The Cold, Cough, and Sore Throat Remedies Market is anticipated to grow at a CAGR of 3.5% with USD 45,300.1 Mn in 2026 and is expected to reach USD 56,500.2 Mn in 2033. The cold, cough, and sore throat medicines market is growing because more people are getting the flu and the common cold, and the world's population is getting older. WHO says that about 1 billion people get seasonal flu every year, with 3–5 Mn of those cases being severe and 290,000–650,000 people dying from respiratory problems. The CDC reported that adults in the U.S. get an average of two to three colds a year. The World Health Organization (WHO) also stats that the number of people over 60 will grow from 1.1 billion in 2023 to 1.4 billion by 2030. This will increase the need for goods that help with respiratory symptoms.
Source: USFDA
Source: CDC
Cold and congestion firms are using technology to improve their delivery systems, which is helping them provide a better user experience and move into related areas like nasal sprays and mists. Better dispensing systems can make things more comfortable, easier to use, and better for kids, which helps brands become more premium and pharmacies sell more. Businesses are embracing these new ideas to move beyond regular tablets and syrups into more valuable forms. This raises the value of both the product mix and the category penetration.
Increasing adoption of organic growth strategies like marketing & brand development has estimated to drive the cold, cough and sore throat remedies market growth in the near future. In this sector, marketing is evolving away from mass advertising and toward science-based doctor involvement and consumer education. This makes the product more trustworthy, boosts the number of recommendations, and gives customers more faith in their choice of branded cough and throat medicines. These kinds of programs also assist corporations change how people treat themselves, especially in markets where people often treat themselves and pharmacists and doctors have a say in what products are available. So, promotion based on awareness immediately helps with faster adoption and higher recurrent demand.
Source: Kenvue Inc.
Rising launch of the new products and innovation of new drug formulation has accelerated the growth of the market size over the forecast period. The cold, cough, and sore throat remedies market size is growing rapidly because of more product launches and better delivery methods. These give customers more targeted, handy, and unique ways to acquire relief. Companies are coming out with new treatments including soft nasal mists, spray-based formats, and over-the-counter (OTC) respiratory therapies that target specific symptoms and make the experience better for users. This helps brands get more shelf space, bring in new customers, and encourage the growth of premium products in the self-care sector. Innovation also makes the market more competitive by adding new claims, formats, and symptom-focused positioning to older remedy categories.
For instance, in March 2026, Glenmark Pharmaceuticals Limited (Glenmark), a research-led, global pharmaceutical company, revealed that the U.S. Food and Drug Administration (U.S. FDA) has given Glenmark Pharmaceuticals Limited (Glenmark) final approval for Fluticasone Propionate Nasal Spray USP, 50 mcg per spray (OTC) and launched for commercial sale on April 1, 2026. The U.S. FDA has decided that Glenmark's Fluticasone Propionate Nasal Spray USP, 50 mcg per spray (OTC) is bioequivalent to the reference listed drug, Flonase Allergy Relief Nasal Spray, 50 mcg per spray, from Haleon U.S. Holding LLC [NDA - 205434]. Glenmark Therapeutics Inc. will sell it in the U.S.
According to According to Nielsen's syndicated statistics for the 52-week period ending March 3, 2026, the Flonase Allergy Relief Nasal Spray, 50 mcg per spray market made over US$384.7 million in sales in 2016.
|
Current Event |
Description and its Impact |
|
Regulatory Shift & Reformulation
|
|
|
Increasing Government Initiatives and Support |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Antihistamines are projected to account for the largest share of cold, cough, and sore throat remedies market in 2026, representing approximately 27.5% of the total volume. The cold, cough, and sore throat remedies products are extensively utilized to relieve sneezing, watery eyes, runny nose, and throat irritation, especially in cold-allergy overlap cases. Their dominance is helped by the ongoing respiratory illness burden in 2026: the U.S. CDC predicted at least 31 million flu cases, 370,000 hospitalizations, and 23,000 deaths by April 4, 2026. NHS Pharmacy First reported 1,757,366 acute sore throat consultations through December 2025, with 138,808 of those happening in December 2025 alone.
Two major factors are supporting the antihistamines segment growth in the market. First, the high seasonal burden of flu, colds, and allergy-like respiratory symptoms continues to support robust demand for OTC symptom-relief medicines, particularly treatments that address sneezing, rhinorrhea, and upper-airway irritation. Second, the fact that antihistamines are easy to get over the counter and that people prefer to take care of themselves means that they will buy them again from pharmacies and stores. Pharmacy-led care models are also making it easier for people to get over-the-counter respiratory medications that don't require a prescription. The FDA's cold, cough, allergy, bronchodilator, and antiasthmatic monograph framework continues to ensure category stability and trust.
For instance, on April 3, 2026, The U.S.FDA required a new warning for cetirizine and levocetirizine about rare, severe itching after long-term use, which shows that they are actively keeping an eye on the situation.
Futhermore, in February 2025, Pharmascience Inc., pharmaceutical company based in Canada revealed the introduction of pms-RUPATADINE (rupatadine), a therapy that helps with both nasal and non-nasal symptoms of seasonal allergic rhinitis (SAR) and perennial allergic rhinitis (PAR), as well as symptoms of chronic spontaneous urticaria (CSU).
To learn more about this report, Request Free Sample
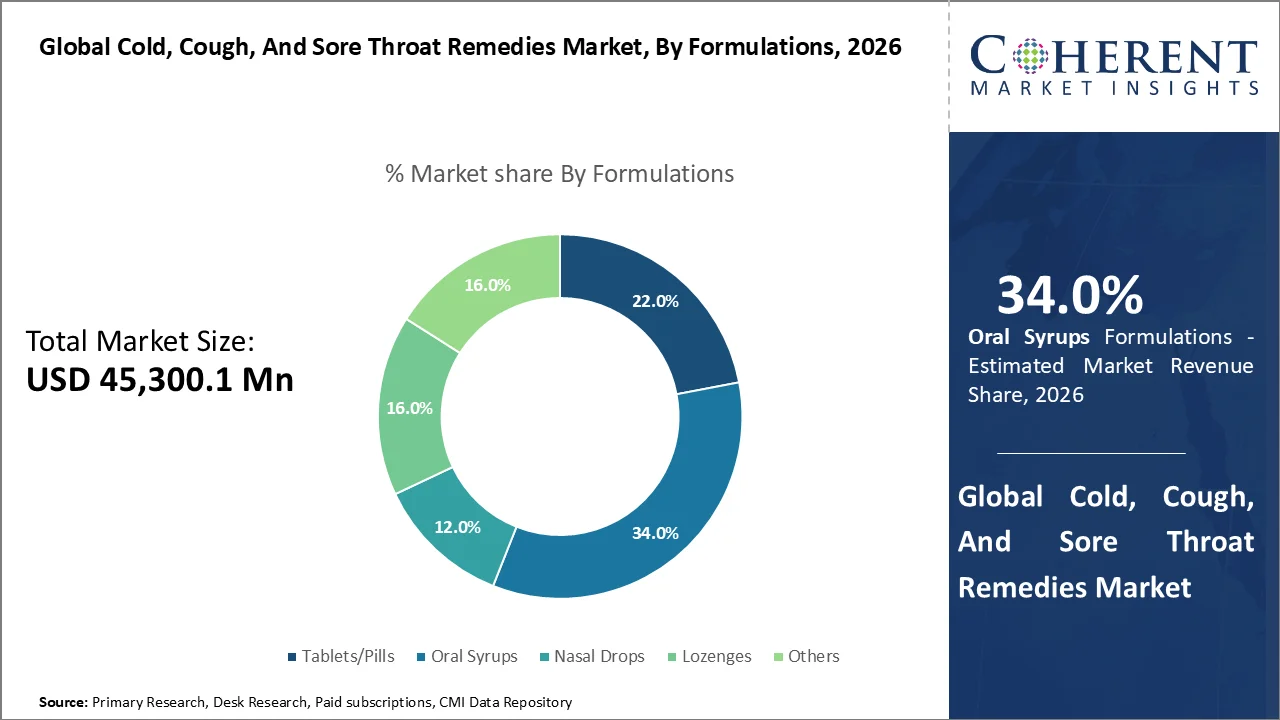
Based on formulation, oral syrups dominate the market, accounting for a significant 34.0% share in 2026, owing to ease of administration, palatable taste & better patient compliance. Oral syrups dominate the market because they are easy to swallow, suitable for both adults and children, and widely used for multi-symptom relief such as cough, congestion, fever, and throat irritation. Their importance is still significant in 2026, when there are a lot of respiratory diseases. The U.S. CDC stated that the flu season from 2025 to 2026 caused at least 47 million illnesses, 610,000 hospitalizations, and 26,000 fatalities by April 2026. NHS Pharmacy First, on the other hand, reported 1,757,366 acute sore throat consultations through December 2025.
Two important factors are helping the oral syrups segment grow. First, syrups are still the most popular choice for kids and families since it's easier to give them to those who have trouble swallowing and for symptom-based dosing than tablets or capsules. Second, producers still use syrups as a main way to give several symptoms, such cough, cold, throat, and nasal congestion relief, all in one product. This makes people want to buy them again. Regulatory and quality oversight also maintains this area commercially significant. For example, the FDA added import limitations for liquid cough and cold medicines in January 2026, showing how big and crucial this type of medicine is.
For instance, on January 21, 2026, U.S. Food and Drug Administration announced that imported liquid cough and cold medicines from India at risk of DEG/EG contamination should be accompanied by test results, showing direct regulatory focus on syrup products. Earlier, Haleon said in its February 27, 2025 full-year results that it launched Theraflu-D Flu Relief Max Strength Syrups in the U.S. with pseudoephedrine.
Drug stores account for the largest share of 38.0% in 2026 because they offer customers easy access to products, advice from pharmacists, and excellent visibility for over-the-counter products all in one place. The size of pharmacy networks and the high demand for respiratory care support their leadership. For instance, in February 2026, National Association of Chain Drug Stores (NACDS) reported that chain members run more than 40,000 pharmacies and fill more than 3 billion prescriptions a year in the U.S. NHSBSA reported 82,212 acute sore throat consultations in December 2025 alone and 9,274 community pharmacies claiming for that pathway, which shows how important pharmacy-led access is.
Source: NACDS
There are two main factors that are helping the drug store category grow. First, a lot of people get sick with respiratory ailments, so they go to pharmacies to get quick relief from their symptoms. The CDC said that by the week of April 4, 2026, there had been at least 31 million flu cases, 370,000 hospitalizations, and 23,000 deaths in the U.S. Second, this channel is getting stronger since pharmacies are now offering consultations, testing, treatment assistance, and medicine supply for common respiratory illnesses. NHS Pharmacy First has seven clinical routes, one of which is for sore throats. Drug stores also benefit from great trust, the ability to go in and buy something, and pharmacist-led over-the-counter recommendations at the time of purchase.

To learn more about this report, Request Free Sample
Asia Pacific account 38.0% market share in 2026, supported by shift towards herbal & natural products, growing popularity of immune boosting products and strong industry presence. Asia Pacific is the largest consumer base for cold, cough, and sore throat remedies. This is because it has a lot of people, a lot of respiratory illnesses, and more access to self-care drugs. According to the World Health Organization (WHO), seasonal flu affects about 1 billion people around the world each year. The WHO's Western Pacific bulletin also stated that flu infections were on the rise in some parts of Asia during the 2025 respiratory season. The Indian government's IDSP influenza surveillance network is still in place to keep an eye on seasonal flu. This shows that there is a big demand for over-the-counter cough, cold, and sore throat medicines in major Asia Pacific markets.
The North America is poised to be as the fastest-growing region through 2026-2033, owing to because there are a lot of over-the-counter drugs available, pharmacies are easy to get to, and respiratory viruses keep people buying cough, cold, and sore throat medicine. According to the Centers for Disease Control and Prevention, during the week of April 4, 2026, at least 31 million Americans had contracted the flu, 370,000 had visited hospitals, and 23,000 had died from it during the 2025–2026 season. In addition, NACDS claims that its members fill over 3 billion prescriptions annually and manage over 40,000 pharmacies. For this reason, pharmacy-led access keeps increasing local sales of products and cures.
The India cold, cough, and sore throat remedies market is undergoing a period of robust expansion. India's large population, growing self-medication trend, and government-funded influenza surveillance system make it a crucial nation in Asia Pacific. Seasonal monitoring is supported by the Government of India's Integrated Disease Surveillance Programme influenza surveillance network, and public health tracking is strengthened by ICMR's ongoing work on infectious diseases and respiratory conditions. For cough syrups, sore throat lozenges, antihistamines, and over-the-counter cold remedies, India is a significant market.
Source: IDSP
The U.S. cold, cough, and sore throat remedies market are projected to grow significantly during the upcoming years. Due to its sizable over-the-counter market, vast pharmacy network, and high prevalence of recurrent respiratory illnesses, the United States is the most significant nation in North America. By early April 2026, the CDC predicted that there will be at least 31 million flu-related illnesses, 370,000 hospital admissions, and 23,000 fatalities. Despite this, the chain pharmacy infrastructure is still quite robust thanks to national retail networks. This facilitates quick product transfer across pharmacies, supermarkets, and internet platforms.
Some of the major key players in Reckitt Benckiser Group PLC, Johnson & Johnson Services, Inc., GlaxoSmithKline plc., Novartis AG, Bayer AG, AstraZeneca plc., Sun Pharmaceutical Industries Ltd., Pfizer Inc., Prestige Brands Holdings, Inc., and Procter & Gamble Co.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 45,300.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 3.5% | 2033 Value Projection: | USD 56,500.2 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Reckitt Benckiser Group PLC, Johnson & Johnson Services, Inc., GlaxoSmithKline plc., Novartis AG, Bayer AG, AstraZeneca plc., Sun Pharmaceutical Industries Ltd., Pfizer Inc., Prestige Brands Holdings, Inc., and Procter & Gamble Co. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
[CDC]
[WHO]
[CDC]
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients