The global budesonide inhaler market size is estimated to reach approximately USD 8,709 Mn in 2026 and is projected to grow at a (CAGR) 6% during the forecast period (2026-2033), reaching by USD 13,099 Mn in 2033.
The global budesonide inhaler market is expected to grow steadily during the forecast period. This is mostly due to rising incidence of asthma, allergic rhinitis, and other respiratory diseases, growing demand for effective and fast-acting inhalation therapies, and advancements in inhaler device technologies.
Budesonide is a synthetic anti-inflammatory glucocorticoid widely used in the treatment of asthma and allergic rhinitis. As an inhaled corticosteroid, it reduces airway inflammation and helps prevent asthma symptoms and exacerbations. Thus, rising prevalence of respiratory disorders and increasing preference for inhalation-based drug delivery systems are expected to drive demand for budesonide inhalers during the forecast period.
For example, a recent 2025 clinical study found that a combination inhaler with budesonide and formoterol lowered asthma attacks in children by almost 45% compared to standard reliever inhalers. This shows the growing clinical preference for budesonide-based inhalers and is likely to boost market growth in the coming years.
Rising popularity of combination therapies is unlocking new revenue streams for budesonide inhaler manufacturers. Budesonide is being increasingly formulated with bronchodilators like formoterol or salmeterol) or incorporated into multi-component inhalers to improve therapeutic efficacy and patient convenience. For instance, Viatris launched Breyna (budesonide and formoterol fumarate dihydrate) inhalation aerosol.
|
Current Event |
Description and its Impact |
|
Healthcare System Transformations |
|
|
Technological Innovation and Competition |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on product type, budesonide inhalants segment is estimated to account for the highest market share of about 62% in 2026. This is mostly due to the widespread adoption of budesonide inhalers for the treatment of asthma and chronic obstructive pulmonary disease (COPD), as well as their proven efficacy, ease of use, and convenience for patients.
Inhalers deliver the drug directly to the lungs, improving treatment results while reducing side effects in the rest of the body. Patients prefer inhalers because they are portable, easy to carry, and can be used anytime, making them ideal for daily management of respiratory conditions. The convenience, fast relief, and effectiveness of inhalers compared to other forms, like tablets or nebulizers, continue to keep them popular in the budesonide inhaler market.
By dosage form, dry powder segment is slated to lead the budesonide inhaler industry, accounting for a market share of 50% in 2026. This is mostly due to its ease of use, improved patient compliance, and better chemical stability compared to other forms.
Dry powder inhalers do not require propellants. This is making them more environmentally friendly and convenient for patients. In addition, the consistent dosing and portability of dry powder inhalers make them a preferred choice for healthcare providers and patients, leading to higher adoption rates worldwide.
To learn more about this report, Request Free Sample
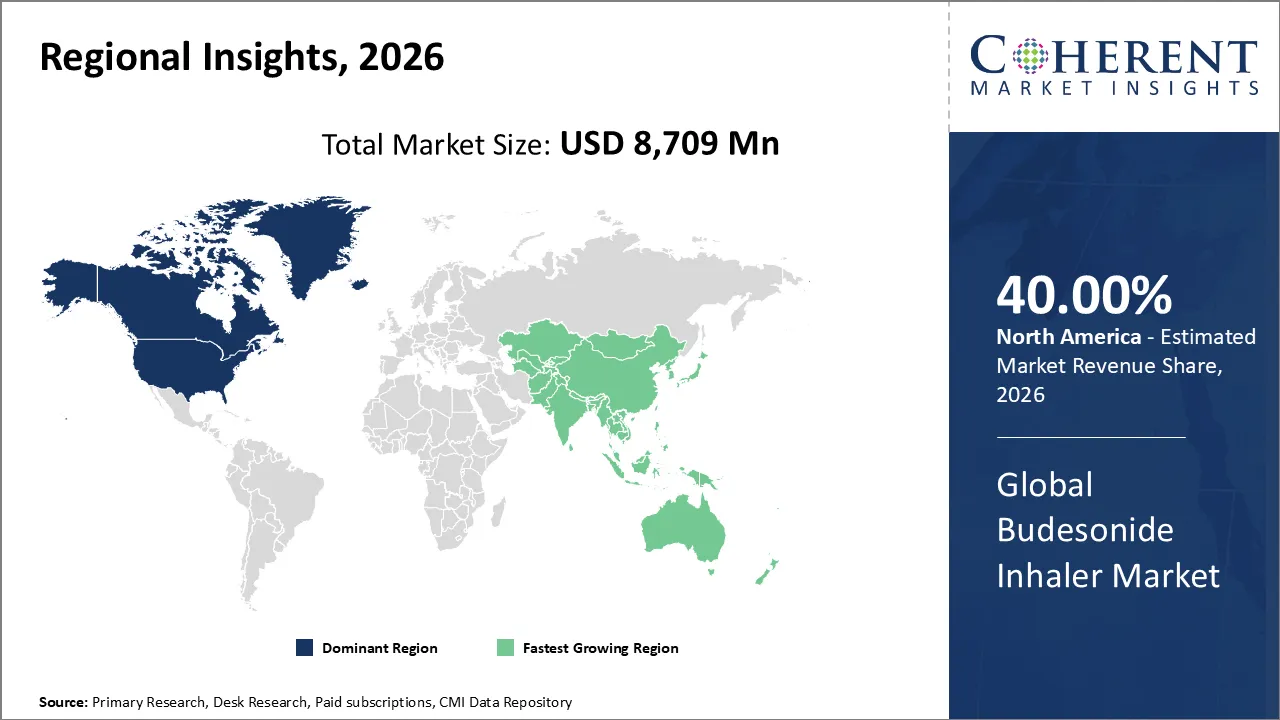
North America is expected to retain its dominance with an estimated share of about 40% in 2026. This is mostly due to high prevalence of respiratory conditions such as asthma and COPD, strong presence of leading pharmaceutical companies, and growing adoption of advanced inhaler technologies in the region.
According to Asthma and Allergy Foundation of America, Asthma remains one of the most common chronic diseases in the United States, affecting more than 28 million people. This high asthma burden is expected to fuel demand for budesonide inhalers in the region during the forecast period. Ongoing research and development by major pharmaceutical companies is paving way for the development of innovative inhaler formulations, further strengthening market growth.
Asia Pacific, spearhead by China and India, is expected to emerge as the most lucrative market for manufacturers of budesonide inhalers during the assessment period. This can be attributed to increasing patient pool, rising popularity of generic budesonide inhalers, favorable government policies, and presence of large contract manufacturing organizations.
Growing urban populations, rising air pollution levels, and increasing access to healthcare are driving regional demand. In addition, strategic partnerships between local manufacturers and global pharmaceutical players are improving production capabilities, making the region very attractive for market expansion.
The U.S. is anticipated to hold a prominent share in the global budesonide inhaler market during the forecast period. This is mostly due to growing incidence of chronic respiratory diseases and rising popularity of advanced inhaler technologies like dry powder inhalers (DPIs) and metered-dose inhalers (MDIs) with better delivery systems. Government initiatives to improve respiratory health, such as funding for asthma management programs and COPD awareness campaigns, are also supporting market growth.
Shift towards preventive care is a key growth-shaping trend in the U.S. budesonide inhaler market. There is a rising emphasis on preventive treatments in the nation as people look to avoid severe exacerbations. This, in turn, is increasing long-term maintenance inhaler usage, including budesonide formulations.
Budesonide inhaler demand in China is expected to increase rapidly during the assessment period. This is due to increasing cases of asthma and other respiratory disorders, especially in urban regions affected by air pollution. Expansion of healthcare infrastructure, rising health insurance coverage, and government policies promoting access to essential medicines are fueling market adoption.
Local pharmaceutical manufacturers in China are increasingly investing in R&D to produce cost-effective inhaler solutions. Similarly, increasing patient awareness regarding preventive respiratory care is positively influencing sales of budesonide inhalers in China.
India’s Budesonide inhaler market is expected to expand steadily, owing to increasing burden of respiratory diseases and rising pollution levels in urban centers. Growing awareness about asthma management, improved access to primary healthcare, and affordability of generic Budesonide inhalers are key drivers. In addition, initiatives by the government and NGOs to improve respiratory health awareness are further stimulating market demand.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 8,709 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6% | 2033 Value Projection: | USD 13,099 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Pfizer Inc., Cipla Limited, Lupin Limited, Cosmo Pharmaceuticals, Takeda Pharmaceutical Company Ltd., Manus Aktteva Biopharma LLP, Abbott Laboratories, Lunan Better Pharmaceutical, Novartis International AG (Sandoz), Mylan N.V., Skyepharma, AstraZeneca Plc., Chiesi Farmaceutici S.p.A, Orion Corporation, Santarus Inc., Synmosa Biopharma Corporation, and Shanghai Sine Pharmaceutical Laboratories Co. Ltd. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The global prevalence of chronic respiratory conditions like asthma and COPD (chronic obstructive pulmonary disease) is increasing globally due to factors such as pollution, smoking, and urbanization. For instance, according to the World Health Organization (WHO), global COPD cases will increase by about 23% through 2050. This growing disease burden is a major demand driver for budesonide inhalers used to control airway inflammation and manage symptoms.
Patients and clinicians in the contemporary world are increasingly becoming aware of respiratory health and the importance of early disease detection. This is supporting the use of inhaled corticosteroids like budesonide as a first-line therapy. In particular, governments in emerging markets such as India and China are launching awareness campaigns and initiatives to address the burden of respiratory diseases and improve access to diagnostics and treatments. These efforts are expected to create a favorable environment for the growth of budesonide inhaler market during the forthcoming period.
Expiry of key patents is paving way for the entry of more generic budesonide inhalers. For instance, Iconovo announced plans to commercialize its generic formoterol/budesonide (a Symbicort equivalent) as part of its 2025 product focus. These generic solutions are making the budesonide therapy more affordable as well as accessible, especially in emerging markets. This broadens the patient base and increases overall market volume.
Advancements in inhalation technologies are creating lucrative growth opportunities for manufacturers of budesonide inhaler market. Smart inhalers with digital tracking and adherence support are gaining traction, especially in developed nations like the U.S., France, and the U.K. These devices can improve patient compliance as well as outcomes. Similarly, advanced inhalers such as breath-actuated devices, dose counters, and those with connectivity features are enhancing drug delivery and patient engagement.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients