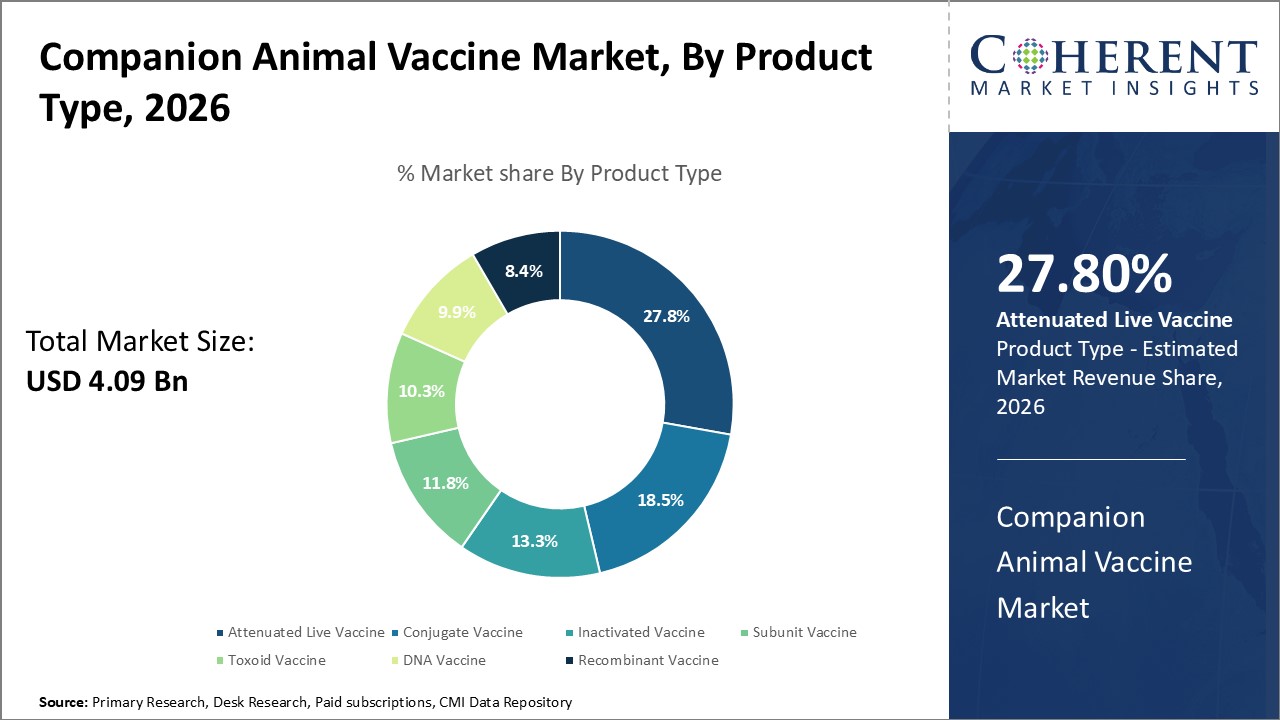
The Companion Animal Vaccine Market is estimated to be valued at USD 4.09 Bn in 2026 and is expected to reach USD 6.56 Bn in 2033, exhibiting a compound annual growth rate (CAGR) of 7.0% from 2026 to 2033.
The Companion Animal Vaccine market is an emerging market that focuses on the manufacturing of vaccines that can be effective in protecting domesticated animals, primarily pets like cats and dogs, from diseases like rabies. It is rapidly growing, owing to a rise in pet ownership, awareness regarding pet health, and the surge in diseases like rabies, distemper, etc.
|
Current Events |
Description and its impact |
|
Advancements in Vaccine Technology and Regulatory Responses |
|
|
Regional Disease Outbreaks Impacting Canine Populations |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Pet insurance companies have started covering core vaccines for dogs and cats, such as rabies, parvovirus, and distemper, particularly under wellness plans or preventive care add-ons. The coverage typically includes the cost of the vaccine itself, as well as the office visit fee. Trupanion, one of the largest pet insurers in North America, offers reimbursement for vaccines as part of their wellness packages. Pets Best provides a Preventative Care Add-On which covers vaccines as part of a broader wellness plan, including dental cleanings and flea treatments. The ASPCA has also contributed to vaccine reimbursement programs by offering low-cost or free vaccines through clinics in underserved areas. Pop-up vaccination clinics are becoming a more common model, and reimbursement for these services may be covered by either government grants or non-profit funding sources to offset veterinary costs.
As of February 2025, a total of 110.49 crore doses have been administered for FMD, 4.57 crore doses for Brucellosis, 25.36 crore doses for PPR, and 0.70 crore doses for CSF in India.
Based on the Species Type, the Feline segment is dominating the market, accounting for around 37.2% of the global market share. The growth of this segment is attributed to the increasing cat ownership across the globe. The Feline category among animals is said to be susceptible to many infections and diseases. This increases the need to get them vaccinated from time to time. For instance, according to a study conducted by Simon F. Haeder, Associate Professor of Public Health, Texas A&M University, in 2024, there are around 4% of dogs and 12% of cats remain unvaccinated against rabies. Around 22% of dog owners and 26% of cat owners are hesitant to get their pets vaccinated. This further propels the companion animal vaccine maret demand.
To learn more about this report, Request Free Sample
On the basis of Product Type, the Attenuated Live Vaccine segment is expected to lead the market in 2026. These vaccines are known for their ability to induce long-lasting immunity. It comprises weakened pathogens that replicate without causing any disease. It is not only cost-effective but is also efficient and is widely adopted in veterinary practices.
In August 2024, Texas-based Low Cost Pet Vax launched its first pop-up vaccination clinics in Miami, Florida. These walk-in clinics, operating on weekends at local business parking lots, offer veterinary-supervised services.
Based on the Distribution Channel, the Veterinary Clinics segment is leading the market. Veterinary clinics act as the first point of contact for pet owners seeking preventive care. Many services are offered by these clinics, such as personalized care, follow-up services, etc., which drive the vaccination compliance. Additionally, there are also collaborations between veterinary clinics and vaccine manufacturers that help in ensuring the availability of vaccines easily and at all times. However, post-pandemic, the consumer behaviour has changed, with rising costs, etc., having taken a toll on the business sustainability. In September 2024, the ASPCA opened its third Community Veterinary Clinic. The clinic aims to provide affordable veterinary care to thousands of pets in underserved areas, improving the health and well-being of local dogs and cats.
To learn more about this report, Request Free Sample
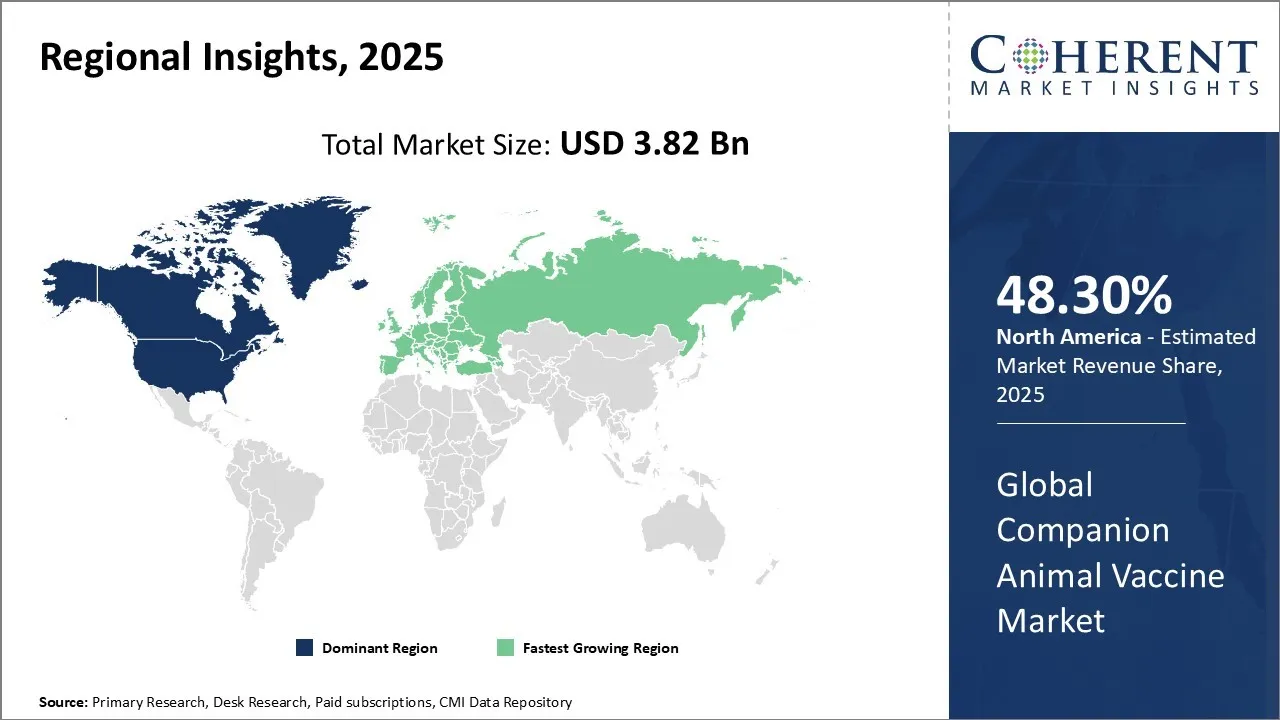
Among regions, North America is estimated to hold a dominant position in the global companion animal vaccine market over the forecast period. North America is estimated to hold 48.3% of the market share in 2026. The global companion animal vaccine market is expected to witness significant growth in the coming years, driven by the high prevalence of orthopedic disorders and dysfunctions, favorable health reimbursement, and increased awareness. The increasing prevalence of neurological disorders is contributing to the growth of the companion animal vaccine market in the North America region.
The U.S. and Canada are the top two countries contributing significantly to the growth of the Companion Animal Vaccine Market of the region. The U.S. is leading the region, owing to its increased efforts in Research & Development. In February 2024, the University of Pennsylvania School of Veterinary Medicine (Penn Vet) launched its mRNA Research Initiative to accelerate the development of veterinary mRNA vaccines and host-directed therapies. The new product launches and approvals of vaccines are expected to increase the number of vaccines produced in the U.S., thereby driving demand for veterinary services in the U.S. Meanwhile, Canada also follows the same trends of pet adoption, and along with it an increase in demand for advanced veterinary vaccines, also drives the market’s growth in Canada.
Europe is the second-largest market. The growth here can be attributed to a variety of different factors, such as widespread pet ownership, a well-established veterinary healthcare infrastructure. The focus of the market in this region is on preventive care, which is also backed by a few government initiatives and pet welfare organizations. Moreover, the presence of several leading pharmaceutical companies also helps the market grow. The two countries that play a pivotal role in shaping the market include Germany and the U.K. Germany is dominating the market in this region due to its large pet population and considerable investment in animal welfare. However, the U.K., too, has a very well-established pet healthcare system, which significantly contributes to the companion animal vaccine market growth.
In November 2024, Ceva Animal Health announced its investment in European vaccine manufacturing with the construction of a new facility in Hungary, boosting the capacity of Ceva Phylaxia.
Introduction of companion animal vaccine with advanced technology in the market is expected to drive the growth of the global companion animal vaccine market over the forecast period. The integration of advanced technologies such as recombinant DNA, mRNA, etc. is leading the market to a new era with lots of innovations on the way. These innovations are expected to improve the efficiency of vaccines, extend the duration of immunity, and reduce side effects. The rise in pet ownership is also significantly contributing to the increase in demand for vaccines, which in turn leads to the development of new and efficient technologies. For instance, in November 2021, Stryker Corporation, a multinational medical technology corporation based in the U.S., launched the Prophecy Infinity Resect-Through Guides for use in total ankle replacement surgeries. The system represents the refinement of Stryker’s industry gold standard technique, allowing surgeons to improve efficiency by reducing potentially time-consuming interoperative steps in a streamlined surgical process.
Key market players are focusing on strategies such as partnership and collaboration to increase production and innovation in companion animal vaccines, which is providing opportunities for new vaccine launches, which is expected to drive demand during the forecast period. For instance, in March 2023, the Biotechnology and Biological Sciences Research Council (BBSRC), a part of UK Research and Innovation (UKRI), and the Bill & Melinda Gates Foundation, a U.S.-based private foundation, announced the creation of a new center at The Pirbright Institute to accelerate the development of animal vaccines to combat emerging and urgent infectious diseases.
There are several advantages to having a companion animal. Pet popularity of stroking cats and dogs is expanding globally, however, pets can occasionally carry hazardous germs that can be transmitted to people and cause sickness. These are known as zoonotic illnesses. As per data for zoonotic diseases in Eastern Europe published in July 2020 by the World Health Organization (WHO), it is estimated that globally, about one billion cases of illness and millions of deaths occur every year from zoonoses. Over 30 new human pathogens have been detected in the last three decades, 75% of which have originated in animals.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 4.09 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.0% | 2033 Value Projection: | USD 6.56 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Elanco Animal Health Incorporated, Boehringer Ingelheim GmbH, Ceva Ltd., Merck & Co. Inc., Virbac, Zoetis Inc., HIPRA, Biogénesis Bago, Ourofino Animal Health, Croda International Plc, KM Biologics, KM Biologics, Sinovac, Kyoto Biken Laboratories, Inc., HESTER BIOSCIENCES LIMITED, Calier, Bioveta, a.s. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Among product types, the Attenuated Live Vaccine segment is dominant during the forecast period in the North America region due to the key players focusing on organic strategies such as the launch of new products, which is expected to drive segment growth over the forecast period. For instance, on June 12, 2023, MSD Animal Health announced approval of INNOVAX-ILT-IBD from the U.S. Food and Drug Administration and the European Commission for the Innovax vaccine, a dual-construct HVT vaccine that provides long-term protection against infectious laryngotracheitis. Such predicted approvals and launches are fueling the global companion animal vaccines market in North America.
*Definition: Companion animal vaccinations are those administered to pets (such as dogs and cats) to stimulate their immune responses. Companion animal vaccines, in other words, are used to improve the functioning of the immune system in pets and other animals.
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients