Ciclesonide Market - Insights
Ciclesonide, are used to prevent the difficulty in breathing, chest tightness, wheezing, and coughing caused by asthma in adults and children 12 years of age and older. It works as a being class of corticosteroid medication to decrease swelling and irritation in the airways allowing easy breathing. Ciclesonide comes in aerosol and nasal spray, and usually need to inhale twice a day. It help to prevent the asthma attack including sudden episodes of shortness of breath, wheezing, and coughing.
A study findings published by the Asthma and Allergy Foundation of America (AAFA) in October 2015 revealed that, air pollution can worsen existing asthma symptoms. According to same source, a study of young campers with moderate to severe asthma reported that, they were 40% more likely to have acute asthma episodes on high pollution summer days as compared to days with average pollution levels. Pollution is common cause of asthma in people, and people face various kinds of pollutants generated through traffic fumes, wood smoke, dust, smoke, and diesel fumes, which make airways inflamed and swollen, triggering asthma symptoms.
The global ciclesonide market size is estimated to be valued at US$ 565.4 million in 2018 and is expected to witness a CAGR of 1.9% over the forecast period (2018–2026).
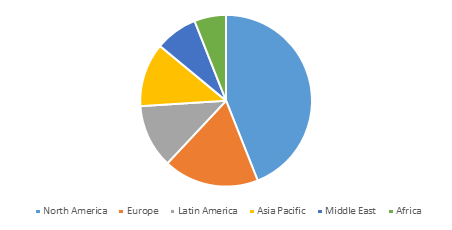
Global Ciclesonide Market Share Analysis (US$ Mn), by Region, 2018
To learn more about this report, Request Free Sample
Source: Coherent Market Insights Analysis (2018)
High Prevalence of Allergy Rhinitis and Asthma is Expected to Augment the Market Growth
Ciclesonide is widely used in the treatment of asthma and allergy rhinitis. Allergy rhinitis and asthma are very common conditions and prevalence of these two disease is very high in countries such as the U.S., U.K., China, and India. Hence, high prevalence of allergy rhinitis is expected to drastically increase demand for ciclesonide and drive its market growth over forecast period. According to the Global Asthma Report published in 2018, an asthma kills around 1,000 people every day and affects as many as 339 million people worldwide. Prevalence of asthma is rising and people in low- and middle-income countries suffer the most.
According to the data published by National Center for Biotechnology Information (NCBI), in January 2018, prevalence of allergy rhinitis in Asia, Europe, the Americas, and Africa region was around 15%–25% among children and adolescents, as well as young adults.
Global Ciclesonide Market Share (%), By Dosage Form: 2018 & 2026
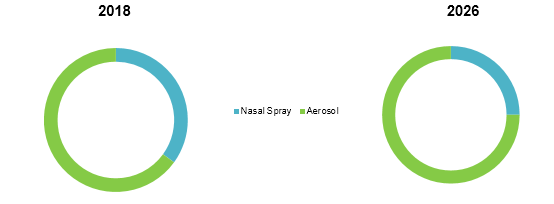
To learn more about this report, Request Free Sample
Source: Coherent Market Insights Analysis (2018)
Ciclesonide is novel inhaled corticosteroids, majorly preferred by asthma and COPD patients, to relive the underlying symptoms. This generates great opportunity for key players in the market to launch ciclesonide-based products.
Key players have opportunities to penetrate in the untapped potential markets where prevalence of allergy rhinitis and asthma is very high and the accessibility of ciclesonide is less due to the low presence of market players. According to Global Asthma Report published in 2018, in some countries of Asia Pacific such as China, India, Indonesia, Malaysia, and Thailand, burden of asthma is substantial, however, asthma remains underdiagnosed and undertreated. According to the same source, many asthma patients are not using inhaled corticosteroids, as these medicines are either inaccessible or unaffordable. Therefore, key players could focus on these markets and fulfill the unmet need of these potential regions to generate high market revenue.
However, presence of alternative mediation and possible risk of pneumonia and other respiratory infections among asthma children who use Safety of inhaled corticosteroids (ICS), may hinder growth of the ciclesonide market.
Ciclesonide is indicated for the treatment and management of allergy rhinitis and asthma. However, availability of some other medication offering better treatment regime are indicated for the treatment of allergy rhinitis and asthma. This is expected to create competition and hinder the adoption of ciclesonide over the forecast period. For instance, GlaxoSmithKline Plc’s drug, namely Veramyst (fluticasone furoate) Nasal Spray is indicated for the treatment of seasonal and perennial allergic rhinitis. NASONEX (mometasone furoate monohydrate) Nasal Spray by Merck & Co., Inc. is indicated for treatment of nasal symptoms of allergic rhinitis.
Furthermore, patent expiration and subsequent launch of generic ciclesonide in the market could hamper growth of ciclesonide in the near future.
With the expiry of branded ciclesonide products such as OMNARIS Nasal Spray and ALVESCO it is expected that generic manufacturers will enter the market. Launch of generic versions of ciclesonide will increase competition and will restrain growth of the market in terms of revenue as generic products are available in less price. For instance, according to the data published in Sepracor Inc. annual report, OMNARIS Nasal Spray and ALVESCO HFA lost their patent in October 2017. Thus, patent lose can affect and hinder the market growth in terms of revenue.
North America is expected to hold a dominate position in global ciclesonide market in 2017, owing to high prevalence of allergy rhinitis and asthma conditions and increasing drug launches in this region. For instance, in 2012, Sunovion Pharmaceuticals Inc. received the U.S. Food and Drug Administration (FDA) approval for its Zetonna (ciclesonide) nasal aerosol for the treatment of allergic rhinitis and launched it in the U.S. market. Furthermore, according to the data published by American Academy of Allergy Asthma & Immunology, in 2016 around 8.3% of children in the U.S. suffered from asthma.
Some of the key players operating in the ciclesonide market include AstraZeneca Plc., Cipla Limited, Sun Pharmaceutical Industries Ltd., and Apotex Inc.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients