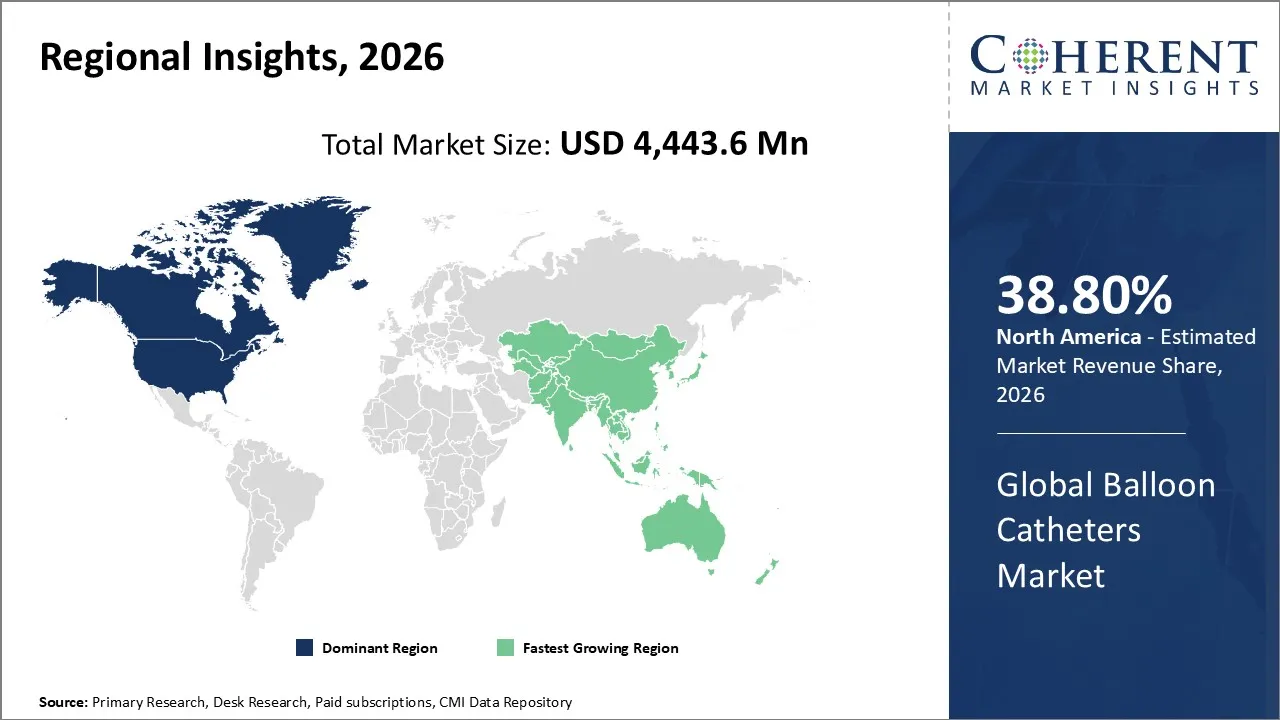
The Balloon Catheters Market is estimated to be valued at USD 4,443.6 Mn in 2026 and is expected to reach USD 6,689 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.3% from 2026 to 2033.
The Balloon Catheters Market is growing steadily worldwide as cardiovascular and peripheral artery diseases become more prevalent, the population ages, and healthcare providers increasingly adopt minimally invasive procedures. Innovations such as drug-coated, scoring, and semi-compliant balloons are improving procedural efficiency and safety. Expanding healthcare infrastructure, supportive reimbursement policies, and the use of balloon catheters in areas beyond cardiology, including urology and neurology, are driving further market growth. North America and Europe dominate the market, while emerging regions continue to increase adoption.
|
Current Events |
Description and its impact |
|
Geopolitical and Regulatory Events |
|
|
Economic and Market Dynamics |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Medicare reimbursement for angioplasty balloon catheters is determined by compliance with Centers for Medicare & Medicaid Services (CMS) guidelines, emphasizing medical necessity, accurate documentation, and correct coding. Coverage is typically provided when the procedure is deemed clinically appropriate and supported by patient records. Reimbursement rates vary based on the care setting, such as inpatient hospitals, outpatient facilities, or ambulatory surgical centers, and are influenced by applicable Diagnosis-Related Groups (DRGs) or Ambulatory Payment Classifications (APCs). Providers must follow CMS billing conventions, including the use of appropriate CPT and HCPCS codes, to ensure timely and accurate payment for balloon catheter–based angioplasty procedures.
Nylon acquired the prominent market share of 45.6% in 2026. Nylon plays a key role in the balloon catheters market because it offers high strength, long-term durability, and reliable performance under elevated inflation pressures. Manufacturers actively select nylon for its optimal balance of flexibility and structural stability, enabling accurate balloon expansion. Its established biocompatibility improves patient safety, while economical manufacturing and easy material availability support broad usage. Furthermore, increasing demand for high-pressure and specialty balloon catheters in cardiovascular and peripheral procedures continues to reinforce the adoption of nylon-based products across the market.
Semi-compliant (Mid pressure) Balloons expected to hold the largest market share of 33.3% in 2026. Semi-compliant (mid pressure) balloons are gaining demand in the balloon catheters market because they deliver controlled expansion while conforming to different vessel anatomies. Clinicians actively choose these balloons for their versatility in lesion preparation and predilatation, as they help limit the use of multiple devices. Increasing numbers of coronary and peripheral interventions, combined with a strong focus on procedural efficiency and safety, are boosting adoption. Ongoing improvements in material design, enhanced trackability, and broader use in minimally invasive procedures are further driving growth in this segment.
To learn more about this report, Request Free Sample
North America dominates the overall market with an estimated share of 38.80% in 2026. Strong adoption of minimally invasive procedures and early uptake of advanced interventional technologies are shaping the North America balloon catheters market. Hospitals and ambulatory surgical centers actively use specialized balloons, including drug-coated and high-pressure types, to enhance treatment outcomes. A robust healthcare infrastructure, high procedure volumes, and an experienced clinical workforce sustain steady demand. Continuous product innovation, the expansion of outpatient services, and the increasing application of balloon catheters in cardiovascular, peripheral, and neurovascular procedures are further defining market trends across the region. For instance, Boston Scientific Corporation received FDA approval for the AGENT™ Drug-Coated Balloon (DCB) to treat coronary in-stent restenosis (ISR) in patients with coronary artery disease, a condition where plaque or scar tissue narrows a stented vessel.
Expanding healthcare infrastructure and increasing awareness of interventional treatments are driving rapid evolution in the Asia Pacific balloon catheters market. Governments and private players are investing more in hospitals and catheterization laboratories, which improves access to advanced procedures. A rising number of patients with cardiovascular and peripheral disorders is boosting procedure volumes. Manufacturers are actively developing cost-effective and technologically enhanced balloon catheters to address regional demand. In addition, supportive government policies, growing medical tourism, and broader adoption of minimally invasive techniques are shaping market trends across both emerging and developed countries in the region. For instance, in September 2025, Edwards Lifesciences launched its next-generation transcatheter heart valve platform in India, advancing aortic stenosis treatment. The new balloon-expandable valve uses proprietary calcification-resistant tissue to reduce calcium buildup and lower the risk of valve deterioration and reintervention.
High utilization of interventional procedures and rapid adoption of advanced medical technologies are driving the United States balloon catheters market. Healthcare providers are actively implementing innovative balloon designs to enhance procedural precision and improve patient outcomes. The emphasis on outpatient care and the growth of ambulatory surgical centers are transforming treatment environments. Ongoing product development, a skilled clinical workforce, and increasing applications of balloon catheters in cardiovascular, peripheral, and neurovascular procedures are further defining key market trends across the country. For instance, Medtronic launched the Arctic Front™ Cardiac Cryoablation Catheter System, the first and only CDSCO-approved cryoballoon catheter for treating atrial fibrillation (AF).
Modernizing healthcare infrastructure and increased investment in advanced catheterization facilities are driving rapid growth in the China balloon catheters market. Rising awareness of cardiovascular and peripheral diseases and higher procedure volumes are boosting demand. Manufacturers are actively launching cost-effective, high-performance balloon catheters to meet local requirements, while medical professionals are adopting minimally invasive interventions to enhance patient outcomes. Government initiatives expanding healthcare access, along with the rise of private hospitals and medical tourism, are further shaping market dynamics across both urban and developing regions in China.
Healthcare providers are increasingly shifting from open surgeries to minimally invasive procedures, such as percutaneous coronary and peripheral interventions. Balloon catheters play a central role in reducing patient recovery time, minimizing complications, and enabling outpatient treatments. Hospitals and surgical centers are investing in advanced balloon technologies to meet this demand, driving innovation in design, flexibility, and precision. This trend is particularly strong in regions with mature healthcare systems seeking efficiency and improved patient outcomes.
The shift toward minimally invasive interventions and outpatient care creates opportunities for balloon catheters designed for efficiency, safety, and rapid recovery. Devices with improved trackability, compact designs, and versatility can meet the needs of ambulatory surgical centers. Companies that innovate to provide multi-functional or single-use balloon catheters tailored for high-volume outpatient settings can capture market share while supporting healthcare providers in enhancing procedural outcomes and operational efficiency.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4,443.6 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.3% | 2033 Value Projection: | USD 6,689 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Abbott Laboratories, Becton Dickinson and Company, Boston Scientific Corporation, Bard Medical Division, B. Braun Melsungen AG, Johnson & Johnson, Teleflex Incorporated, Medtronic, Plc, Edwards Lifesciences Corporation, and Vascular Solutions, Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Manisha Vibhute is a consultant with over 5 years of experience in market research and consulting. With a strong understanding of market dynamics, Manisha assists clients in developing effective market access strategies. She helps medical device companies navigate pricing, reimbursement, and regulatory pathways to ensure successful product launches.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients