The Global Smoking Cessation and Nicotine De-addiction Products Market is estimated to be valued at USD 22.81 Bn in 2026 and is expected to reach USD 70.52 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 17.5% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
Increasing health concerns related to smoking, rising awareness about available smoking cessation products, and initiatives by public and private organizations to curb tobacco consumption are major factors fueling demand.
The market is witnessing positive trends with more people looking to quit smoking and reduce tobacco consumption. Growth in digital platforms promoting anti-smoking campaigns and availability of various nicotine replacement therapies and e-cigarettes are giving consumers multiple options to choose from for their nicotine de-addiction needs. This is expected to drive further uptake of smoking cessation products during the forecast period.
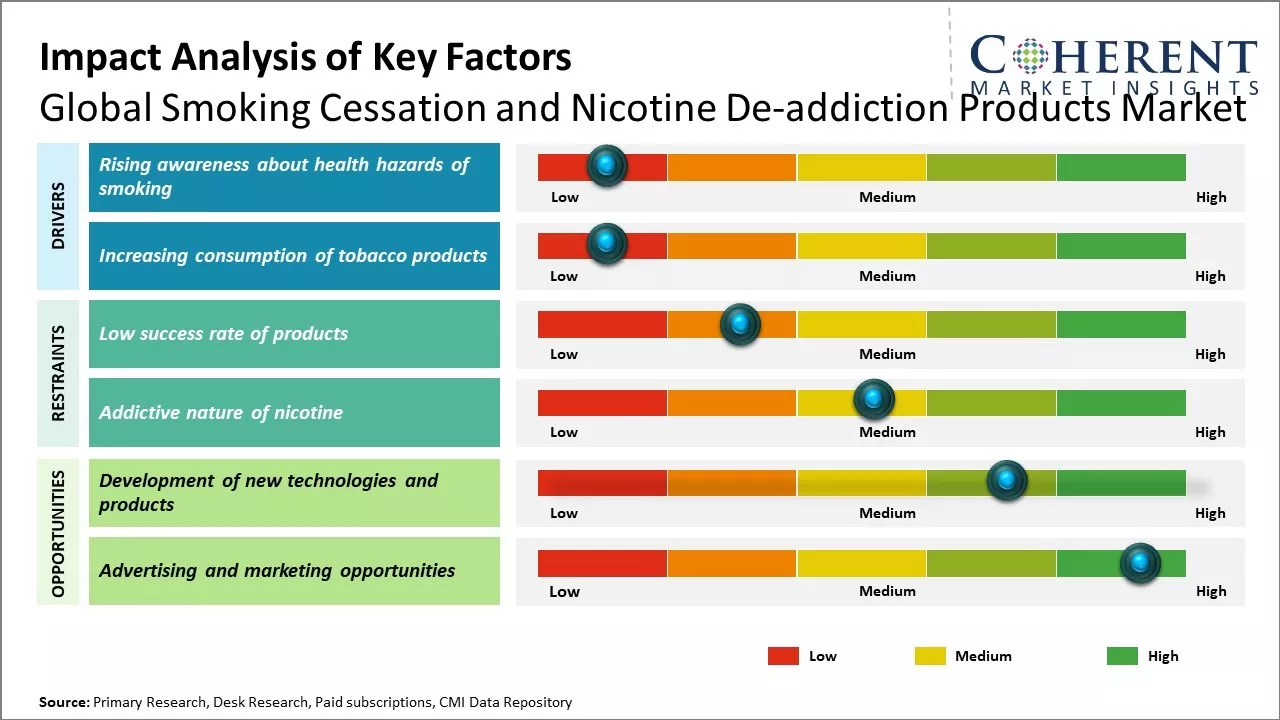
Rising awareness about health hazards of smoking
With extensive research highlighting the ill-effects of smoking on human health, awareness about smoking-related diseases has increased significantly among the global population in recent years. Various healthcare organizations and anti-tobacco advocacy groups have launched education initiatives through multiple channels to educate people, especially the youth, about direct correlation between smoking and life-threatening conditions like lung cancer, respiratory diseases, cardiovascular problems and more. Governments of many countries have made it mandatory to print pictorial warnings on tobacco product packages informing consumers about health risks. Mass media campaigns regularly caution people against taking up smoking through impactful messages. Studies have found that such awareness campaigns have convinced many current smokers to try quitting and also discouraged potential new users from picking up the habit. As people increasingly recognize smoking as a serious risk factor for diseases rather than just a casual habit, the motivation to seek cessation products and therapies is seen to rise proportionately across global markets. For instance, in 2021, the Global Adult Tobacco Survey (GATS) in India indicated a notable increase in awareness about the harmful effects of tobacco between 2009-2010 (GATS I) and 2015-2016 (GATS II). Key findings show that awareness levels varied significantly across demographics such as age, gender, education, wealth, urban-rural divide, and regional disparities. Overall, awareness among smokers, smokeless tobacco users, and non-users increased, with significant improvements observed in gender, age groups, residential areas, and geographical regions. However, comprehensive awareness remains suboptimal, suggesting the need for targeted health campaigns focusing on regional disparities, adopting a gender-neutral approach, and targeting younger populations.
Get actionable strategies to beat competition: Request Free Sample
Increasing consumption of tobacco products
Rising tobacco consumption worldwide has become a major public health issue. According to the WHO, over 1.3 billion people use tobacco globally with over 8 million deaths attributed to direct tobacco use annually. The health consequences of tobacco use such as cancers, cardiovascular, and respiratory diseases have seen steady rise. This is prompting many smokers to think about quitting smoking. At the same time, governments and health agencies across nations are promoting effective smoking cessation programs to help current smokers quit tobacco. All of these factors are directly fueling the growth of the global smoking cessation and nicotine de-addiction products industry. As per recent data published by CDC, nearly 70% of current U.S. adult smokers said they want to quit completely in 2020, up from 50-60% over the last decade. This shows that more smokers are willing to adopt de-addiction methods like nicotine replacement therapy and e-cigarettes. Major players in the industry are tapping into this opportunity by continuously expanding their product portfolios to suit diverse needs of smokers.
Key Takeaways from Analyst:
The global smoking cessation and nicotine de-addiction products market is expected to witness steady growth over the forecast period driven by rising awareness about health hazards of smoking. Growing initiatives by various governmental and non-governmental organizations to educate people are generating more demand. North America currently dominates the market owing to stringent regulations and bans on smoking in public places. Europe is another major market benefiting from rising quit rates. However, Asia Pacific is likely to emerge as the fastest growing market over the coming years supported by large population base and growing health consciousness.
On the other hand, the availability of inexpensive illicit alternatives and social acceptance of smoking in certain cultures remain key challenges. Changing consumer preferences from nicotine replacement therapies to e-cigarettes could also impact demand patterns in the near future. Nonetheless, the launch of novel products catering to new consumer segments and convenience of e-cigarettes are opening new opportunities. Additionally, as health expenditures rise with aging populations, medical professionals and insurers are emphasizing on preventive treatment options such as smoking cessation products. Advancements in nicotine delivery mechanisms ensuring high customer satisfaction without completely eliminating the smoking experience could further boost the market prospects. In overall terms, the outlook for the global market appears positive.
Market Challenges: Low success rate of products
The low success rate of smoking cessation products is a major challenge restraining the growth of the global smoking cessation and nicotine de-addiction market. Currently, most smoking cessation products and therapies have very limited efficacy, with success rates of only 20-30% on average. This means the majority of people are unable to quit smoking even after trying approved therapies and products. The low success rates undermine users' confidence in these solutions and discourage many smokers from even attempting to quit. As a result, smokers remain hesitant to invest time and money into products and programs that have a high likelihood of failing. This significantly reduces the overall demand for smoking cessation offerings. Additionally, the recurring nature of relapses also diminishes repurchase rates for replacement therapies and nicotine gums/patches, limiting revenue opportunities for market players. An added concern is that the few products showing some efficacy, such as varenicline (Chantix) and combination nicotine replacement therapies, involve high costs that many smokers are reluctant or unable to afford. This cost hurdle further exacerbates accessibility and compliance issues. For example, according to 2020 data from the CDC, almost 70% of U.S. adult smokers tried to quit in the past year, but only 55% of those tried to use counseling and/or medication due to concerns over costs. The lack of affordable and effective smoking cessation options continues to enable the prevalence of smoking worldwide.
Market Opportunities: Development of new technologies and products
The development of new technologies and products present a great opportunity for innovation in the global smoking cessation and nicotine de-addiction market. As smokers increasingly seek less harmful alternatives to traditional cigarettes, technological advancements are enabling novel solutions aimed at reducing tobacco-related harm. For instance, heated tobacco products such as IQOS devices that heat but do not burn tobacco, allowing for inhalation of nicotine vapors without smoke. While still containing nicotine, early research suggests these heated tobacco products expose users to significantly lower levels of harmful chemicals compared to cigarette smoke. Such reduced risk products, if backed by robust scientific evidence, have the potential to meaningfully impact public health by providing smokers an alternative way to satisfy their nicotine craving with less risk. Similarly, the e-cigarette category continues to rapidly evolve. New generations of e-cigarettes are improving nicotine delivery capabilities to better mimic the feel of smoking, which helps increase their effectiveness as smoking cessation aids. Product developments like pod-based e-cigarettes with pre-filled nicotine cartridges have made these devices even more convenient and easier for smokers to use. This increased user-friendliness expands the addressable market size. The latest e-liquids are also being formulated with novel nicotine salts to provide a smoother vaping experience. More effective nicotine delivery from such innovations could increase the success rate of quit attempts.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
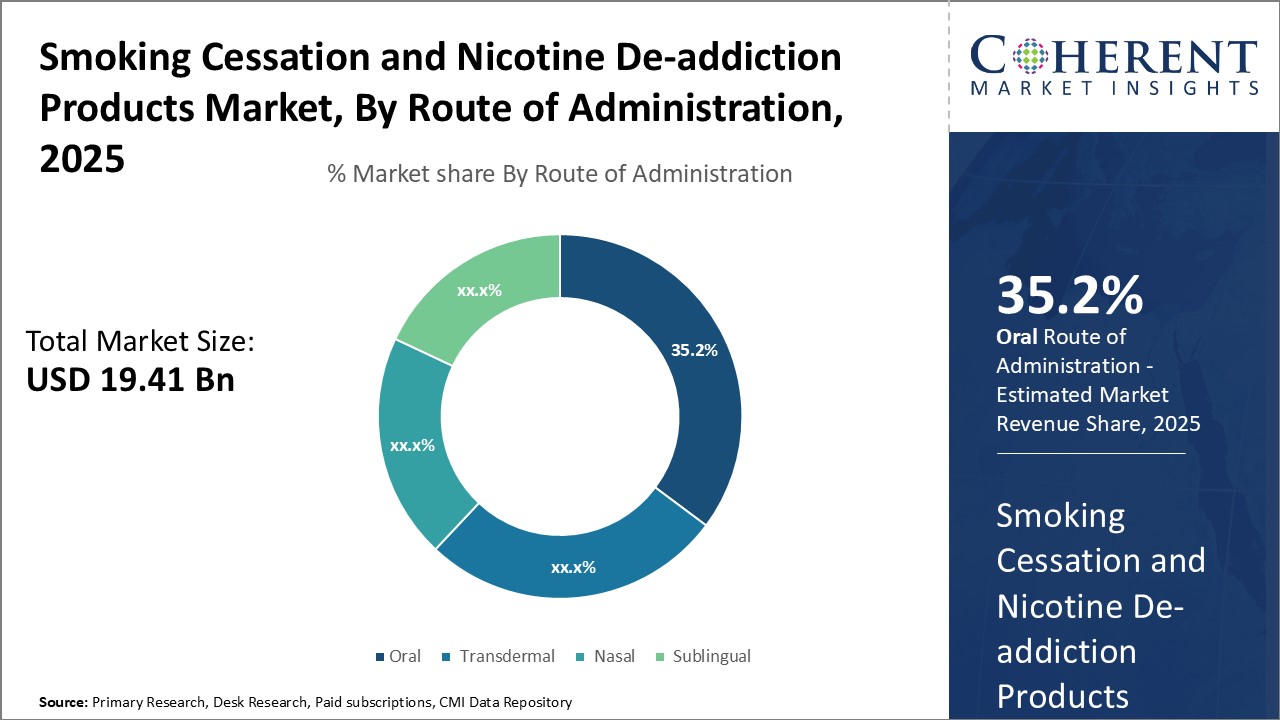
By Route of Administration - Convenience Drives Oral Segment Growth
In terms of Route of Administration, the Oral segment is estimated to hold the highest share of 36.2% in 2026 owing to its convenience of administration. Oral products such as nicotine gums, lozenges, pills and mouth sprays provide an easy option to consume nicotine replacement therapy anytime and anywhere without any complex administration process. Users can opt for oral products discreetly even at workplaces or in public without feeling embarrassed. This self-administrable feature makes oral products highly portable and suitable for around-the-clock usage from morning to evening as per the craving levels. Moreover, oral products do not require prescription and are widely available over-the-counter at affordable price points, further augmenting their adoption. Strong distribution network and loyal customer base have enabled oral products to gain mass acceptance among people trying to quit smoking as well those willing to reduce nicotine consumption gradually.
To learn more about this report, Request Free Sample
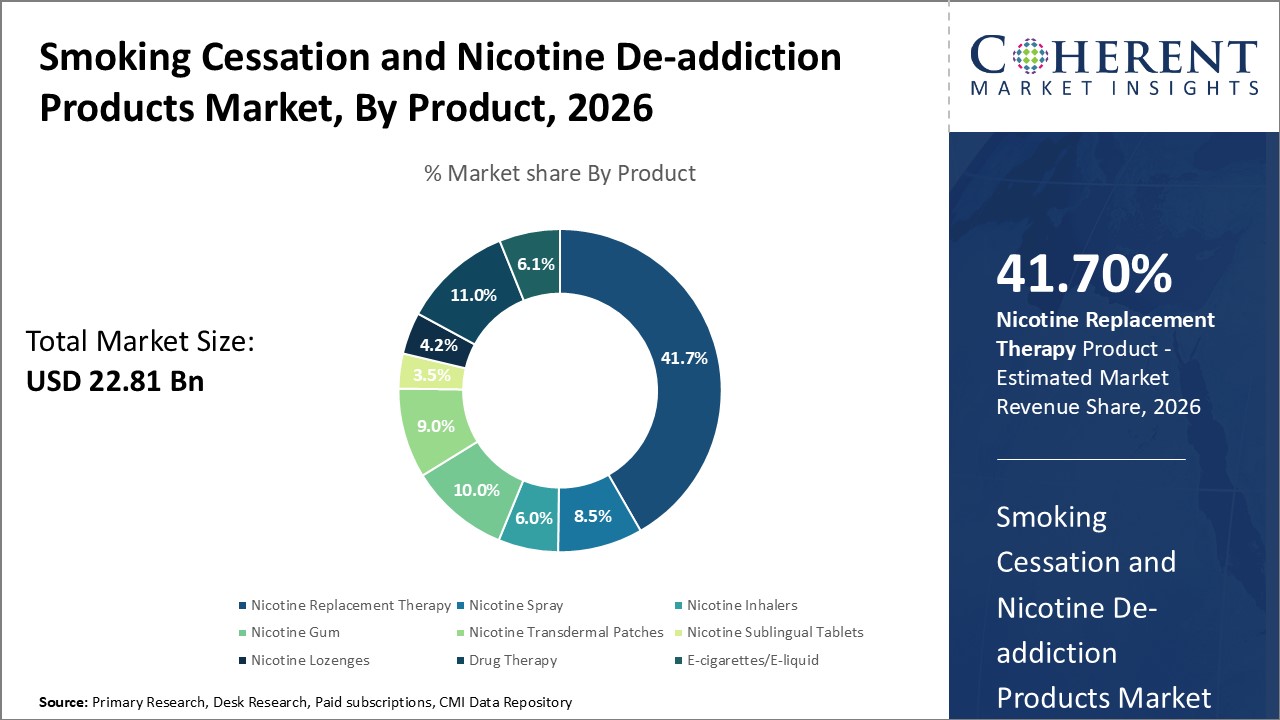
By Product - Nicotine Replacement Therapy Dominates Due to Well-established Standards
In terms of Product, the Nicotine Replacement Therapy segment is estimated to hold the highest share of 41.70% in 2026 owing to its well-established industry standards and safety protocols. Nicotine replacement products such as gums, patches and lozenges provide controlled and measured dose of nicotine to curb withdrawal symptoms without intake of toxic combusted smoke. Regulatory bodies worldwide have approved various NRT formats after extensive research and trials establishing their safer nature compared to smoking. Manufacturers adhere to high quality and packaging standards to maintain consistency of nicotine dosage across batches. Nicotine replacement also allows titration of dosage as per requirements, facilitating gradual reduction of nicotine dependence. The longstanding usage and recognition as first line of treatment have made NRT highly preferred among healthcare professionals and patients seeking clinically proven smoking cessation aids.
By Distribution Channel- Hospital Accessibility Bolsters Hospital Pharmacy Segment
In terms of Distribution Channel, the Hospital Pharmacy segment is estimated to hold the highest share of 41.62% in 2026 due to enhanced accessibility for patients. Hospital pharmacies are predominantly present within the premises of healthcare centers, allowing patients to directly procure smoking cessation products during doctor consultations or post-admission care without any additional travel. This eliminates logistical hurdles for individuals undergoing treatment or recovering from smoking-related ailments. Moreover, hospital pharmacies stock a validated range of products approved by attached medical facilities. Users can be confident about the authenticity and quality of products available within trusted healthcare settings. Pharmacists at hospitals also play an educative role, guiding patients on appropriate product selection and correct usage. The healthcare setups also facilitate monitoring of treatment progress through periodic follow-ups by doctors, improving treatment effectiveness through hospital pharmacy channel.
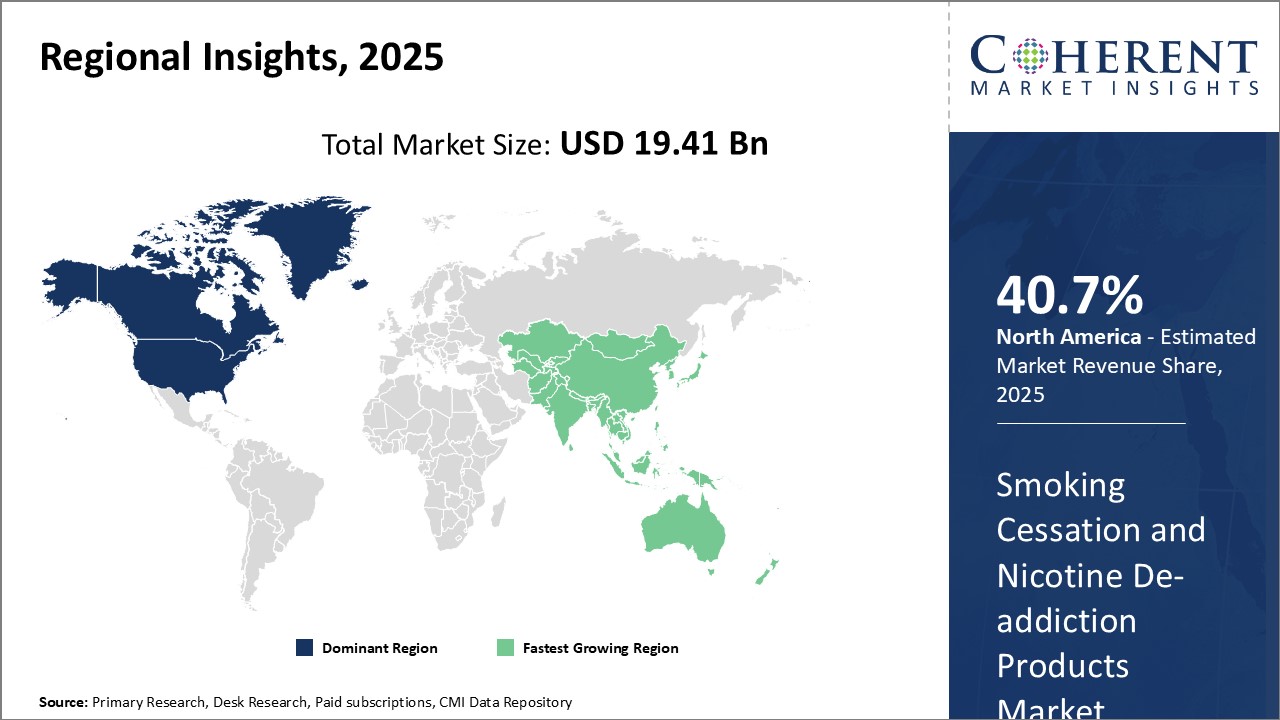
Need a Different Region or Segment? Request Free Sample
North America has established itself as the dominant region in the global smoking cessation and nicotine de-addiction products market with an estimated market share of 40.2 % in 2026. The U.S. accounts for the lion's share of the North American market. This is driven by growing health consciousness among consumers and strong regulatory guidelines around tobacco control. Government initiatives such as imposing high taxes on cigarettes along with effective anti-tobacco campaigns have raised awareness about the dangers of smoking. Some states have even banned indoor smoking in public places which has encouraged many smokers to quit.
The Asia Pacific region is positioned as the fastest growing market for smoking cessation and nicotine de-addiction products globally. China accounts for the lion’s share of the overall Asia Pacific market due to its large population of smokers. In recent years, the country has implemented new regulations on tobacco control and public awareness drives to promote a healthy lifestyle. This is encouraging Chinese smokers to opt for alternative nicotine delivery products. Furthermore, India and other Southeast Asian nations are also contributing significantly to the regional market growth.
Smoking Cessation and Nicotine De-addiction Products Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 22.81 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 17.5% | 2033 Value Projection: | USD 70.52 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
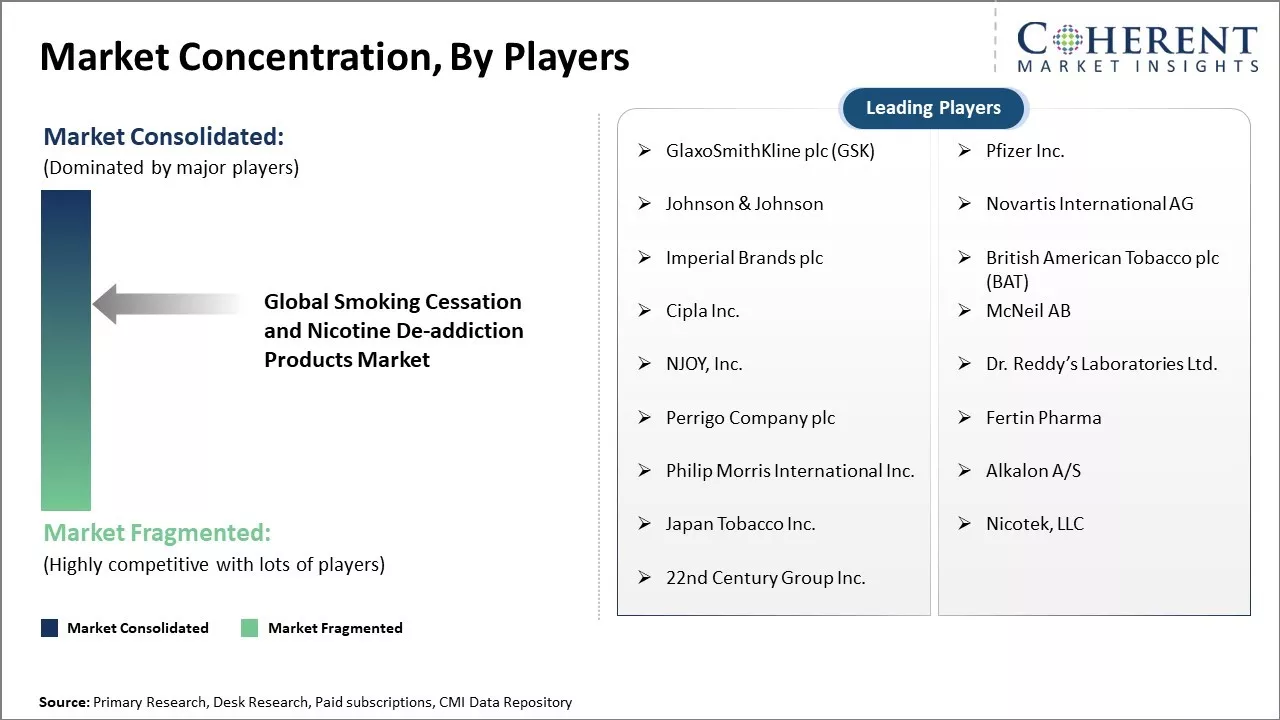
| Companies covered: |
GlaxoSmithKline plc (GSK), Pfizer Inc., Johnson & Johnson, Novartis International AG, Imperial Brands plc, British American Tobacco plc (BAT), Cipla Inc., McNeil AB, NJOY, Inc., Dr. Reddy’s Laboratories Ltd., Perrigo Company plc, Fertin Pharma, Philip Morris International Inc., Alkalon A/S, Japan Tobacco Inc., Nicotek, LLC, 22nd Century Group Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: The global smoking cessation and nicotine de-addiction products market consists of a wide range of products that help people quit smoking and relieve withdrawal symptoms caused by nicotine addiction. This includes nicotine replacement therapy products like nicotine gum, patches, lozenges, and inhalers. It also includes prescription smoking cessation aids like Bupropion and Varenicline, as well as apps and websites offering digital therapies for smoking cessation. The market also comprises non-nicotine medications and therapies to help treat nicotine dependency worldwide.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients