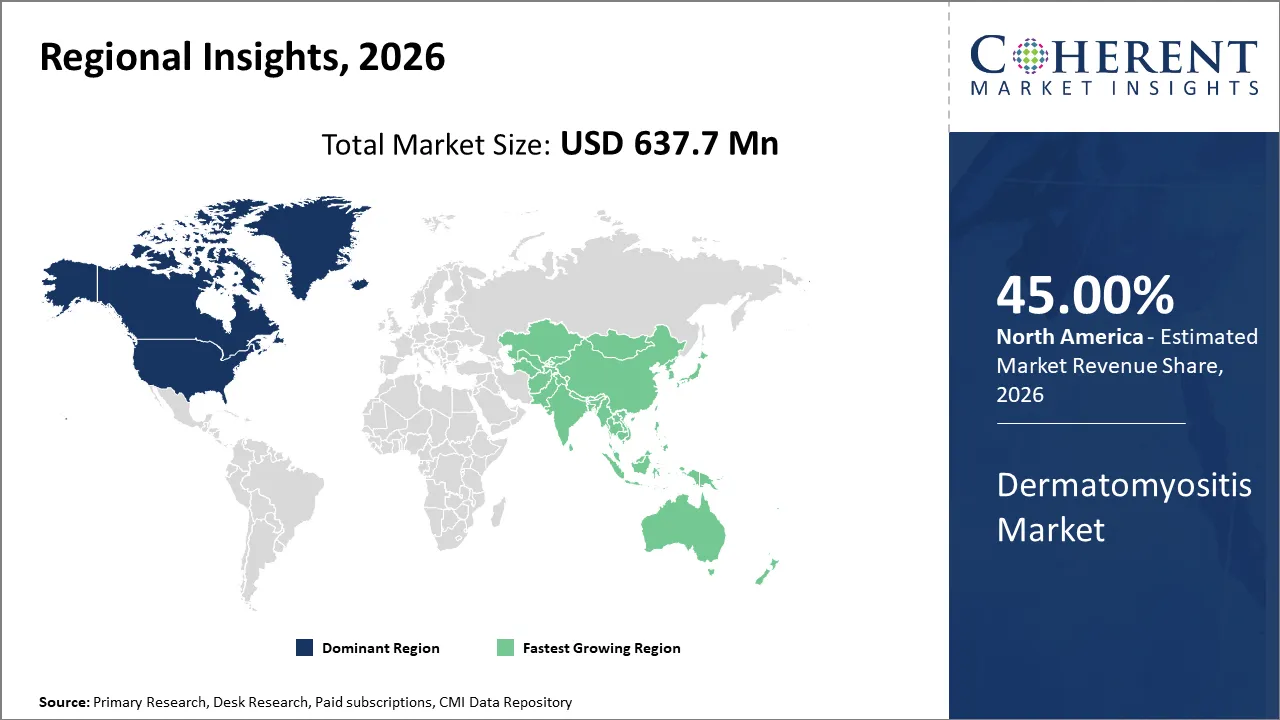
The global dermatomyositis market size is expected to reach approximately USD 637.7 Mn in 2026 and USD 931.6 Mn by 2033, growing at a CAGR of 5.5% throughout the forecast period (2026-2033)
An inflammatory autoimmune disorder characterized by muscle weakness and skin rash is called dermatomyositis. Dermatomyositis attacks muscle and skin, causing muscle weakness, fatigue, and a characteristic skin rash; in some patients, it can also affect the lungs, swallowing, and overall physical function. The dermatomyositis market is mainly driven by rising recognition of the disease’s high unmet need, because patients often require long-term treatment and may also develop serious complications such as interstitial lung disease, dysphagia, and malignancy-associated disease. The dermatomyositis market is gaining strategic importance because it has high unmet clinical need, complex diagnosis, and long-term management requirements, which are driving demand for earlier detection, specialist care, biomarker-led testing, and targeted therapies. Its orphan-disease status, growing clinical research activity, and increasing focus on precision immunology are also encouraging innovation of new drug, partnerships, and premium treatment positioning.
|
Current Event |
Description and its Impact |
|
Increasing drug approval by the regulatory bodies |
|
|
Launch of Advanced Therapy for Treatment |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Based on type, adult dermatomyositis is expected to lead the market, accounting for a revenue share of 60% in 2026. The adult dermatomyositis segment dominates because the adult patient pool is larger than the pediatric pool, and adults more often present with complications that require broader, longer, and more intensive medical management. The increased clinical complexity of adult illness is another important factor. While juvenile dermatomyositis is relatively infrequently linked to malignancy, adult dermatomyositis is much more strongly associated with cancer, and reviews point out that cancer-associated dermatomyositis is a well-established problem in adults. Since more regular screenings, imaging, specialist consultations, and longer follow-ups are required, this increases the market demand for adult care.
By treatment type, the anti-inflammatory medication segment is slated to account for a prominent market share of 50% in 2026. The anti-inflammatory medications are extensively used for dermatomyositis treatment owing to their first-line role in disease management and rapid action on suppression of muscle and skin inflammation. Another important factor is that most patients get anti-inflammatory drugs early and often, no matter how bad their disease is. High-dose glucocorticoids are frequently used in conjunction with methotrexate or other immunosuppressants for treating dermatomyositis in both adults and children, according to guidelines by the regulatory bodies. Because inflammation control is the cornerstone of treatment, particularly at diagnosis and during flare-ups, the anti-inflammatory medicine segment dominates the dermatomyositis market. Systemic glucocorticoids continue to be the first-line treatment for dermatomyositis, according to current reviews and clinical references. This is because systemic glucocorticoids operate fast to reduce muscle inflammation, relieve weakness, and control active illness before longer-term, steroid-sparing therapies fully take effect.
Based on distribution channel, hospital pharmacies segment led the global dermatomyositis market with share of 50% in 2026. The hospital pharmacies segment dominates as the dermatomyositis treatment often involves specialist-managed therapies such as immunosuppressants, corticosteroids, biologics, and especially intravenous immunoglobulin (IVIg), which commonly require hospital-based prescribing, monitoring, infusion administration, and reimbursement control. Because dermatomyositis is a rare autoimmune disease that usually requires specialist-led treatment, including IVIg infusions and other advanced immunomodulatory therapies, which are primarily dispensed and monitored through hospital settings rather than retail pharmacies, the hospital pharmacies segment dominates the dermatomyositis market distribution landscape.
To learn more about this report, Request Free Sample
North America is expected to dominate the global dermatomyositis market, accounting for a share of 45% in 2026. The region benefits from a well-developed network of specialized myositis centers and referral hospitals in the U.S. This network supports earlier diagnoses, collaborative care involving multiple medical specialties, and greater use of advanced treatments. Due to availability of advanced infrastructure and presence of leading key players, there is greater use of advanced infusion-based therapy supports higher market value. The growth of the North America dermatomyositis market is driven by strong specialist care availability, robust hospital and infusion infrastructure, higher awareness of rare autoimmune diseases, and the presence of U.S.FDA-approved treatment options, all of which improve diagnosis rates, treatment uptake, and long-term disease management in the region.
Asia Pacific region is expected to be the fastest growing region in the dermatomyositis market in the near future due to rising research and development activity and increasing adoption of advanced therapeutic treatment. For instance, in August 2025, Pfizer, a pharmaceutical company, announced the positive response of the dazukibart drug for the treatment of dermatomyositis and polymyositis and its enrollment in the phase 3 clinical trial. This trial has multiple recruiting sites in Asia‑Pacific, including China (e.g., Anhui Provincial Hospital, Peking Union Medical College Hospital, Renji Hospital Shanghai Jiao Tong University), India (Ernakulam, Kerala), and Japan (Tokyo) reflecting Asia‑Pacific participation. According to evaluation published by the National Center for Biotechnology Information evaluations in 2024, biologics, JAK inhibitors, and other more recent therapy modalities for dermatomyositis and juvenile dermatomyositis are gaining popularity. Regional market expansion quickens as Asia Pacific's tertiary-care facilities increase access to these more expensive treatments. Access to immunology testing, the expansion of tertiary hospitals, and rheumatology/dermatology referral routes are all contributing to the diagnosis and treatment of previously overlooked or postponed cases. This is not evidence of present market domination, but rather a market-growth element. This trend of change is supported by the research on diagnostic delay and current advancements in classification.
The U.S. country dominates within the North America dermatomyositis market due to large patient pool increase the burden for the treatment. According to the research data published in 2024 by the National Center for Biotechnology Information, the U.S. has one of the highest reported rates of adult dermatomyositis incidence and prevalence worldwide, with an adult incidence of 15.2 per million person-years and a prevalence of 21.4 per 100,000, indicating a higher demand for treatment than other nations in the region. Coordinated care from rheumatology, dermatology, neurology, pulmonology, and infusion-based treatment is frequently necessary for dermatomyositis. Due to the presence of advanced infrastructure and an advanced specialized environment for treating myositis problems is shown in U.S.-based facilities like the Johns Hopkins Myositis Center and The Myositis Association's physician network, clinical research is more active in the U.S. In the U.S., ClinicalTrials.gov lists numerous dermatomyositis studies and extension trials, demonstrating increased ongoing research activity, early acceptance of innovative medicines, and improved access to experimental treatment alternatives.
China dermatomyositis market is driven by the growing epidemiology and launched of advanced therapy in the country. China's policy framework has progressively supported rare-disease medications through insurance inclusion, price negotiations, VAT reductions, quicker review/approval support, and hospital formulary requirements for collaborative-network institutions. This facilitates the conversion of clinical need into real commercial demand. Anti-MDA5 dermatomyositis is comparatively more common in Asian populations, which is a clinically significant factor. According to a 2025 review, it is responsible for more than 20% of occurrences of dermatomyositis in Asians, and 50% to 100% of Asian MDA5-DM patients develop fast progressive interstitial lung disease. This raises the need for specialized diagnosis, hospitalization, immunosuppressive treatment, and follow-up. The Asia-Pacific dermatomyositis market is dominated by China because of its large addressable patient pool, extensive network of tertiary hospitals, improved infrastructure for diagnosing and referring rare diseases, improved access to treatment through policy support, and increased clinical demand for specialized management of severe dermatomyositis subtypes.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 637.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.5% | 2033 Value Projection: | USD 931.6 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
AstraZeneca Plc., Bristol-Myers Squibb Company, Octapharma AG, Corbus Pharmaceuticals Holdings, Inc., CSL Limited, Ermium Therapeutics, Kezar Life Sciences Inc., Mallinckrodt Plc., Néovacs, Horizon Therapeutics plc., and Pfizer Inc. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Expansion of funding activities and inflammatory myopathies has estimated to drive the growth of the dermatomyositis market by improving awareness, stimulating research, enabling infrastructure, and ultimately expanding both treatment development and patient access. The funding aids in the discovery of new patient subgroups, biomarkers, and treatment targets. It can aid in the initiation of clinical studies, increasing the possibility that novel treatments will be approved by regulators. Early funding lowers the cost of entry for new biotech firms looking to create therapies tailored to diabetes mellitus. In combination, non‑profit funding drives growth in the dermatomyositis market by increasing diagnosis rates and patient identification, enhancing reimbursement and policy support, and attracting broader investment and commercial interest.
For instance, in December 2025, according to the data published by The Myositis Association, has funded 68 international research projects with close to USD10 million. From basic science to clinical applications, these initiatives cover a wide range of myositis-related illnesses and specialties. Our average grant award is over USD120,000, which is a significant investment that has helped patients find fresh hope, develop research programs, and establish careers. 43% of TMA-funded initiatives have benefited non-U.S. investigators, demonstrating our dedication to international cooperation. Early-career researchers have benefited from 32% of funds, fostering the development of the next generation of myositis specialists. Many of the 22 Fellowship Grants that have been given out have resulted in long-term leadership in the industry.
Increasing approval and launch of new drug product has estimated to drive the growth of the market over the forecast period. By enhancing treatment credibility, reimbursement access, physician adoption, patient identification, and pipeline investment, product launches and regulatory approvals hasten the growth of the dermatomyositis market and transform an underpenetrated rare-disease segment into a more organized commercial market. Additionally, approvals benefit the market by increasing diagnosis and awareness, motivating specialists to find qualified patients sooner, and driving treatment facilities to develop regimens tailored to individual diseases. Successful launches can draw increased interest from investors and R&D.
For instance, in March 2026, Priovant Therapeutics, a clinical-stage biotechnology company, unveiled that the U.S. Food and Drug Administration (FDA) has approved New Drug Application (NDA) for brepocitinib to treat dermatomyositis (DM) and has given the application Priority Review. The third quarter of 2026 has been designated by the FDA as the Prescription Drug User Fee Act (PDUFA) target action date. By the end of September 2026, the business plans to introduce the medication in the US.
Due to the rising prevalence of dermatomyositis, there is increase in the demand for the biologic therapies. Growth in immunosuppressants and precision immunology treatment is major structural driver of the dermatomyositis market because both expand the patient pool, improve treatment outcomes, and attract pharmaceutical investment. As the precision immunology medicines target specific immune pathways instead of broad suppression it has higher demand in the market. Expansion of combination treatment strategies enables better disease management and driving market adoption.
Growing emergence in regions like Asia Pacific, Latin America, and the Middle East are experiencing rapid growth in healthcare access, driven by significant investments in specialized care centers and rheumatology departments. The increasing demand for diagnostic tools and targeted therapies will stimulate growth in the market. The ability to detect and manage dermatomyositis early will increase the adoption of both existing and novel treatments. Healthcare infrastructure development will allow wider access to treatments, enabling the use of innovative drugs, expanding patient access, and accelerating market penetration. Therefore, expansion in emerging markets has created significant opportunity for the growth of the dermatomyositis market in the near future.
Increasing prevalence and launch of advanced technology for the diagnosis of the dermatomyositis has created significant opportunity for the growth of the in the near future. For instance, in January 2026, according to the data published by the National Center for Biotechnology Information and Fortune journals, it has been estimated that a rare inflammatory myopathy, dermatomyositis is expected to have an annual incidence rate of 1 to 15 cases per million people and a prevalence of 2 to 20 cases per 100,000 people. Early diagnosis makes therapy possible, which is essential for slowing the progression of the disease and averting consequences. Patients may have better long-term results if treatment is started early in the illness. The precision and promptness of diagnoses are being greatly enhanced by technological developments in diagnostic instruments including MRI, skin biopsy methods, and autoantibody tests. More early-stage diagnoses will result from this increased awareness, enabling earlier therapies and improved patient outcomes.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients