The blockchain based solutions for security labels market size is anticipated to grow at a CAGR of 6.5% with USD 29.7 Bn in 2026 and is expected to reach USD 48.60 Bn in 2033. The primary drivers are largely defined by a global arms race against sophisticated counterfeiting (1 in 10 medicines), increasing regulatory pressure for product provenance, and a shift toward digital-first supply chain transparency.
Radio Frequency Identification (RFID) is projected to account for the largest blockchain based solutions for security labels market share of 45.6% in 2026. This is due to the fact that it allows for automated, non‑line‑of‑sight identification and data capture that enhances blockchain‑backed traceability and anti‑counterfeiting across complex supply chains. RFID tags are mainly used for tracking goods, managing inventory, and improving visibility. Thus, making them a preferred choice where secure labeling is critical. Radio Frequency Identification (RFID) , thereby reflecting rising demand for tracking technologies that can integrate with distributed ledgers for improved security.
The U.S. Drug Supply Chain Security Act (DSCSA) mandates an electronic, interoperable system to identify and trace prescription drugs at the unit level, thus propelling adoption of secure technologies. The FDA’s DSCSA pilot projects, including the MediLedger effort, demonstrated that blockchain‑based solutions can meet interoperability requirements and validate product authenticity throughout the supply chain.
To learn more about this report, Request Free Sample
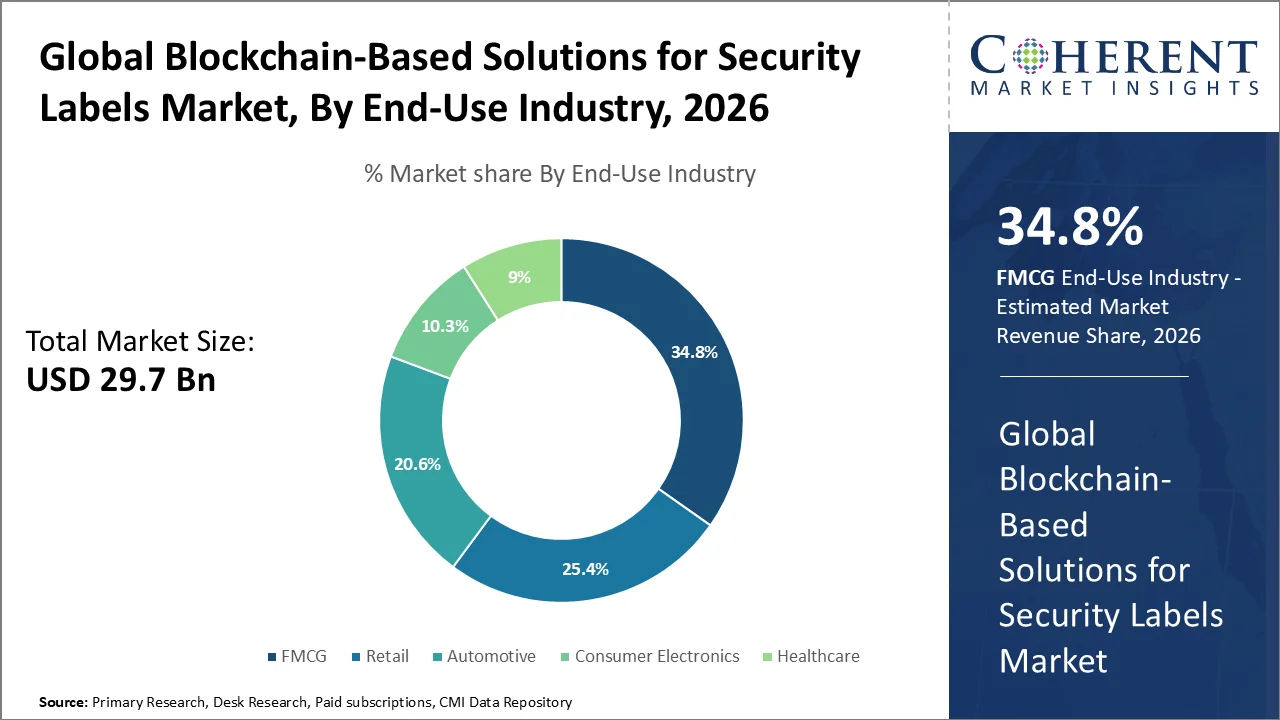
On the basis of end-use industry, FMCG leads with a major 34.8% share in 2026. The segment’s growth is due to its heavy dependence on fast, verifiable, and tamper-resistant labeling in food, beverage, personal care, and household product supply chains. The U.S. FDA’s Food Traceability Rule requires covered firms to maintain additional traceability records and provide requested data within 24 hours, thus boosting demand for secure digital label systems which can strengthen recall readiness and lot-level visibility.
The counterfeit pressure is also contributing to this adoption. OECD and EUIPO reported that global trade in counterfeit goods reached about USD 467 billion in 2021, equal to 2.3% of world imports, with food, cosmetics, and other everyday consumer goods among the affected categories. As FMCG companies manage high product volumes, short replenishment cycles, and rising compliance scrutiny, blockchain-enabled security labels are gaining traction for authenticity verification, traceability, and supply-chain transparency thereby accelerating blockchain based solutions for security labels market demand across global retail and distribution networks worldwide.
The rise in use of blockchain-based solutions for security labels is substantially contributing to the growth in the country. The blockchain technology provides secure and tamper-proof tracking tools to help pharmaceuticals, electronics, and luxury goods industries address the rising counterfeiting risks as well as improve supply chain transparency. The Drug Supply Chain Security Act (DSCSA) requires electronic tracking systems to identify and track prescription drugs, with rules to be followed by 2023.
According to the FDA, blockchain has been part of pilot projects testing interoperable and secure product traceability solutions. Notably, the MediLedger FDA Pilot Project tested blockchain’s potential in confirming product authenticity and ownership transparency. As regulations demand enhanced traceability and authenticity across critical sectors, blockchain-based solutions are set to drive the market due to the increasing demand for reliable and verifiable labels.
Immutable digital twin frameworks are improving blockchain based solutions for security labels by turning each protected item into a persistent, tamper-evident digital record. These can be verified in logistics, manufacturing, retail, as well as after-sales stages. This is becoming more important as the UN Transparency Protocol notes counterfeit trade accounts for an estimated 2% to 5% of global trade value. Pharmaceuticals, electronics, apparel, and food are among the most affected sectors. In the U.S., FDA says DSCSA requires interoperable, electronic package-level tracing for certain prescription drugs, thus speeding up the demand for serialisation-linked blockchain records as well as secure label authentication.
In Europe, the European Blockchain Services Infrastructure announced in February 2026 that traceability features for digital product passports are being developed. The European Parliament in March 2026 stressed rapid Digital Product Passport rollout for critical products sold online. Together, these developments demonstrate how immutable digital twins can enhance provenance, automate compliance checks, and make advanced security labels more scalable, trusted, as well as commercially valuable. An EU Observatory report also notes blockchain can enhance digital product passports with secure, transparent, immutable lifecycle information, directly supporting anti-counterfeit label ecosystems.
|
Current Event |
Description and its Impact |
|
India’s 2025 QR/Barcode Authentication Enforcement for Drug Packaging |
|
|
2025–2026 EU Digital Product Passport Consultation and ESPR Rollout |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
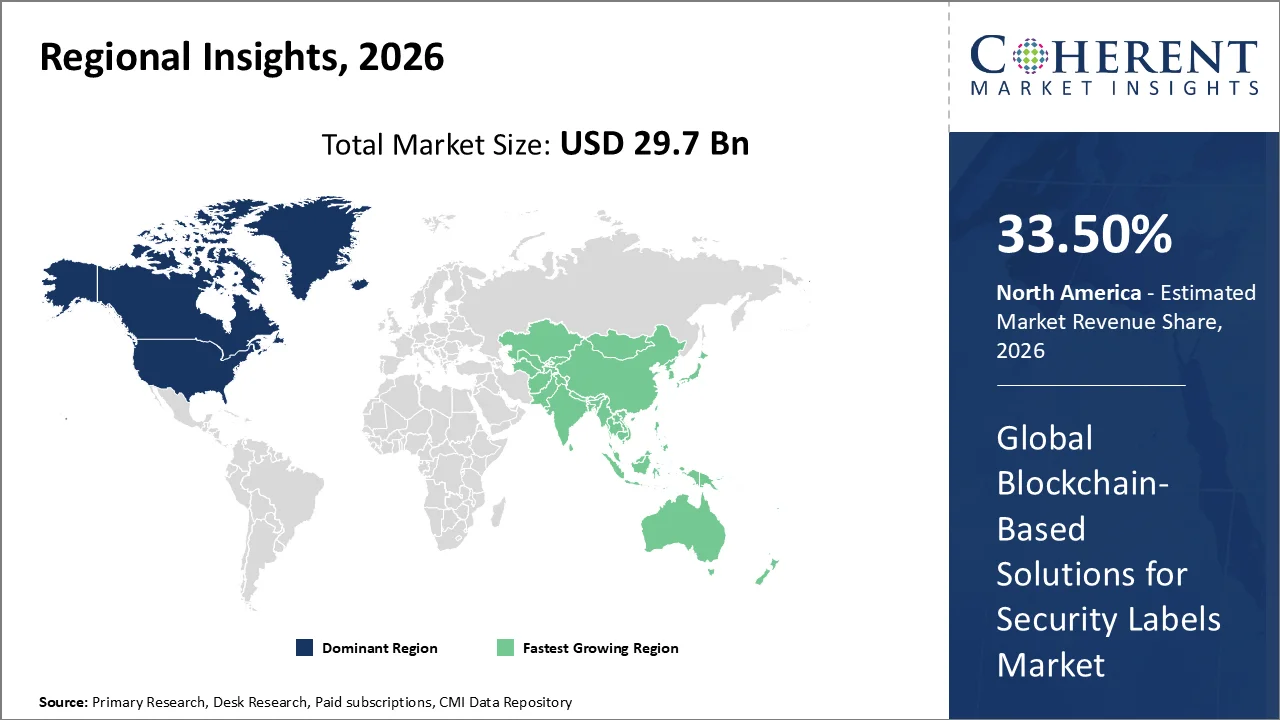
The North America region accounts for 33.50% of the blockchain based solutions for security labels market share in 2026. The region’s growth is owing to robust regulatory backing and the expanding adoption of blockchain technology in industries in order to enhance traceability and security. The FDA's DSCSA pilot program, which evaluated blockchain solutions for product tracking, delivered promising results in improving product authenticity and counterfeit prevention within the healthcare sector. This initiative highlights blockchain’s potential to ensure secure supply chains and accurate product verification, thereby propelling further interest and integration in other industries as well.
In addition, GS1 US reports that nearly 300,000 businesses in 25 industries are dependent on its standards, thereby highlighting the growing acceptance of blockchain in the U.S. supply chains. The National Institute of Standards and Technology (NIST) also strengthened this momentum by publishing new traceability frameworks in 2025. This aims at enhancing product provenance and boosting confidence in the authenticity of goods.
These regulatory advancements, coupled with the rising demand for secure labeling solutions in sectors like pharmaceuticals and FMCG, are main drivers of the blockchain based solutions for security labels market in North America.
Asia Pacific is expected to witness strong growth in blockchain based solutions for security labels market over the forecast period. Rising counterfeit exposure across pharmaceuticals, cosmetics, food, and consumer goods is a major growth driver. The World Customs Organization reported that Asia Pacific was the most frequent departure region for medicine seizures, accounting for 30% of incidents, or 1,603 cases, involving 27 million items.
Enforcement activity is also intensifying across the region. Japan Customs recorded 33,019 IP-rights import seizures in 2024, its highest level in the published series, while South Korea announced stronger measures in March 2026 for counterfeit cosmetics, including recalls and disposal. Regional institutions are also advancing digital traceability.
In 2025, APEC moved a blockchain-based shrimp traceability project to strengthen Asia-Pacific seafood value chains. Combined with Singapore Customs updated IP border-enforcement framework in February 2026, these developments are accelerating adoption of blockchain-enabled security labels across Asia Pacific markets. This momentum supports export chains needing verifiable origin, serialization, and tamper-evident authentication.
The U.S. is experiencing significant growth in blockchain based solutions for security labels market. The growth is mainly owing to the rising threat of counterfeiting and the need for supply chain transparency. As per the U.S. Customs and Border Protection (CBP) more over USD 5.4 billion worth of fake goods were seized in 2024, thus highlighting the increasing need for secure labeling technologies. In response, the FDA’s Drug Supply Chain Security Act (DSCSA) provides a structured approach to product traceability. This framework is likely to speed up the adoption of blockchain-based solutions for pharmaceutical labeling, thus ensuring enhanced security in the years ahead.
In addition, the U.S. Department of Homeland Security’s National Intellectual Property Rights Coordination Center (IPR Center) remains active in combating counterfeit goods, advocating for blockchain-based solutions to ensure digital provenance and tamper-evident labeling. These initiatives, coupled with increasing federal investments in supply chain security, are expected to accelerate the market growth for blockchain-based security labels across the U.S.
China blockchain based solutions for security labels market is advancing steadily, supported by the country’s stronger push toward traceability, anti-counterfeiting, and digital compliance. China’s national drug-safety plan calls for broader lifecycle digital management and gradual implementation of unique device identification, which directly strengthens demand for secure, serialized labels. The National Healthcare Security Administration said China had aggregated 15.806 billion drug traceability codes by January 2025, while Shanghai reported more than 50 live blockchain scenarios and 107 projects under development in April 2025.
Some of the major key players in blockchain based solutions for security labels market are 3M Company, Avery Dennison Corporation, CCL Industries Inc., Honeywell International Inc., UPM Raflatac, Inc., OpSec Security, Inc., Tesa SE, Brady Corporation, Covectra, Inc., and Mega Fortris Sdn. Bhd.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 29.7 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.5% | 2033 Value Projection: | USD 48.60 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
3M Company, Avery Dennison Corporation, CCL Industries Inc., Honeywell International Inc., UPM Raflatac, Inc., OpSec Security, Inc., Tesa SE, Brady Corporation, Covectra, Inc., and Mega Fortris Sdn. Bhd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Suraj Bhanudas Jagtap is a seasoned Senior Management Consultant with over 7 years of experience. He has served Fortune 500 companies and startups, helping clients with cross broader expansion and market entry access strategies. He has played significant role in offering strategic viewpoints and actionable insights for various client’s projects including demand analysis, and competitive analysis, identifying right channel partner among others.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients