The blood based biomarker market was valued at USD 8.5 Bn in 2026 and is forecast to reach a value of USD 16.5 Bn by 2033 at a CAGR of 7% between 2026 and 2033.
The blood-based biomarker market is rapidly expanding, driven by several key factors that reflect broader shifts in healthcare and diagnostics. A major growth driver is the increasing prevalence of chronic and complex diseases such as cancer, cardiovascular disorders, and neurological conditions, which create demand for earlier, more precise detection methods that blood biomarkers can provide. Early disease detection and monitoring through minimally invasive blood tests is now a priority in clinical care, pushing adoption across hospitals, clinics, and research settings.
|
Current Event |
Description and its Impact |
|
Precision Medicine Initiatives and collaborations |
|
|
Regional Expansion in Alzheimer’s Disease Diagnostics |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
In terms of type, the diagnosis biomarker segment is expected to lead the market with 35.2% share in 2026. These biomarkers are widely used for early disease detection, particularly for serious conditions such as cancer, cardiovascular disorders, and neurological diseases. Early detection significantly improves treatment outcomes, guides clinical decision-making, and enhances overall patient care.
The strong clinical importance of diagnostic biomarkers, combined with rising global chronic disease prevalence and advancements in diagnostic technologies, continues to drive high demand in this segment.
For instance, according to the World Health Organization (WHO), approximately 70% of healthcare decisions are based on diagnostic test results, yet only 3–5% of healthcare budgets are allocated to diagnostic services. This imbalance highlights the critical role diagnostics play in healthcare systems worldwide. In many countries, clinicians lack access to basic diagnostic tools and laboratory tests necessary to support patients. Even where such services are available, vast geographic distances and isolated communities often make testing difficult, limiting timely diagnosis and treatment.
In terms of disease, the cancer segment is expected to hold 55.5% share of the market in 2026. Reflecting the critical role of biomarkers in early detection, monitoring, and treatment guidance. Blood-based biomarkers, such as circulating tumor DNA and tumor-associated proteins, allow non-invasive detection and real-time tracking of tumor progression, making them indispensable in oncology.
For instance, as per the National Library of Medicine, late-stage diagnosis remains a major challenge in lung cancer, with only 20–30% of cases detected early. Liquid biopsy technologies can complement imaging by identifying molecular changes indicative of malignancy, enabling earlier diagnosis and more effective treatment interventions. The adoption of these advanced diagnostic tools underscores the dominance of cancer in the blood-based biomarker market and highlights both the clinical importance and commercial demand for precise, minimally invasive testing.
In terms of application, the drug discovery segment is projected to account for 46.4% share of the market in 2026. Blood-based biomarkers allow pharmaceutical and biotechnology companies to monitor disease progression, treatment response, and patient stratification in a minimally invasive way, reducing the time and cost of developing new therapies.
To learn more about this report, Request Free Sample
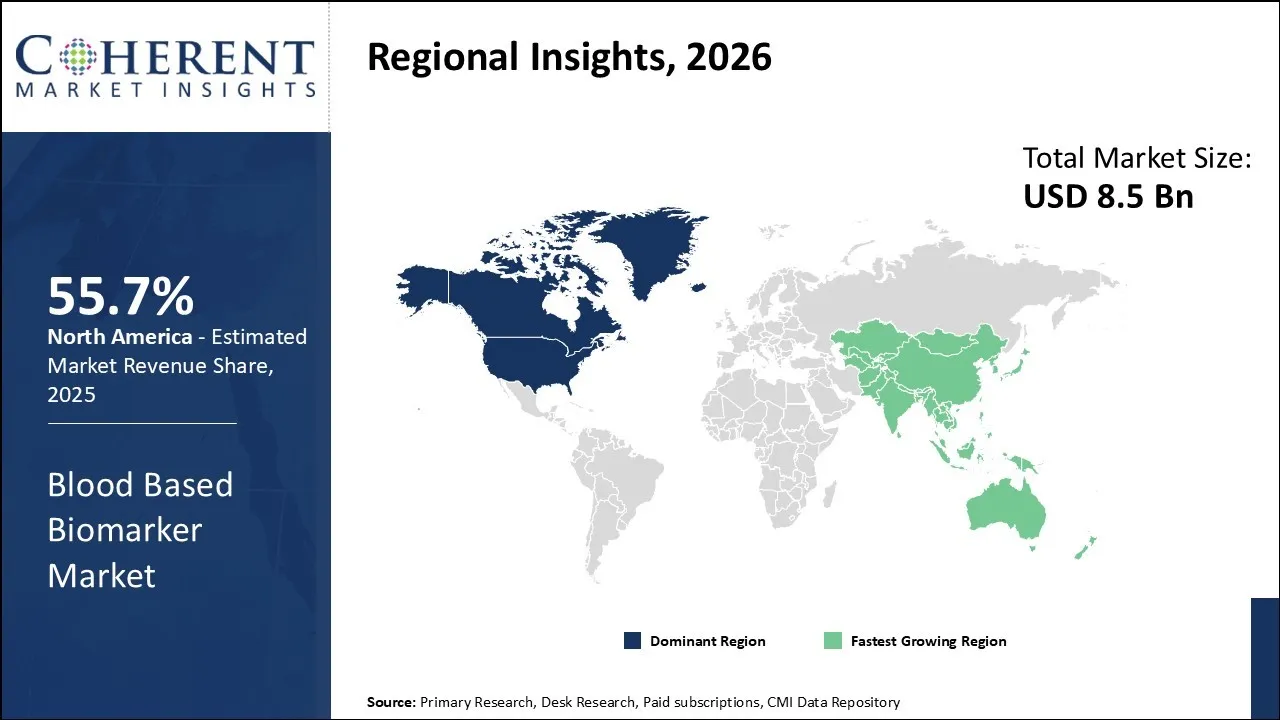
North America is expected to dominate the Blood Based Biomarker market with 55.7% share in 2026, driven by its concentration of major pharmaceutical and biotechnology companies, diagnostic laboratories, and research institutions that foster continuous innovation in biomarker discovery and validation. The presence of world-class medical centers across the United States and Canada has established sophisticated clinical pathways for integrating biomarkers into personalized medicine protocols, particularly in oncology and precision therapeutics.
For instance, according to a WIPO report published in 2025, North America, historically the global R&D leader, now accounts for around 28% of global research and development activity, followed by Europe (20%) and Central and Southern Asia (3.25%), highlighting the region’s continued strength in scientific innovation.
Asia Pacific is expected to exhibit the fastest growth, rising cancer incidence rates linked to aging populations, urbanization, lifestyle changes, and increased environmental exposures have created urgent demand for earlier detection and monitoring capabilities that blood-based biomarkers uniquely provide. Countries including China, India, and Japan are experiencing accelerated healthcare modernization, with government investments in diagnostic infrastructure and laboratory capabilities expanding access to advanced biomarker testing technologies.
For instance, in March 2025, Eisai, a pharmaceutical company based in Tokyo, Japan, announced that it will share its latest research on lecanemab (LEQEMBI), a treatment for Alzheimer’s disease, at the 2025 AD/PD™ International Conference in Vienna, Austria, and online from April 1–5. The company will present 16 research updates, including 6 oral presentations, covering lecanemab and other Alzheimer’s research. This highlights Asia’s contribution, as Eisai’s work in Japan is driving important developments in biomarkers and treatments for neurological diseases.
the US contributes the highest share in the blood-based biomarker market in the region owing to its advanced healthcare infrastructure, extensive research funding, and a well-established biotechnology and pharmaceutical industry. The US has a robust ecosystem that fosters innovation in molecular diagnostics, particularly in blood-based biomarker research, which is increasingly used for early disease detection, personalized medicine, and monitoring therapeutic responses.
For instance, initiatives such as the Cancer Biomarker Awareness Day hosted by the ACCC and the LUNGevity Foundation in November 2025 aim to educate clinicians and patients about the importance of cancer biomarkers. This highlights ongoing efforts in the US to increase biomarker adoption and awareness, supporting the country’s leading position in the blood-based biomarker market.
South Korea contributes the highest share in the blood-based biomarker market owing to its well-established healthcare infrastructure, strong emphasis on biotechnology innovation, and government-backed initiatives that promote precision medicine and early disease detection. South Korea’s robust investment in R&D, particularly in the genomics and diagnostics fields, has positioned it as a regional leader. partnerships and distribution agreements are helping expand access to advanced blood-based biomarker technologies.
For instance, in December 2025, Quanterix and Emocog entered an exclusive agreement in Korea to distribute the Simoa platform for blood-based Alzheimer’s diagnostics, enabling wider use of highly sensitive biomarker testing in clinical and research settings. This move highlights how collaboration between technology developers and local distributors is driving adoption, improving diagnostic capabilities, and supporting market growth in the region.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8.5 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7% | 2033 Value Projection: | USD 16.5 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Diadem srl., Proteomedix, Cleveland Diagnostics, F. Hoffmann-La Roche Ltd, GENFIT, Nutech Cancer Biomarkers India Pvt Ltd, SysmOex Corporation, Minomic, Creative Diagnostics, Eisai Co., Ltd., and C2N Diagnostics |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The rising prevalence of chronic diseases and cancer is a primary driver fueling the expansion of the blood-based biomarker market. Chronic ailments such as diabetes, cardiovascular diseases, and neurodegenerative disorders have seen a marked increase globally, largely due to aging populations, lifestyle changes, and environmental factors. Blood-based biomarkers offer a minimally invasive, cost-effective, and rapid diagnostic tool to detect and monitor these conditions early, enabling timely intervention and personalized treatment approaches.
The growing demand for early disease detection and diagnosis is significantly propelling the expansion of the blood-based biomarker market. Early detection of diseases such as cancer, cardiovascular ailments, and neurodegenerative disorders is critical in improving patient outcomes by enabling timely intervention. Blood-based biomarkers offer a minimally invasive, cost-effective, and accessible approach to detect biochemical changes in the body long before symptoms manifest, revolutionizing traditional diagnostic paradigms.
The integration of artificial intelligence (AI) and machine learning (ML) in biomarker discovery represents a transformative opportunity within the blood-based biomarker market by significantly accelerating the identification and validation process of novel biomarkers. Blood-based biomarkers offer a minimally invasive and cost-effective solution for early diagnosis, prognosis, and therapeutic monitoring across numerous diseases, including cancer, cardiovascular disorders, and neurodegenerative diseases.
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients