The global medical power supply devices market is estimated to be valued at US$ 1,941.5 Mn in 2026 and is expected to reach US$ 2,624.5 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 4.4% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Rising healthcare infrastructure development and expansion of healthcare facilities globally are expected to drive the demand. In addition, increasing expenditure on healthcare equipment as a result of the growing number of surgeries performed worldwide will also boost the adoption of medical power supply devices. Further, increasing incidence of chronic diseases and growing geriatric population are other key factors expected to propel the market growth. However, higher costs associated with medical devices may hamper market growth to some extent. Nonetheless, ongoing technological advancements in terms of enhanced features, reliability, and efficiency are anticipated to offer potential opportunity for market growth over the coming years.
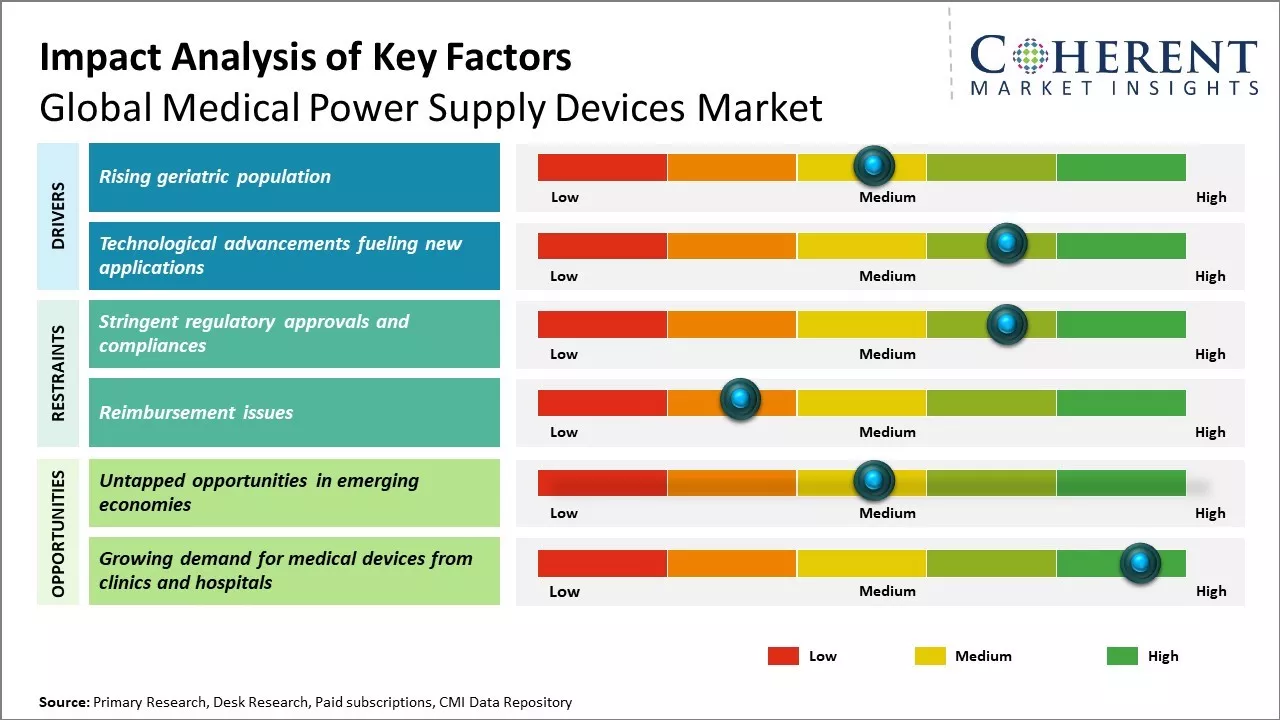
Rising geriatric population
The global geriatric population is on a steady rise. With advancing age comes numerous health issues and ailments that require close medical monitoring and support. As such, the proliferating senior demographic is increasingly relying on life-sustaining and monitoring medical devices. Power supply forms the backbone of operation for the majority of such devices. Whether it is portable oxygen concentrators for respiratory conditions, apnea monitors for sleep disorders, and infusion pumps for drug administration or defibrillators for cardiac events, reliable power is imperative for optimal performance and patient safety. The steady growth in aging population worldwide is translating into increased demand for diverse powered medical equipment at home as well as in hospitals and long-term care facilities. This makes a strong case for advanced and regulatory-compliant medical power supply solutions that can keep pace with rising healthcare needs of the senior citizens.
Get actionable strategies to beat competition: Request Free Sample
Technological advancements fueling new applications
Technological progress in the domain of healthcare has been nothing short of remarkable over the past decade. Miniaturization of electronic components, more powerful and energy-efficient processors as well as enhanced connectivity standards are enabling a wide range of point-of-care diagnostic tools and therapeutic devices. Wearables and wireless monitoring sensors are making inroads for remotely tracking vitals as well as managing chronic illnesses. Medical practitioners are unveiling innovative powered tools for advanced surgical procedures, non-invasive therapies and personalized treatment protocols. Meanwhile, the frontiers of assisted reproductive technologies, regenerative medicines, neuroscience, and genetic/molecular research continue expanding rapidly. All these ground breaking lines of work increasingly rely on customized power supply designs that must adhere to stringent International Organization for Standardization (ISO) , International Electrotechnical Commission (IEC), and regulatory norms for use on humans.
Key Takeaways from Analyst:
Key drivers for this growth include the rise in precision and digitally-controlled medical equipment which require sophisticated power solutions. Additionally, the aging population is increasing the demand for home healthcare devices which utilize compact power supplies. Emerging economies are witnessing higher investments in healthcare infrastructure that will translate to new hospital builds and equipment upgrades.
However, market growth could be restrained by stringent regulatory norms regarding safety certifications. Medical power supplies are classified as Class III devices and require extensive testing and approvals from agencies like the Food and Drug Administration before sales. Additionally, these devices have higher compliance costs. Strong competition from local and regional players also poses a challenge.
The North America region currently dominates the global medical power supply devices market and is expected to remain the largest region over the forecast period. This can be attributed to heavy investments in healthcare as well as demand for technologically advanced medical devices from the large healthcare spending population in the U.S. and Canada. Asia Pacific is poised to be the fastest growing regional market due to healthcare reforms, government focus on public health, as well as growing medical tourism in countries like India, China, and Malaysia.
Market Challenges: Stringent regulatory approvals and compliances
Stringent regulatory approvals and compliance requirements are one of the major factors restraining the growth of the global medical power supply devices market. The medical devices industry operates in a highly regulated environment with strict rules and protocols mandated by regulatory authorities across different countries and regions. Complying with these regulations involves extensive documentation, rigorous testing, and lengthy certification procedures before medical power supply devices can be marketed and sold.
This rigorous approval process leads to significant delays in launching new and upgraded products. Medical devices often require regulatory clearances from bodies like the U.S. FDA which has a standard review period of 180 days. However, complex devices frequently encounter longer review periods of 540 days or more. This prolonged approval timeline restricts medical device makers from quickly responding to the evolving needs of the healthcare sector and technological advancements. It also increases the costs for companies to stay compliant with frequent changes in regulatory guidelines.
Regulatory compliances are an ongoing requirement not just for product launch but throughout the lifecycle of a device. Medical power supply devices need to adhere to stringent performance, safety and quality standards. Manufacturing facilities selling into regulated markets are subject to periodic audits and plant inspections from bodies like the U.S. FDA and EU's MDR to ensure compliance is being maintained. The documentation and oversight involved in such compliances impose a significant resource strain on device makers. It influences their pricing strategies and overall operational expenses which impacts returns.
Market Opportunities: Untapped opportunities in emerging economies
Untapped opportunities in emerging economies present a major avenue for growth in the global medical power supply devices market. Regions such as Asia Pacific, Latin America, Middle East and Africa are experiencing significant development and infrastructure upgrades in their healthcare sectors. This acts as a catalyst for increased adoption of advanced medical devices that require reliable power supply units.
Many developing nations are working towards expanding healthcare access for their growing populations. This involves establishing new hospitals and clinics, particularly in rural and underserved areas. Given the critical nature of medical equipment, a steady and high quality power source is imperative. Therefore, increased healthcare investment naturally translates to higher procurement of medical power supplies that can withstand variable conditions and ensure uninterrupted operations. Countries are actively working to meet the United Nations' sustainable development goal of achieving universal health coverage by 2030.
On the manufacturing and technology front, emerging markets now have more cost competitive options for assembling medical power supply devices within their local industries. This allows addressing the specific needs of local healthcare facilities in a self-sufficient manner. The ability of global medical power supply brands to customize offerings, conduct local R&D and forge manufacturing partnerships will play a key role in tapping these rapidly growing yet complex emerging markets.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
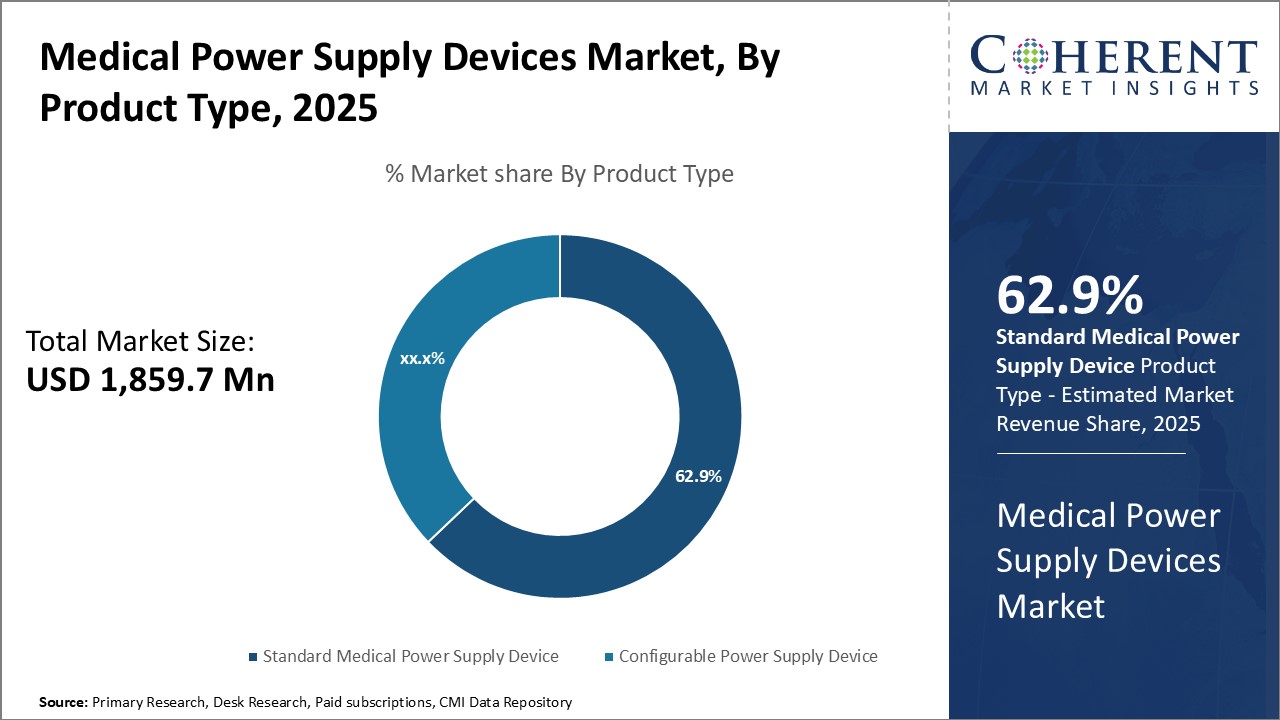
Insights By product type - Technological advancements driving Standard Medical Power Supply Device growth
In terms of product type, the standard medical power supply device segment contributes 63.9% of the market share in 2026 owing to continuous technological developments in the device. Standard medical power supply devices power a majority of basic medical equipment used across various healthcare settings. Being a mature category, manufacturers are focusing on integrating newer features to expand capabilities. Miniaturization of components has allowed designing of compact devices with higher output. Additionally, enhanced safety features such as overvoltage and overcurrent protection have increased adoption. With growing healthcare infrastructure development worldwide, demand for reliable yet affordable standard power supply devices is increasing. Various product innovations including energy efficient designs and remote monitoring functionality are expanding replacement potential of existing standard devices. Further gains can be expected as medical technology progresses and requirements for standardization of supply across equipment increases.
To learn more about this report, Request Free Sample
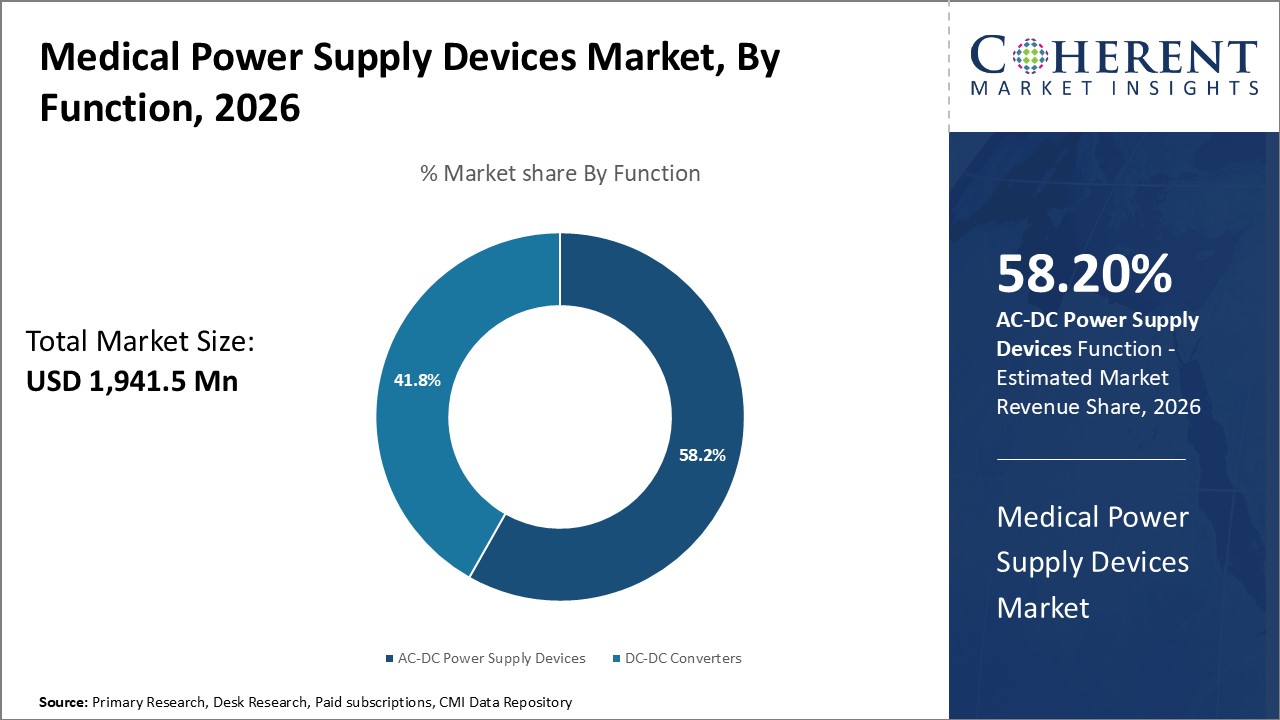
Insights By function - Rapid expansion of healthcare infrastructure is favoring AC-DC Power Supply Devices
In terms of function, the AC-DC power supply devices segment contributes 58.2% of the market share in 2026 benefitting from infrastructure investments in the healthcare sector. As hospitals and clinics upgrade to incorporate more electrical medical equipment, need for stable AC-DC converted power is surging. Most critical and life-saving devices requiring power interfaces are AC-DC compatible. With governments allocating higher budgets for modernizing healthcare facilities, installations of new state-of-the-art medical technology is growing. These installations demand compatible robust AC-DC converters to ensure uninterrupted performance of equipment. Additionally, rising medical tourism and presence of larger healthcare chains operating networked facilities is driving standardized solutions increasing preference for high-capacity multi-output AC-DC devices. Such centralized power supply infrastructure also facilitates centralized remote monitoring.
Insights By end user - Increasing role of hospitals driving adoption of Medical Power Supply Devices
In terms of end user, the hospitals segment contributes 43.6% of the market share in 2026 owing to the growing importance of hospitals in modern healthcare delivery systems. Advanced medical interventions necessitate specialized equipment and drives hospitals to expand infrastructure and amenities. This expands in-house requirement for sophisticated medical power supply devices capable of supporting diverse equipment. Additionally, hospitals acting as nodal centers see larger footfall of patients requiring access to specialized care. This necessitates reliable uptime of specialized equipment which is enabled by robust medical-grade power supply systems. Hospitals also lead equipment modernization and upgrading more frequently to incorporate new technologies. This replacement cycles further boost periodic demand for medical power supply devices. Stable inputs also allow hospitals to function round the clock without compromising on services. Comprehensive and critical inpatient care nature of hospitals makes them the biggest end-users for medical-grade power supply devices.
Need a Different Region or Segment? Request Free Sample
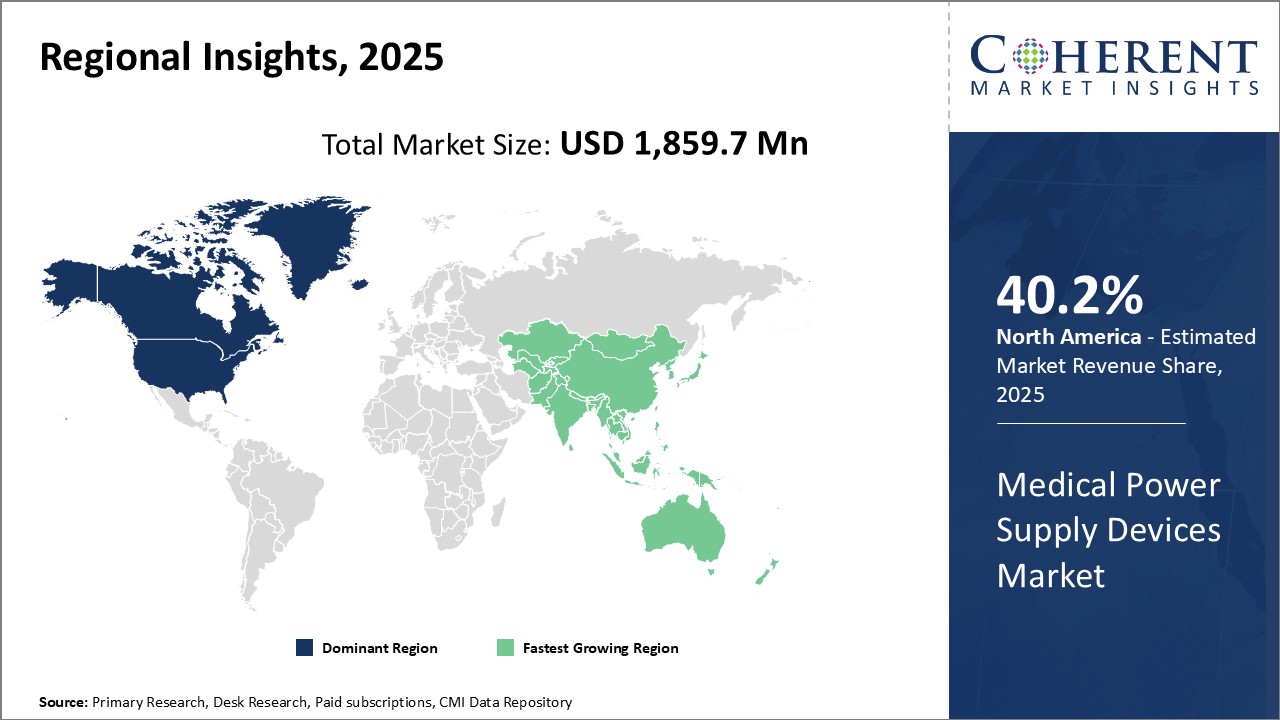
North America has established itself as the dominant region in the global medical power supply devices market. The region is expected to hold 40.2% of the market share in 2026. Being home to leading MedTech companies and having a presence of advanced healthcare infrastructure and facilities, North America enjoys strong industry presence. The region has seen significant technological advancements in recent years pertaining to safety of medical power supply devices. This has resulted in sophisticated devices with stringent quality standards becoming widely adopted.
Apart from the strong manufacturing base, North America also has a large consumer base due to accessibility of quality healthcare. This has further strengthened the demand for cutting-edge medical devices including power supply equipment. Several manufacturers in the region also export their products to international markets, augmenting regional exports. With high purchasing power of consumers, devices made for North American markets tend to be costlier as companies can charge premium prices.
The Asia Pacific region has emerged as the fastest growing market for medical power supply devices globally. Countries like China, India, Japan, and South Korea have witnessed tremendous expansion of their healthcare industries in the last decade. Rising medical tourism and growing focus on preventative healthcare have amplified the need for advanced medical devices across Asia Pacific. Being a vast region with over half the world's population, the potential for market expansion remains high.
The presence of leading global MedTech players through local manufacturing units has enhanced the availability of innovative products. This has positively impacted the adoption of technologically advanced power supply devices. Asia Pacific also gains from relatively lenient regulatory norms and low production costs, improving its cost competitiveness in international markets. Additionally, the growing export orientation of regional economies is a key factor driving the medical power supply devices market.
Medical Power Supply Devices Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 1,941.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 4.4% | 2033 Value Projection: | USD 2,624.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
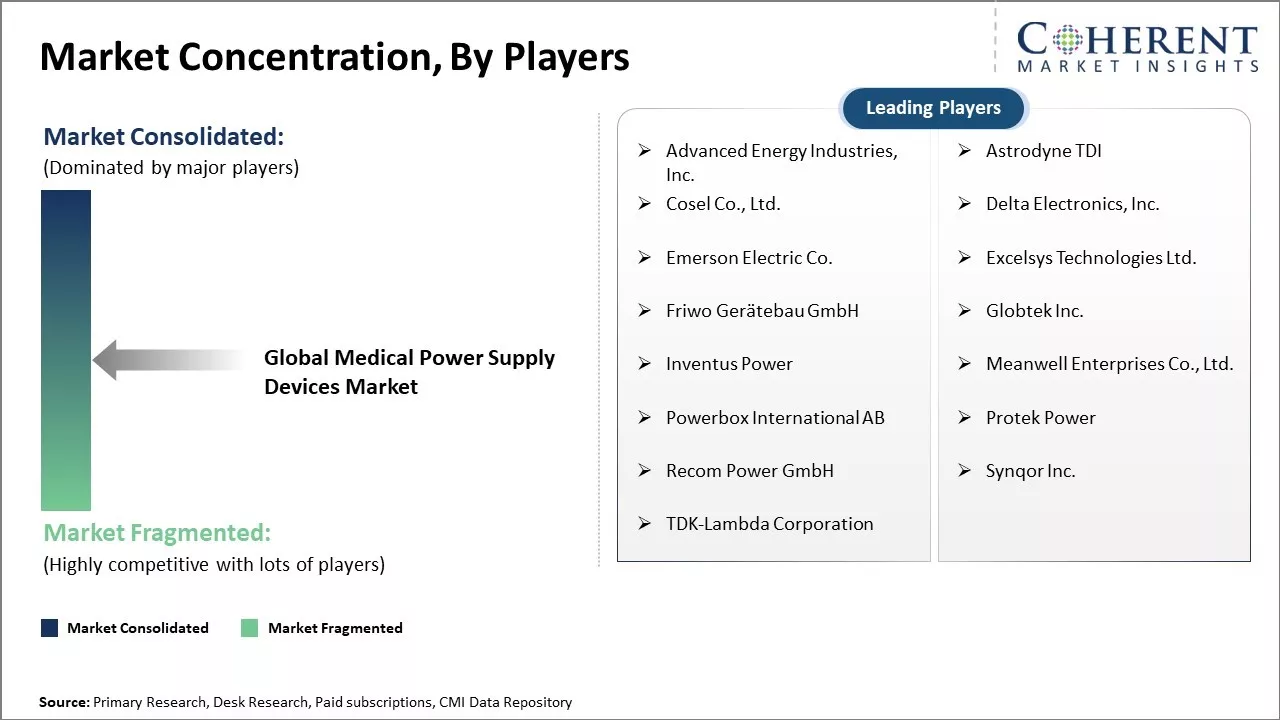
| Companies covered: |
Advanced Energy Industries, Inc., Astrodyne TDI, Cosel Co., Ltd., Delta Electronics, Inc., Emerson Electric Co., Excelsys Technologies Ltd., Friwo Gerätebau GmbH, Globtek Inc., Inventus Power, Meanwell Enterprises Co., Ltd., Powerbox International AB, Protek Power, Recom Power GmbH, Synqor Inc., and TDK-Lambda Corporation |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: The global medical power supply devices market consists of companies that manufacture and sell a variety of power supply devices that are used to provide regulated electrical power to medical equipment. This includes AC-DC and DC-DC converters, battery/charger assemblies, enclosures, and other ancillary components. The devices ensure medical equipment functions reliably and safely by regulating voltage fluctuations and providing stable, clean power. The market has seen steady growth in line with rising healthcare expenditures and demand for advanced medical technologies worldwide.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Suraj Bhanudas Jagtap is a seasoned Senior Management Consultant with over 7 years of experience. He has served Fortune 500 companies and startups, helping clients with cross broader expansion and market entry access strategies. He has played significant role in offering strategic viewpoints and actionable insights for various client’s projects including demand analysis, and competitive analysis, identifying right channel partner among others.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients