The global Contract Research Organization (CRO) services market size is projected to grow from USD 82.86 Bn in 2026 to USD 137.52 Bn by 2033, registering a compound annual growth rate (CAGR) of 7.5% during the forecast period (2026–2033). This growth is primarily driven by increasing pharmaceutical and biotechnology R&D outsourcing, rising complexity and volume of clinical trials, growing demand for cost-efficient drug development processes, and expanding adoption of advanced clinical trial methodologies including decentralized and data-driven research models.
The Clinical Research Services segment is expected to hold the highest market share among all segments of the Contract Research Organization (CRO) services market by 2026, accounting for around 55% of the overall market share. This is attributed to the growing complexities, sizes, and geographical spread of clinical trials ranging from phases I to IV, and outsourcing activities.
In September 2025, IQVIA launched its Clinical Trial Financial Suite (CTFS), an AI-powered platform designed to streamline budgeting, forecasting, and payment workflows across global clinical trials.
To learn more about this report, Request Free Sample
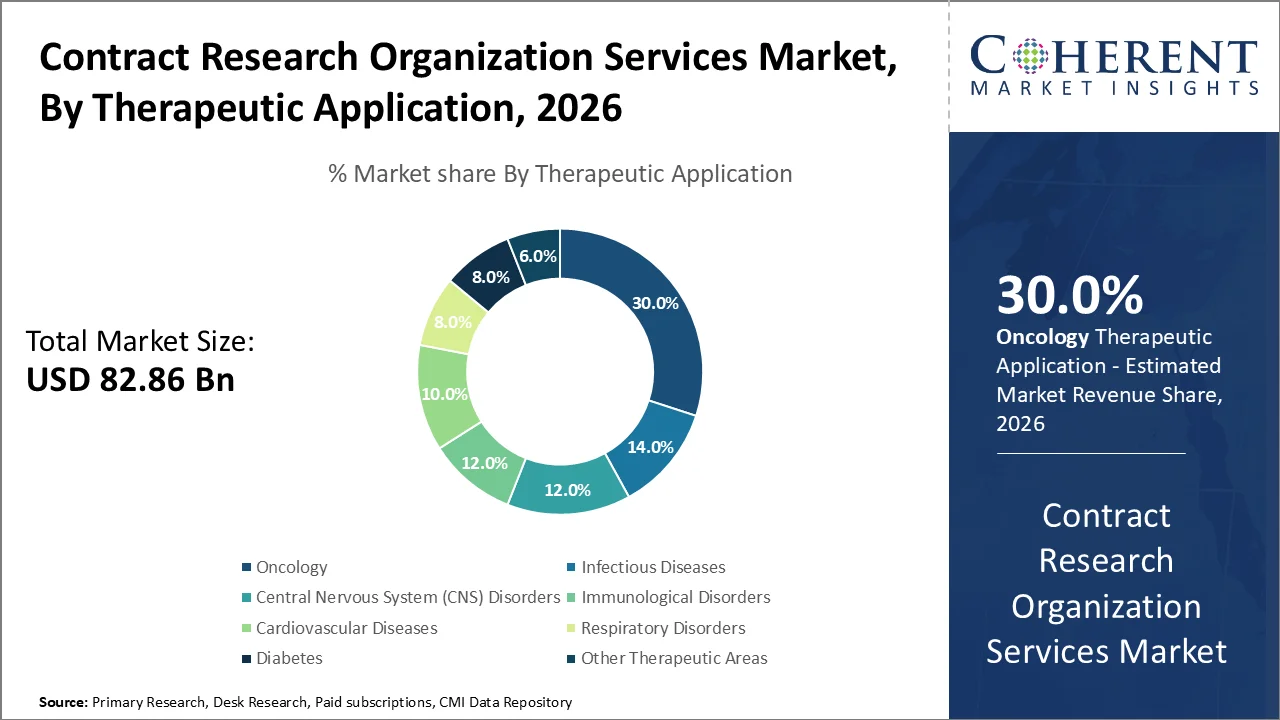
The oncology segment is expected to remain the leading area within the CRO services market in 2026, accounting for around 30% of the share. This is largely driven by the rising global burden of cancer, along with a strong pipeline of oncology therapies, including immunotherapies, targeted treatments, and personalized medicine approaches. About 1 in 5 people globally will develop cancer during their lifetime, while mortality risk is estimated at 1 in 9 for men and 1 in 12 for women. By 2050, the global cancer burden is projected to increase significantly, with new cases reaching around 33 million annually and deaths rising to approximately 18.2 million per year.
In addition, oncology consistently represents a significant portion of clinical trials worldwide. According to information provided by ClinicalTrials.gov, cancer research is one of the most conducted clinical trials. Therefore, drug manufacturing firms have been directing huge R&D capital towards oncology, hence maintaining high demand for outsourced research assistance in oncology.
In August 2025, IQVIA highlighted continued expansion of its oncology-focused clinical research capabilities, emphasizing advanced trial design, real-world evidence integration, and AI-enabled oncology development platforms to support faster cancer drug approvals and precision medicine programs across global markets.
Pharmaceutical and Biopharmaceuticals companies segment hold the largest market share in terms of CRO services in the year 2026, accounting for about 72% of overall market demand owing to their increased tendency to outsource clinical research and other R&D-related activities in order to lower development costs.
Moreover, major pharmaceutical companies have started to outsource much of the clinical trial work, and it is projected that 70% or more of clinical trial tasks are outsourced via CRO collaboration. This phenomenon can be attributed to the increasing intricacy of biologic drugs, gene therapy drugs, and personal medicine development processes.
Pharmaceutical and biotech companies are increasingly outsourcing to CROs clinical research functions due to the high cost of discovering drugs and the need to improve efficiency in bringing them to the market. Increasing complexity of clinical trials, especially in oncology and orphan diseases, further boosts the demand for CROs.
Additionally, a report by Tufts Center for the Study of Drug Development (CSDD) highlights that outsourcing to CROs has significantly increased, with over 70% of clinical trial activities in large pharma companies now being outsourced to external partners.
The increase in the cases of chronic diseases like cancer has led to the rise in the number of contracts between CROs and clients. The field of oncology continues to be a leader compared to others as a result of immunotherapy, targeted therapy, and personalized medicine.
In addition, according to the database of ClinicalTrials.gov, over 470,000 clinical trials have been registered worldwide, with oncology being one of the most numerous groups of trials.
|
Current Event |
Description and its Impact |
|
2026 U.S. FDA Regulatory Modernization and Accelerated Clinical Trial Approvals |
|
|
Expansion of Decentralized and Hybrid Clinical Trials (DCTs) |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
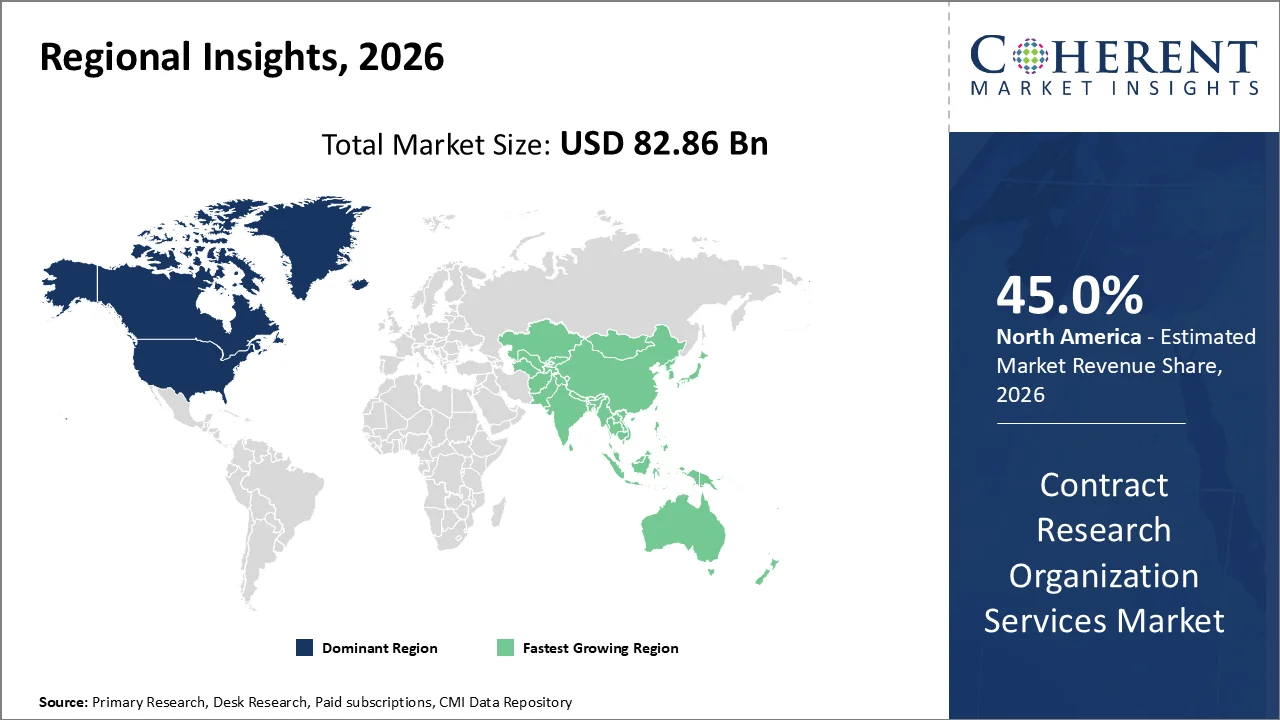
The North American CRO services market is forecast to lead the global CRO services market in 2026, occupying 45% of the market, owing to the existence of prominent pharmaceutical and biotech companies, higher number of clinical trials, and superior regulatory environment. The region enjoys an established research, development environment and outsourcing practices among drug developers.
The United States leads clinical research activity globally, hosting a significant proportion of registered clinical trials. According to ClinicalTrials.gov, the U.S. accounts for over 30% of all active clinical studies worldwide, reinforcing strong CRO demand.
The Asia-Pacific region is expected to be the fastest-growing CRO services market over the forecast period of 2026-2033, exhibiting a CAGR of around 10%. This impressive growth rate is attributed to the outsourcing of clinical trials by pharmaceutical and biotech organizations across the globe, lower costs compared to western nations, enhanced healthcare infrastructure, and access to large naïve patient pools that ensure rapid patient enrollment and effective trial management.
According to IQVIA, the Asia-Pacific region is increasingly preferred for global clinical trials due to lower operational costs, faster recruitment timelines, and strong scalability, making it a strategic destination for CRO expansion.
In March 2025, ICON plc, announced expansion of its Asia-Pacific clinical research operations, strengthening its presence across India, China, and Southeast Asia to support increasing demand for outsourced clinical trials.
The United States CRO services market has witnessed a notable growth owing to the pharmaceutical R&D environment, high number of clinical trials, and outsourcing of drug development processes. The US has been home to a considerable percentage of clinical research worldwide, with more than 30% of total registered clinical trials being carried out in the US (ClinicalTrials.gov). This further confirms that the US has been the biggest CRO market across the globe. Increasing R&D costs, estimated at around USD 2.6 billion on average for each drug development process (Tufts CSDD), have compelled pharma and biotech firms to outsource their clinical operations to CROs.
In July 2025, Labcorp (U.S.-based global CRO and diagnostics company) announced strong second-quarter 2025 results and highlighted continued expansion of its clinical research and oncology testing services in the United States.
The Japan CRO services industry will see constant growth during the forecast period owing to its health care sector, regulatory environment, and fast-growing older population. Japan’s population is known as one of the oldest populations worldwide, with nearly 30% of the population aged above 65 years. This demographic trend has led to increased clinical research in fields such as cancer, neurological disorders, and chronic diseases.
According to IQVIA, Japan continues to play a critical role in Asia-Pacific clinical research due to its high-quality standards, advanced healthcare system, and strong integration with global pharmaceutical development networks.
Some of the major key players in Contract Research Organization Services are IQVIA, LabCorp, PAREXEL, ICON plc, PPD, PRA Health Sciences, Syneos Health, Charles River, Wuxi PharmaTech, Medpace Holdings, SGS, Envigo, and MPI Research.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 82.86 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 7.5% | 2033 Value Projection: | USD 137.52 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
IQVIA, LabCorp, PAREXEL, ICON plc, PPD, PRA Health Sciences, Syneos Health, Charles River, Wuxi PharmaTech, Medpace Holdings, SGS, Envigo, and MPI Research. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients