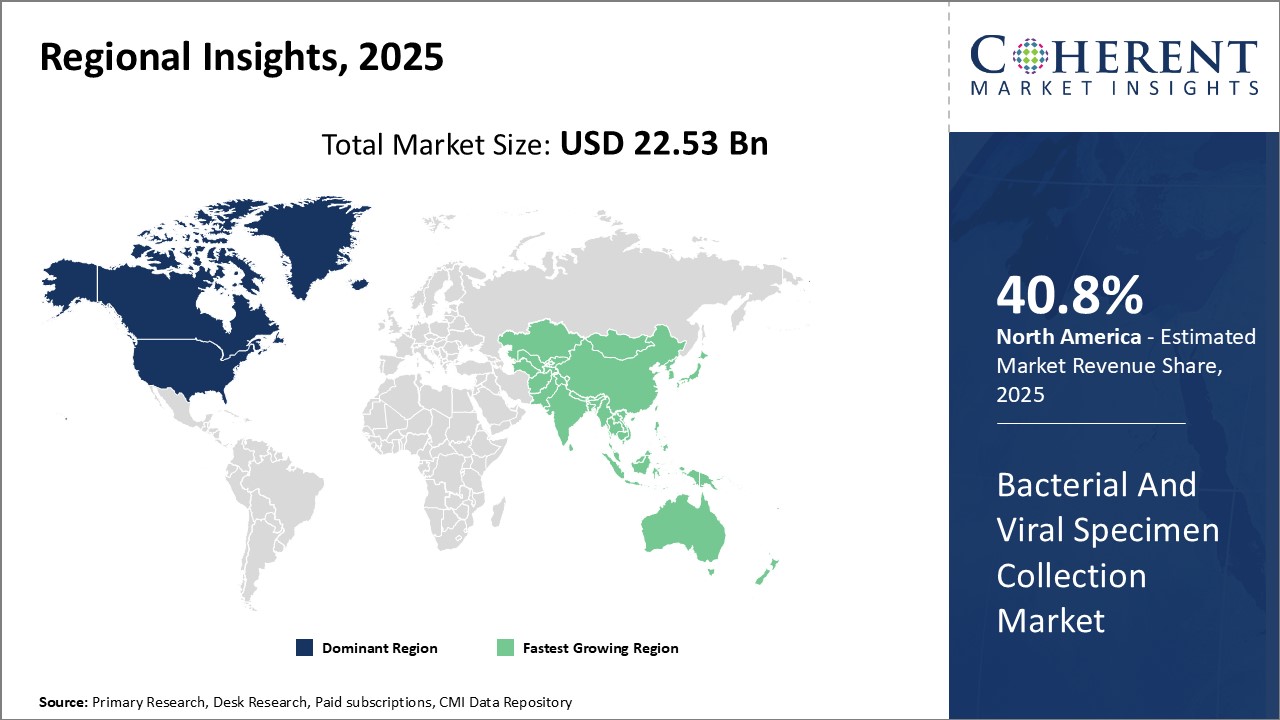
North America dominates the global bacterial & viral specimen collection market currently with an estimated market share of 40.8% in 2026, due to high prevalence of infectious diseases in the U.S. and Canada, coupled with heavy spending on healthcare and well-established healthcare infrastructure. Both the countries have large number of clinical diagnostic labs and hospitals, which are high consumers of specimen collection products. Moreover, North America is home to several leading market players who have their global headquarters located in the region.
Asia Pacific region has emerged as the fastest growing market for bacterial & viral specimen collection. Rising healthcare expenditure, growing geriatric population prone to chronic illnesses and infections, and increasing focus on modern healthcare services can drive the market growth. Presence of many developing nations such as China and India with huge populations can drive the market growth. These countries are witnessing rapid establishment as well as upgradation of healthcare facilities and diagnostic labs.
Japanese market stood out in the Asia Pacific bacterial & viral specimen collection space due to higher per capita healthcare spending and robust healthcare infrastructure comparable to developed western countries. Although specimen collection products in Japan attract premium prices, strong demand from large hospitals and diagnostics chains ensures stable sales volumes.
Improving accessibility to modern medicines and diagnostics across small towns and rural regions can create new avenues. Large diagnostics chains and hospital groups in Asia Pacific countries are increasingly adopting advanced specimen collection solutions to enhance convenience and quality of patient care. This rise of organized healthcare provides widespread impetus to the bacterial & viral specimen collection market across the fast-growing Asia Pacific region.
Bacterial And Viral Specimen Collection Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 25.62 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 13.7% | 2033 Value Projection: | USD 62.93 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
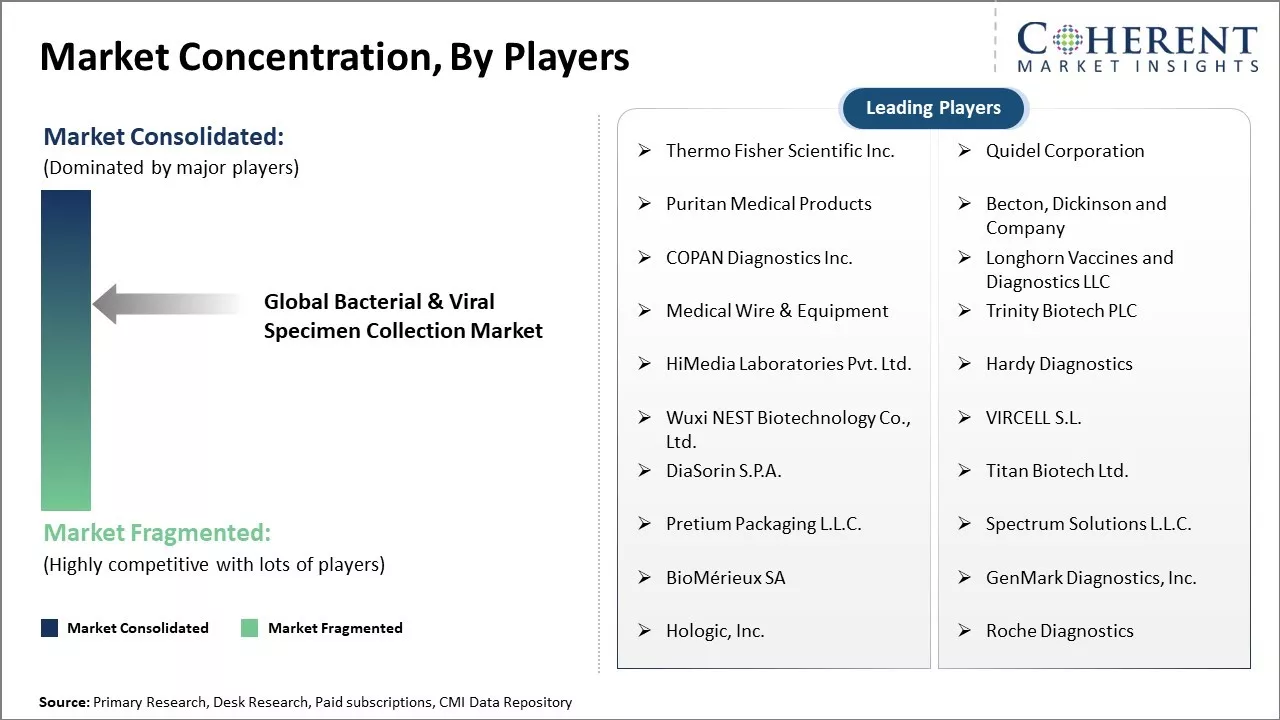
| Companies covered: |
Thermo Fisher Scientific Inc., Quidel Corporation, Puritan Medical Products, Becton, Dickinson and Company, COPAN Diagnostics Inc., Longhorn Vaccines and Diagnostics LLC, Medical Wire & Equipment, Trinity Biotech PLC, HiMedia Laboratories Pvt. Ltd., Hardy Diagnostics, Wuxi NEST Biotechnology Co., Ltd., VIRCELL S.L., DiaSorin S.P.A., Titan Biotech Ltd., Pretium Packaging L.L.C., Spectrum Solutions L.L.C., BioMérieux SA, GenMark Diagnostics, Inc., Hologic, Inc., Roche Diagnostics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global Bacterial & Viral Specimen Collection Market provides essential tools and kits for the collection and transportation of bacterial and viral specimens. This market includes a diverse range of products such as swabs, viral transport media, specimen collection tubes, and viral collection kits. These items are designed to help healthcare facilities efficiently gather and safely transport specimens, ensuring accurate laboratory testing and diagnosis of bacterial and viral infections. Proper specimen collection and transportation are critical for obtaining reliable laboratory results and delivering effective patient care.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients