Infertility is a global health issue as its affects both men and women. Female infertility is defined as the condition wherein a female cannot get pregnant even after one year of unprotected sexual activity. Infertility in females occur due to rising age, polycystic ovarian syndrome (PCOS), obesity, and multiple miscarriages.
Global Female Infertility Treatment Drugs Market - Impact of the Coronavirus (COVID-19) Pandemic
Coronavirus (COVID-19) outbreak was first reported on December 31, 2019, in Wuhan, China. The World Health Organization declared COVID-19, as pandemic on March 11, 2020. According to the Coronavirus Disease (COVID-19) Weekly Epidemiological Update by the World Health Organization, over 169 million cases and 3.5 million deaths due to coronavirus disease (COVID-19) were reported till May 30, 2021 across the globe.
Impact of COVID-19 on Demand and Supply of Female Infertility Treatment Drugs
The emergence of the global pandemic has triggered fear that the global and extended pharmaceutical supply chain would crumble. In practice, it has proved remarkably resilient with drug shortages kept to a minimum. In addition, the pandemic has exposed some weaknesses in the supply chain that must be addressed as the world recovers and prepares for what the future holds.
The heavy reliance on China for raw materials, intermediates, and APIs as well as India for generic drug products remains a significant concern. Distribution is also affected by disruptions in the global supply chain, as are clinical trials. A high level of ambiguity is expected to remain until the spread of the SARS-CoV-2 virus is halted.
Furthermore, this heavy reliance of drugs leads to drug shortage in the U.S. For instance, the U.S. FDA reported an increase in the number of drug shortages from 2017 to 2018, a contrast to the declines observed since 2011. The drug shortages are lasting longer and can be even more than eight years in some cases. Over 60% of shortages in 2019 were attributed to supply disruptions due to manufacturing and/or quality issues due to covid-19 pandemic.
In addition to the reduction in manufacturing activity, the pharmaceutical supply chain is being impacted by distribution challenges created by reduced transportation capacity. Disruptions at ports, quarantined crews, and labor shortages due to lockdowns are all contributing factors.
The global female infertility treatment drugs market is estimated to be valued at US$ 2,649.8 Mn in 2021 and is expected to reach US$ 3,512.8 Mn by 2028, registering a CAGR of 4.1% during the forecast period (2021-2028)
Figure 1: Global Female Infertility Treatment Drugs Market Y-o-Y Growth Comparison (%), by Route of Administration, 2018-2028
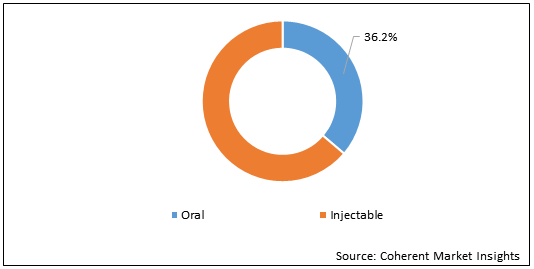
To learn more about this report, Request Free Sample
The increasing rate of infertility is the major factor that is expected to drive the market growth over the forecast period.
Factor such as the increasing infertility rates globally is expected to augment the growth of global female infertility treatment drugs market over the forecast period. For instance, according to the report published in Galadari Printing and Publishing LLC, 2018, the prevalence of infertility increased with a significant decline in demographics ranging from 6.6 births per woman in the 1970s to 5.22 children in 1980-1985 and 1.73 from 2015-2020, registering an overall decrease of 160%.
Moreover, increasing number of research and development activities for improving the effectiveness of female infertility treatment drugs is expected to aid in the growth of the market over the forecast period. For instance, in October 2018, Myovant Sciences, a subsidiary of Sumitovant Biopharma, announced the presentation of data from a Phase 1 trial of MVT-602, a novel kisspeptin-1 receptor agonist in development as a potential treatment for female infertility in women as part of assisted reproduction, such as in vitro fertilization (IVF) at 2018 American Society for Reproductive Medicine (ASRM) Annual Congress. Results of the study showed that administration of MVT-602 in healthy premenopausal women in the follicular phase produced a dose-related increase in luteinizing hormone concentrations and expected effects on follicle-stimulating hormone and estradiol).
The rising number of product approvals and launches support the research and development of new drugs for female infertility treatment which is expected to drive the growth of the global female infertility drugs market over the forecast period. For instance, in February 2016, Merck KGaA, a Germany-based science and technology company in healthcare, life science and performance materials, received approval from the European Medicines Agency (EMA) for the GONAL-f prefilled pen 2.0. GONAL-f (follitropin alfa), a recombinant human follicle-stimulating hormone (r-hFSH) approved for the treatment of infertility in both males and females. In November 2018, Merck & Co. Inc., announced that the company received approval for a new version of GONAL-f (follitropin alfa injection) prefilled pen from the U.S. Food and Drug Administration (U.S. FDA). Follitropin alfa helps stimulate healthy ovaries to produce eggs. It is known as GONAL-f RFF Redi-jectTM prefilled pen in the U.S.
In addition, increasing number of available reimbursement facilities by manufacturer and government propel the market growth of female infertility treatment drugs. For instance, Sanofi S.A. provides reimbursement for CLOMID under the Pharmaceutical Benefits Scheme (PBS) in Europe and Australia, in order to make the medicines available at affordable prices for patients. PBS provides timely, reliable, and affordable access to fertility medications to the people in Europe and Australia. The medicine is prescribed for the treatment of ovulatory failure in selected infertile women who wish to become pregnant.
Female Infertility Treatment Drugs Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2020 | Market Size in 2021: | US$ 2,649.8 Mn |
| Historical Data for: | 2017 to 2020 | Forecast Period: | 2021 to 2028 |
| Forecast Period 2021 to 2028 CAGR: | 4.1% | 2028 Value Projection: | US$ 3,512.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Ferring Pharmaceuticals, Merck & Co., Inc., Bayer AG, Abbott Laboratories, Par Pharmaceutical, Inc., Sumitovant, Livzon, Zydus Cadila, Oxolife, and Mankind Pharma |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Female Infertility Treatment Drugs Market – Restraints
However, growth of the female infertility drugs market may be hampered, owing to the risks associated with female infertility treatment drugs and increasing number of product recalls. For instance, on March 2, 2021 Ferring Pharmaceuticals voluntarily initiated recall of their infertility treatment drug Menopur 75 IU.
Global Female Infertility Treatment Drugs Market – Regional Analysis
On the basis of region, the global female infertility treatment drugs market is segmented into North America, Latin America, Europe, and Asia Pacific.
North America is expected to show growth in the global female infertility drugs market over the forecast period, owing to rising number of product launch and increase in female infertility rates. For instance, according to the Centers for Disease Control and Prevention (CDC) report of 2018, around 6% of women aged 15 to 44 years in the U.S. were unable to conceive after one year of continuous intercourse.
Europe female infertility treatment drugs market is estimated to be valued at US$ 1,035.1 Mn in 2021 and is expected to increase to US$ 1,355.0 Mn by 2028, exhibiting a CAGR of 3.9% during the forecast period. Approval of novel products by regulatory authorities and availability of government or private policies for aiding infertility treatment in the region is expected to drive growth of the Europe female infertility treatment drugs market in the near future. For instance, National Health Service (NHS), a government owned organization provides free infertility treatment to women. Private players such as Manchester Fertility Services Ltd. and CARE Fertility are involved in providing financial assistance for infertility treatment in U.K.
The female infertility treatment drugs market in the Asia Pacific is expected to gain momentum during the forecast period, owing to an increasing prevalence of infertility rates in the Asian population. For instance, according to the Indian Society of Assisted Reproduction (ISAR) report of 2017, infertility affects around 10 to 14% of the Indian population, with higher rates in urban areas where one out of six couples are affected from it.
Figure 2: Global Female Infertility Treatment Drugs Market Y-o-Y Growth (%) Comparison, by Region, 2018 – 2028
To learn more about this report, Request Free Sample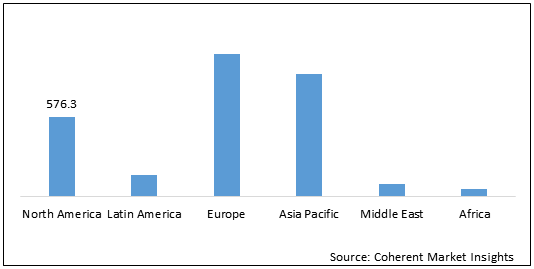
Global Female Infertility Treatment Drugs Market – Competitive Landscape
Major players operating in the global female infertility treatment drugs market include Ferring Pharmaceuticals, Merck & Co., Inc., Bayer AG, Abbott Laboratories, Par Pharmaceutical, Inc., Sumitovant, Livzon, Zydus Cadila, Oxolife, and Mankind Pharma.
Share
Share
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients