Critical Limb Ischemia Treatment Market Size and Trends
The global critical limb ischemia treatment market is estimated to be valued at USD 6.05 Bn in 2026 and is expected to reach USD 11.06 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 9.0% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
The critical limb ischemia treatment market is expected to witness prominent growth over the forecast period. The rising geriatric population worldwide and the growing prevalence of peripheral artery disease are major factors contributing to the increased demand for critical limb ischemia treatment. Furthermore, the increasing adoption of minimally invasive surgeries and the growing number of regulatory approvals for advanced treatment options are expected to provide new opportunities for players in the market. However, the high cost of treatment and lack of reimbursement policies in developing nations may hinder the market growth.
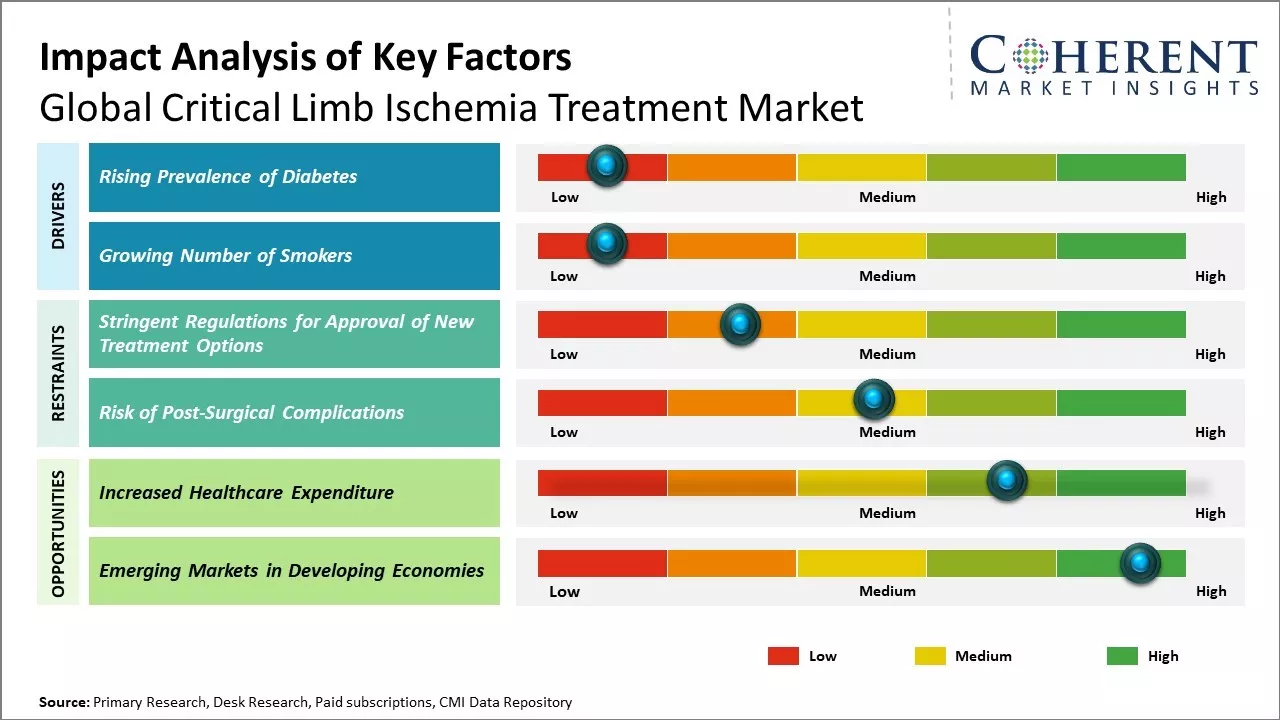
Rising Prevalence of Diabetes
The global prevalence of diabetes has been steadily increasing over the past few decades. According to estimates, over 460 million adults were living with diabetes in 2019, compared to around 108 million in 1980. Type 2 diabetes in particular has risen dramatically worldwide due to increasing levels of obesity and sedentary lifestyles. Diabetes greatly elevates the risk of developing peripheral artery disease and critical limb ischemia, as high blood sugar levels can damage and narrow the arteries supplying blood to the legs and feet over time. It is projected that by 2030, diabetes will affect over 700 million people globally if preventive measures are not taken. As more people are afflicted by this chronic condition, cases of CLI Critical Limb Ischemia are also expected to grow substantially. Early treatment of diabetes and maintaining optimal blood sugar control can help reduce complications, but many patients may still develop PAD and require CLI Critical Limb Ischemia treatment. The growing diabetes epidemic worldwide will therefore act as a key driver for the critical limb ischemia market in the coming years. The increasing prevalence of diabetes poses a heightened risk for patients developing chronic limb-threatening ischemia (CLTI) due to peripheral arterial disease. For instance, with diabetes rates projected to rise, as indicated by the International Diabetes Federation's 2022 statistics, from 32 million in the U.S. to 36 million by 2050, there is a growing concern over the potential damage caused by high blood sugar levels to nerves regulating heart and blood vessel functions. This scenario is anticipated to drive the demand for more effective treatments, thereby stimulating the market growth in arterial disease management.
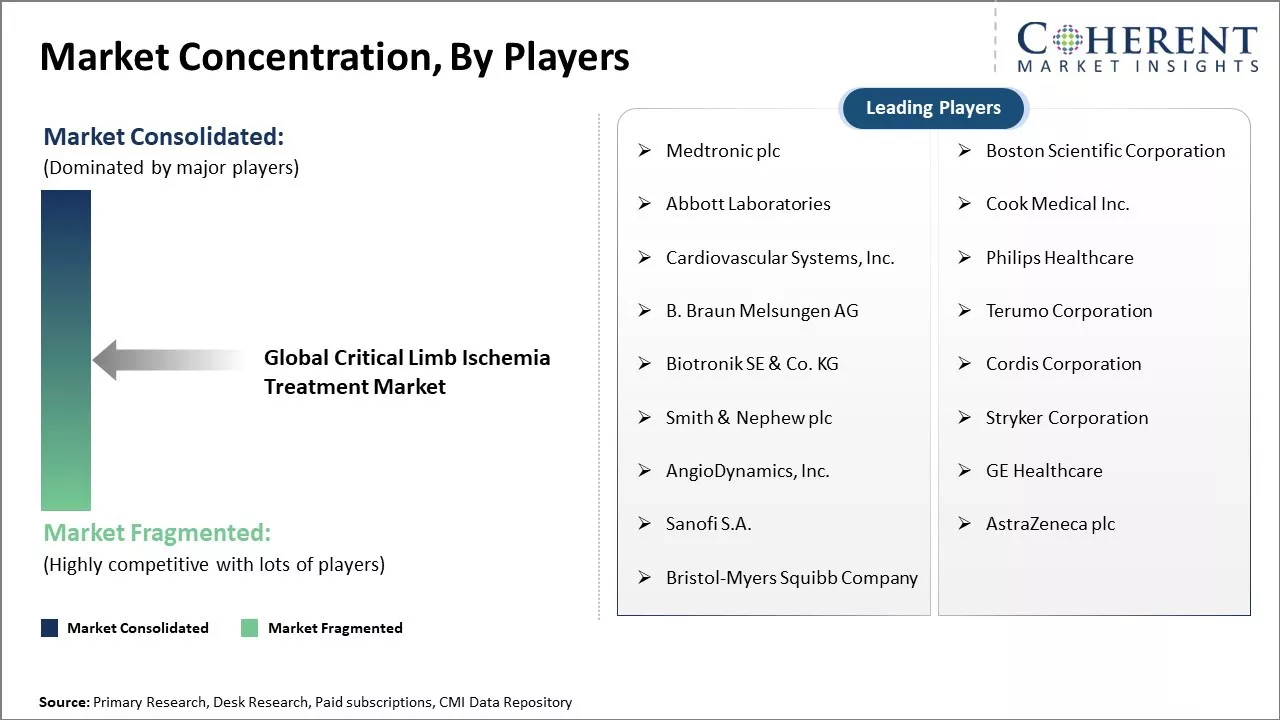
Market Concentration and Competitive Landscape
Get actionable strategies to beat competition: Request Free Sample
Growing Number of Smokers
The growing number of smokers around the world is having a significant impact on the critical limb ischemia treatment market. As smoking is one of the major risk factors that can lead to peripheral artery disease (PAD) and critical limb ischemia, the rising prevalence of smoking will fuel the need for effective treatment options for this condition. PAD occurs when arteries in the legs or arms become narrowed due to buildup of fat, cholesterol, calcium or other substances over many years of smoking. Smoking constricts the blood vessels and reduces blood flow to arms and legs. It damages the inner lining of arteries and promotes formation of blood clots. All of these smoking-related effects increase the chances of developing critical limb ischemia, which is the most severe form of PAD that occurs when limbs do not receive adequate blood flow. According to recent data from the World Health Organization, In July 2023 approx one in five adults smoke tobacco on a daily basis globally. While smoking rates are declining in high-income countries, they are still elevated and rising in many low and middle-income countries. Every year over 8 million people die due to tobacco use. If current trends continue, tobacco is expected to kill more than 1 billion people globally in the 21st century. The growth in the overall number of smokers indicates that more people will develop critical limb ischemia due to PAD in the coming years due to prolonged smoking.
Key Takeaways from Analyst:
The global critical limb ischemia treatment market is expected to experience steady growth over the forecast period driven by the rising prevalence of peripheral artery disease and diabetes globally. The growing geriatric population who are more prone to CLI will also fuel market expansion. Advancements in revascularization treatments including drug-coated balloons and stents are providing improved treatment outcomes for patients boosting adoption.
However, high treatment costs associated with CLI therapies continue to restrain the market growth to a certain extent. Lack of reimbursement coverage in developing nations poses significant barrier. Moreover, alternative treatment options like amputation also take away share from revascularization procedures.
The North America region currently dominates the Critical Limb Ischemia treatment market owing to the presence of major players and availability of advanced treatment options. Asia Pacific is likely to witness fastest gains supported by improving access to healthcare facilities and increasing expenditure in major countries. Growing medical tourism industry in India and China will benefit market prospects in the Asia Pacific region.
Development of innovative drug-eluting balloons and stents with higher effectiveness and lower restenosis rates provides lucrative opportunities for key players. Investments into stem cell therapy and gene therapy research could yield breakthrough results over long-term.
Market Challenges: Stringent Regulations for Approval of New Treatment Options
Stringent regulations for the approval of new treatment options is one of the major factors restraining the growth of global critical limb ischemia treatment market. The approval process for any new drug or treatment in this market is extremely lengthy and complex. Clinical trials and vast amount of clinical data is required to prove the safety and efficacy of new treatment modalities before they can be cleared for market use. This prolonged approval timeline poses significant challenges for pharmaceutical and medical device companies. For example, according to the World Health Organization, the average time required for approval of a new drug in the U.S. is over 7 years. This includes several phases of clinical trials involving thousands of subjects and gathering vast amount of data on clinical endpoints such as reduction in amputation rates, improvement in pain levels and overall quality of life. Any minor issues or deficiencies found during regulator reviews can significantly delay the approval process. This lengthy and expensive drug development process acts as a deterrent for many small to mid-sized companies to invest in development of new critical limb ischemia treatments.
Market Opportunities: Increased Healthcare Expenditure
Increased healthcare expenditure on critical limb ischemia treatment presents a great opportunity for the Global Critical Limb Ischemia Treatment Market. Critical limb ischemia is a severe blockage of arteries that reduces blood flow to the limbs. It can lead to pain and serious complications including sores, infections and amputation if left untreated. As per the World Health Organization, the global prevalence of peripheral artery disease or PAD, of which critical limb ischemia is a severe form, is around 200 million cases. It is estimated that 10–20% of PAD patients will progress to critical limb ischemia if left untreated. With rapid aging of the population globally and rising prevalence of diseases like diabetes that increase the risk of critical limb ischemia, there is expected to be a significant increase in the number of critical limb ischemia cases in the coming years. At the same time, healthcare expenditure is also projected to increase substantially across many countries. For example, healthcare spending as percentage of GDP in the U.S. is projected to increase from 17.7% in 2018 to 19.4% by 2023 according to data from The Centers for Medicare and Medicaid Services. Similarly, healthcare spending as percentage of GDP in China, Germany and India is also projected to increase in the coming years.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
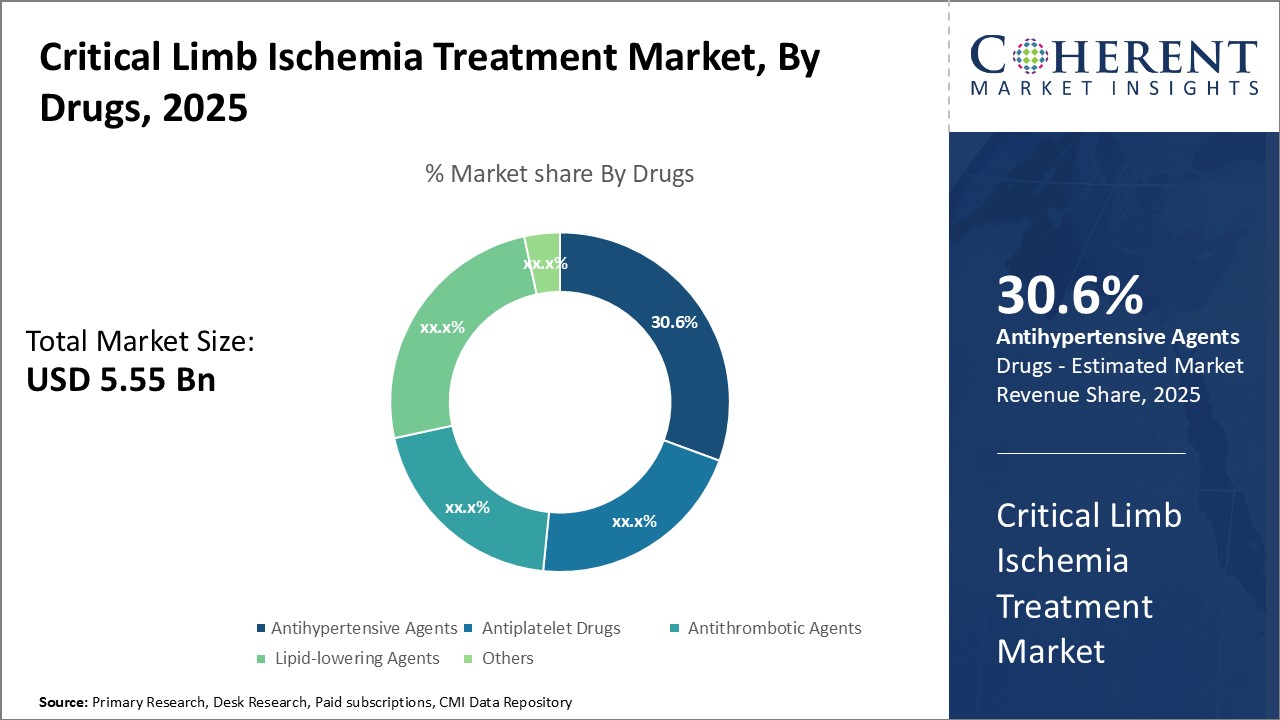
By Drugs - Drug Efficacy and Safety Drives the Antihypertensive Agents Segment
In terms of drugs, antihypertensive agents is expected to contribute the highest share of 31.6% in 2026 owing to their strong efficacy and safety profile. Antihypertensive drugs help lower blood pressure and reduce strain on heart and blood vessels. Lowering blood pressure also helps improve blood flow to critical limbs affected by ischemia. Among antihypertensive classes, Angiotensin Converting Enzyme (ACE) inhibitors are widely used for their vasodilating properties. ACE inhibitors work by inhibiting the conversion of angiotensin I to angiotensin II, thereby relaxing blood vessels and improving blood flow. Calcium channel blockers are another popular class that dilate blood vessels by blocking calcium ions. They help treat patients with symptoms of intermittent claudication associated with critical limb ischemia. The proven efficacy and safety record of antihypertensive drugs in management of cardiovascular conditions drives their higher adoption rate compared to other drug classes.
To learn more about this report, Request Free Sample
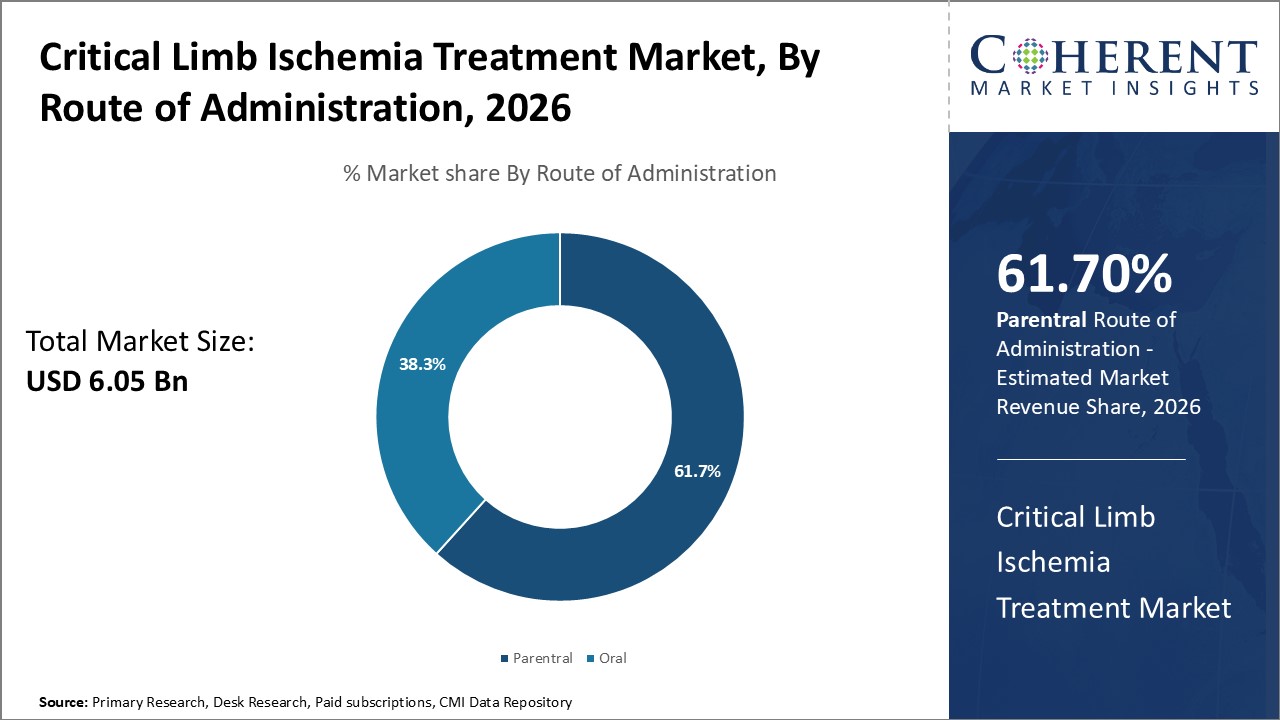
By Route of Administration- Route of Administration Influences the Parenteral Segment
In terms of route of administration, parenteral is expected to contribute the highest share of the market, accounting for 61.7% in 2026 owing to direct absorption into bloodstream. Critical limb ischemia is a severe condition requiring prompt treatment to restore blood flow. Oral administration might not provide adequate or rapid relief from symptoms. Parenteral administration through injections allows drugs to directly enter systemic circulation without needing to pass through gastrointestinal tract and liver metabolism. This ensures consistent and predictable drug levels within the therapeutic window. It also leads to the faster onset of action. For patients with nutritional deficiencies or inability to swallow, parenteral route provides a more feasible option for drug delivery. These advantages make parenteral the preferred mode of administration for managing critical limb ischemia.
By End User- Hospitals Lead Due to Diagnostic Facilities
In terms of end user, hospitals is expected to contribute the highest share of the market, accounting for 41.7% in 2026 owing to the availability of advanced diagnostic and treatment facilities. Critical limb ischemia diagnosis requires specialized tests and imaging-like ankle brachial pressure index, duplex ultrasound, angiogram, etc. to accurately assess the blood flow status. Hospitals are well equipped with state-of-the-art diagnostic laboratories integrated with expert medical personnel to conduct these tests. They also have vascular surgeons and interventional radiologists who can efficiently treat the underlying conditions through endovascular or surgical procedures like angioplasty, stenting or bypass grafts. Post procedure care in hospital setup further improves clinical outcomes. Access to comprehensive care drives patients particularly to large hospitals over other end users for critical limb ischemia management.
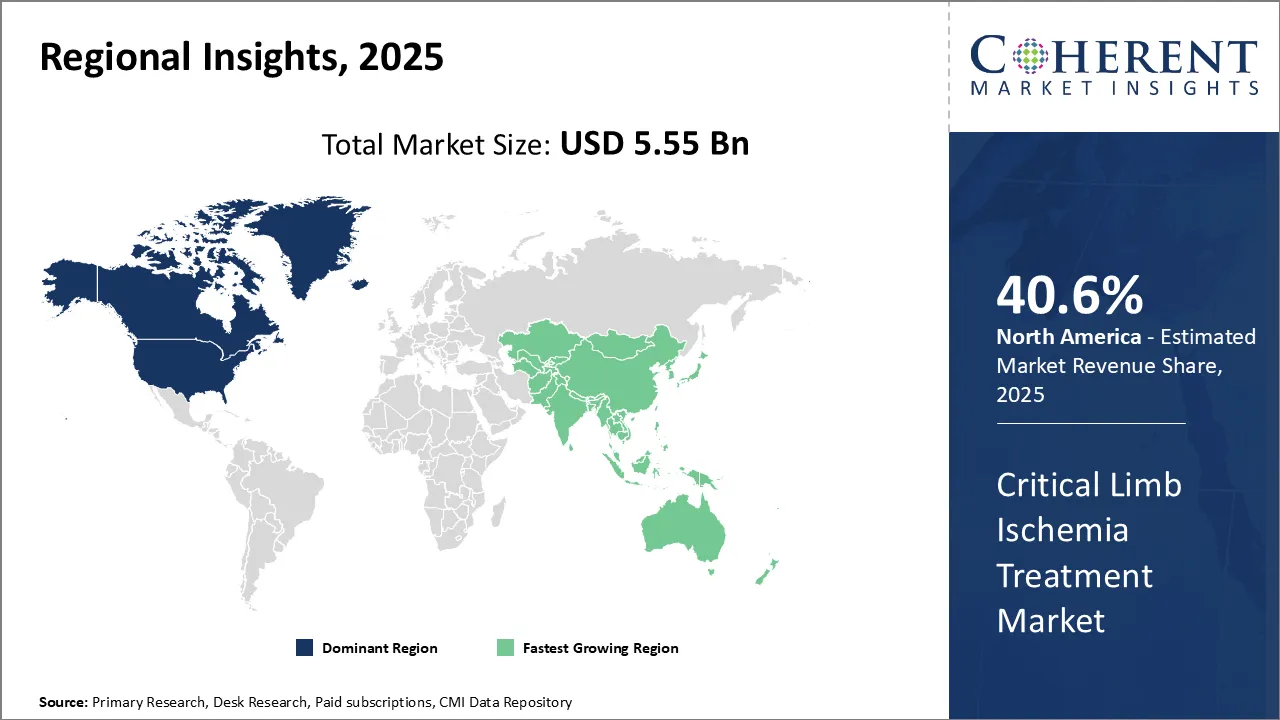
Regional Insights
Need a Different Region or Segment? Request Free Sample
North America currently dominates the global critical limb ischemia treatment market. The region is expected to account for 40.6% in 2026 and is expected to continue its stronghold over the forecast period. The region has a well-established healthcare infrastructure and high healthcare expenditure which enables early adoption of latest medical technologies. There is a strong presence of key market players in the region who continue to invest heavily in R&D for developing advanced treatment options. Additionally, growing geriatric population and associated rise in the prevalence of diabetes and peripheral artery disease have been primary factors driving the growth of treatments in North America.
Among emerging regions, Asia Pacific is poised to exhibit the fastest growth through the next five to seven years. Rapid economic development and improving access to healthcare across major Asian countries have increased demand for critical limb ischemia treatment. India and China together account for over one-third of global diabetes cases, fueling growth opportunities. Initiatives by regional governments to strengthen healthcare systems along with increasing healthcare spending are positively impacting the market growth. Further, growing medical tourism across Asia Pacific due to the availability of low-cost treatment is attracting patients from developed markets of North America and Western Europe. This has encouraged local players to invest in building capabilities to cater to international standards. While pricing pressures exist, the large patient pool and rising affordability are aiding Asia Pacific to emerge as the fastest growing regional market.
Market Report Scope
Critical Limb Ischemia Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 6.05 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 9.0% | 2033 Value Projection: | USD 11.06 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, Cook Medical Inc., Cardiovascular Systems, Inc., Philips Healthcare, B. Braun Melsungen AG, Terumo Corporation, Biotronik SE & Co. KG, Cordis Corporation, Smith & Nephew plc, Stryker Corporation, AngioDynamics, Inc., GE Healthcare, Sanofi S.A., AstraZeneca plc, and Bristol-Myers Squibb Company |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Market Segmentation
- Drugs Insights (Revenue, USD Bn, 2021 - 2033)
- Antihypertensive Agents
- Antiplatelet Drugs
- Antithrombotic Agents
- Lipid-lowering Agents
- Others
- Route Of Administration Insights (Revenue, USD Bn, 2021 - 2033)
- Oral
- Parentral
- End User Insights (Revenue, USD Bn, 2021 - 2033)
- Hospitals
- Specialty Clinics
- Ambulatory Surgery Centers
- Nursing Homes
- Others (Rehabilitation Centers,etc.)
- Regional Insights (Revenue, USD Bn, 2021 - 2033)
- North America
- U.S.
- Canada
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Europe
- Germany
- U.K.
- Spain
- France
- Italy
- Russia
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- Australia
- South Korea
- ASEAN
- Rest of Asia Pacific
- Middle East
- GCC Countries
- Israel
- Rest of Middle East
- Africa
- South Africa
- North Africa
- Central Africa
- North America
- Key Players Insights
- Medtronic plc
- Boston Scientific Corporation
- Abbott Laboratories
- Cook Medical Inc.
- Cardiovascular Systems, Inc.
- Philips Healthcare
- B. Braun Melsungen AG
- Terumo Corporation
- Biotronik SE & Co. KG
- Cordis Corporation
- Smith & Nephew plc
- Stryker Corporation
- AngioDynamics, Inc.
- GE Healthcare
- Sanofi S.A.
- AstraZeneca plc
- Bristol-Myers Squibb Company
Sources
Primary Research Interviews:
- Vascular surgeons and interventional radiologists
- Hospital and ASC procurement managers
- Endovascular device sales reps
- Wound and limb-preservation specialists
Databases:
- iData Research (peripheral vascular)
- GlobalData Medical Devices
- Definitive Healthcare
- IQVIA hospital procurement data
Magazines:
- Endovascular Today
- Vascular Disease Management
- Cardiovascular Business
- Medical Device & Diagnostic Industry (MD+DI)
Journals:
- Journal of Vascular Surgery
- Circulation
- JACC: Cardiovascular Interventions
- European Journal of Vascular and Endovascular Surgery
Newspapers:
- The Wall Street Journal
- Reuters
- Financial Times
- The New York Times
Associations:
- Society for Vascular Surgery (SVS)
- Society of Interventional Radiology (SIR)
- Cardiovascular and Interventional Radiological Society of Europe (CIRSE)
- Society for Cardiovascular Angiography and Interventions (SCAI)
Public Domain Sources:
- U.S. FDA 510(k) and PMA databases
- SEC filings (BD/Bard, Boston Scientific, Cordis, Abbott, Medtronic)
- ClinicalTrials.gov
- CMS Medicare claims for endovascular procedures
Proprietary Elements
- CMI Data Analytics Tool, Proprietary CMI Existing Repository of information for last 10 years.
Share
Share
About Author
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Missing comfort of reading report in your local language? Find your preferred language :
Transform your Strategy with Exclusive Trending Reports :
Frequently Asked Questions
EXISTING CLIENTELE
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients
