The coronavirus treatment drugs market size is anticipated to grow at a CAGR of 13.3% with USD 24.8 Bn in 2026 and is expected to reach USD 62 Bn in 2033. The market is growing with the increasing adoption of oral antiviral treatments and monoclonal antibodies. As per the FDA's 2023 approval, the drugs like Paxlovid (nirmatrelvir/ritonavir) and Veklury (remdesivir) continue to be vital in treating COVID-19, especially among high-risk populations. The continuous clinical trials along with the new treatments like molnupiravir and baricitinib, also contribute to the market’s growth.
On the basis of drug class, the anti-viral drugs segment accounts for the largest coronavirus treatment drugs market share of 45.2% in 2026. The segment’s growth is due to widespread use of both oral and intravenous antiviral treatments such as Paxlovid (nirmatrelvir/ritonavir) and Veklury (remdesivir), which are FDA-approved to reduce the risk of hospitalization and death in patients with mild to moderate COVID-19. These antiviral drugs have played a significant role in managing COVID-19 cases since their authorization.
The recent clinical developments continue to strengthen the position of antiviral treatments. For instance, in November 2022, .
In addition, new monoclonal antibody as well as biologic treatments are navigating regulatory pathways with the FDA, aiming for potential Biologics License Applications (BLAs) for next-generation COVID-19 therapies. These ongoing approvals, clinical trial progress, and the high demand for antiviral prescriptions ensure the continued dominance of the antiviral drug class in the overall market for COVID-19 treatment.
To learn more about this report, Request Free Sample
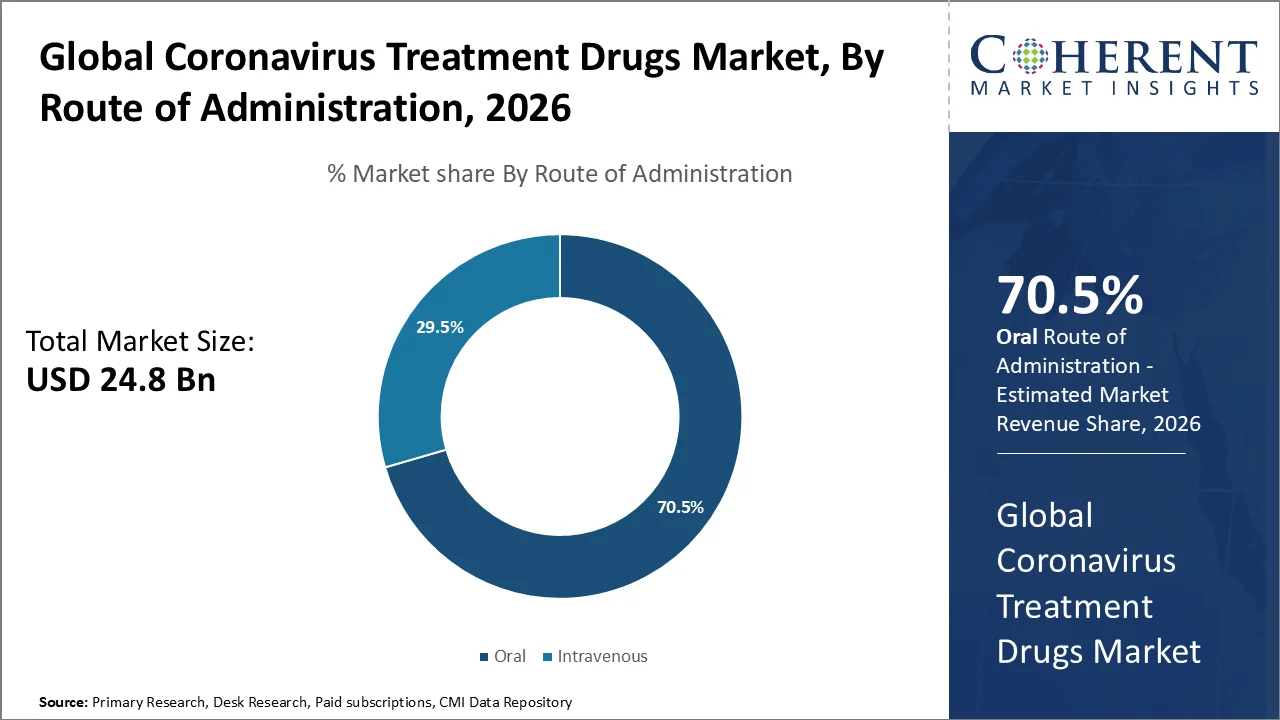
The oral segment accounts for the largest Coronavirus Treatment Drugs Market share of 70.5% in 2026. This growth is driven by the shift toward convenient, early-stage, prescription-based COVID-19 treatment outside hospital settings. The growth is led by oral antivirals which can be initiated soon after diagnosis, thus reducing the burden on infusion centers as well as improving access for high-risk adults.
For instance, in May 2023, the U.S. FDA approved Paxlovid (nirmatrelvir/ritonavir) for mild-to-moderate COVID-19 in adults at high risk of progression to severe disease. Earlier, in December 2021, the FDA granted EUA to molnupiravir (Lagevrio) for certain high-risk adults when other authorized treatments were not accessible or clinically appropriate, thereby broadening oral therapeutic options.
The pipeline momentum further supports this segment, with oral antiviral ibuzatrelvir under Phase III evaluation for COVID-19 treatment. Meanwhile, ensitrelvir/Xocova gained full approval in Japan in March 2024. It became available in Singapore through a special access route in 2023, and has a U.S. FDA PDUFA target date of June 16, 2026 for post-exposure prevention.
The U.S. continues to be at the forefront of COVID‑19 treatment advancements mainly driven by continual research and regulatory updates. The CDC as well as the FDA have authorized several antiviral medications like nirmatrelvir/ritonavir (Paxlovid), remdesivir (Veklury), and molnupiravir (Lagevrio in order to treat mild-to-moderate COVID‑19 in high-risk patients. These treatments are critical to managing COVID‑19, with the CDC recommending their early initiation within five days of symptom onset to prevent progression to severe disease.
The recent real-world data from the CDC reveals that from June 2023 to September 2025, 31% of high-risk COVIDThese statistics highlight the growing role of antiviral medications in preventing severe COVID‑19 outcomes in the U.S.
In addition to these treatments, the ongoing clinical research is continually exploring new antiviral regimens and combination therapies to improve efficacy, particularly for patients with emerging variants of the virus. The National Institutes of Health (NIH) is supporting numerous clinical trials to assess new therapies, including combination treatments that pair antivirals with immune modulators for more robust responses.
COVID-19 Treatment Drugs Market: Breakthrough in RNA ‑Targeted Antiviral Drug Discovery
One of the major breakthroughs in the industry is the development of RNA-targeted antiviral drugs. For instance, in October 2025, the researchers from the Wertheim UF Scripps Institute discovered a promising antiviral compound that specifically targets the viral RNA machinery of SARS-CoV-2. This new drug is a part of a growing wave of next-generation treatments, thus offering the potential for more effective as well as broad-spectrum therapies compared to current drugs. Unlike traditional antiviral drugs, this compound works by interfering directly with the virus’s genetic material and thus prevents any replication.
This advancement could pave the way for more potent treatments for both COVID-19 as well as also for other RNA viruses. It is a considerable step forward as it enhances the ability to treat viral infections with a higher efficacy along with fewer side effects. In addition, the discovery signals a shift in drug development strategies, thereby putting a greater emphasis on genetic mechanisms rather than solely relying on traditional approaches. This innovation is expected to propel further advancements in antiviral therapies as well as boost the market substantially.
|
Current Event |
Description and its Impact |
|
Continued U.S. FDA Emergency Use Authorization Framework for COVID‑19 Therapeutics |
|
|
WHO Ongoing Guideline Development for COVID‑19 Therapeutics |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
To learn more about this report, Request Free Sample
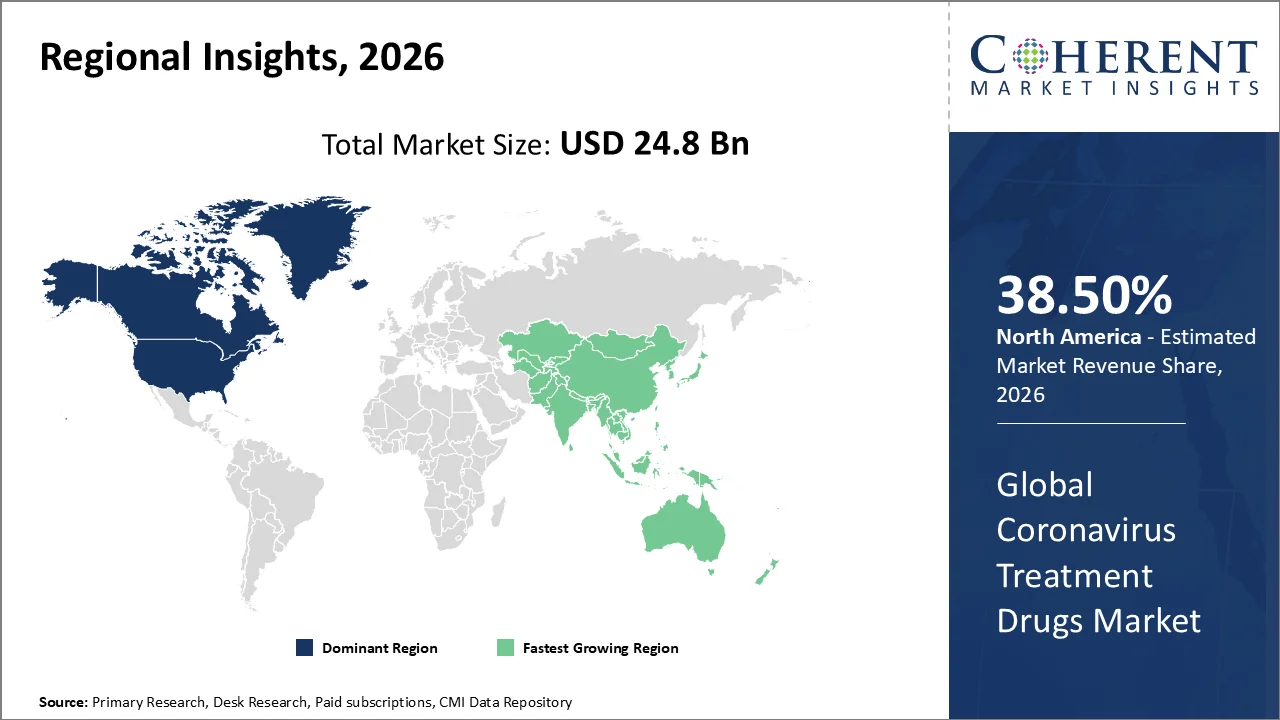
The North America region accounts for 38.50% of the market in 2026. The region’s growth is backed by the region’s broad access to antiviral therapies, strong regulatory frameworks, and ongoing clinical guidance. In the U.S., the Food and Drug Administration (FDA) authorized oral antivirals such as Pfizer’s Paxlovid and Merck’s Molnupiravir for use in non
In Canada, Health Canada has authorized key COVIDThese treatments are available under terms aligned with regulatory review and safety monitoring.
Clinical prescribing guidance in Ontario confirms that oral Paxlovid and intravenous Remdesivir are recommended for eligible high
The Asia Pacific region is poised to be as the fastest-growing region through 2026-2033 driven by continued high-risk patient management, wider oral antiviral access, and regulator-led safety monitoring. The World Health Organization (WHO)'s global COVID-19 risk assessment, released in February 2026, stated that by end-2025, public-health risk remained moderate, while deaths and hospitalizations had declined due to population immunity and improved clinical management, keeping antivirals relevant for vulnerable group.
In China, the National Medical Products Administration (NMPA) granted conditional approval for Pfizer’s Paxlovid for mild-to-moderate COVID-19 adults at higher risk in February 2022 while Azvudine became the country’s first domestically developed oral COVID-19 antiviral under emergency authorization and treatment-protocol inclusion.
Japan is also strengthening prescription governance. The Pharmaceuticals and Medical Devices Agency (PMDA)’s 2024 review supported Xocova Tablets 125 mg, and the Ministry of Health, Labour and Welfare (MHLW) issued precaution revisions for Xocova and Lagevrio in December 2024, including pregnancy-related cautions and Shionogi.
Australia’s Therapeutic Goods Administration (TGA) provisionally approved Paxlovid in January 2022 and Singapore’s Health Sciences Authority (HSA) granted interim authorization in February 2022 for Paxlovid for high-risk adults.
The United States Coronavirus Treatment Drugs Market is driven by updated outpatient care protocols emphasizing early antiviral prescribing for high‑risk COVID‑19 patients. FDA‑approved oral antiviral Paxlovid remains a core treatment option for mild‑to‑moderate COVID‑19 in adults at risk for severe illness, starting within five days of symptom onset.
CDC’s February 2026 analysis shows that from June 2023 through September 2025, 16%–23% of COVID‑19 patients aged ≥65 years received an antiviral prescription during low incidence periods, rising to 37%–38% during high incidence periods, indicating significant seasonal and access variation. Among treated older adults, 99% received antivirals within seven days of diagnosis and 80% were prescribed nirmatrelvir/ritonavir (Paxlovid), while 13% received molnupiravir and 7% received remdesivir, according to official outpatient electronic health record data.
These trends signal expanding utilization yet persistent underuse relative to eligible high‑risk populations.
India’s Coronavirus Treatment Drugs Market continues to be shaped by official clinical guidance and real‑time surveillance of COVID‑19 caseloads, while domestic regulatory agencies maintain oversight of approved antiviral therapies. According to the Ministry of Health and Family Welfare (MoHFW) COVIDThe MoHFW has published national clinical management guidance updating COVID‑19 protocols, and earlier advisories from MoHFW recommended rational use of remdesivir in select moderate to severe hospitalized patients as a reserve antiviral, underscoring structured utilisation in treatment settings. Meanwhile, India's Drug Controller General has granted accelerated approval of remdesivir (e.g., COVIFOR) for restricted medical use to meet urgent treatment needs during the pandemic response.
Some of the major key players in Coronavirus Treatment Drugs are Microsoft, Facebook, Inc., Apple, Inc., Oracle, Seclore, Fasoo, VERA, Adobe Inc., Open Text Corporation, DivX, LLC, HP Labs, Dell Inc.,VOBILE INC., RealNetworks, Inc., IBM Corporation, General Electric, Axtia Technologies, Union FinTech, and Conax Technologies
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 24.8 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 13.3% | 2033 Value Projection: | USD 62 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Serum Institute of India, Pfizer Inc., AstraZeneca plc., Bharat Biotech International Limited, Cadila Healthcare Limited, Johnson & Johnson, Moderna, GlaxoSmithKline plc., CanSino Biologics, Sinovac Biotech, Novavax, Inc., Glenmark Pharmaceuticals Limited, Merck & Co., Inc. and Dr. Reddy's Laboratories. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients