The Europe allogenic human chondrocyte market is anticipated to grow at a CAGR of 10.4% with USD 3,122.1 Mn in 2026 and is expected to reach USD 6,161.9 Mn in 2033. The Europe allogeneic human chondrocyte market is driven by the high burden of osteoarthritis (OA) and musculoskeletal disorders among aging populations, particularly knee and hip OA, where prevalence reaches about 10% for knee and 6% for hip in Europe, as reported in a systematic review published in January 2026. In July 2022, according to the data published by the WHO, musculoskeletal conditions constitute a leading cause of disability and rehabilitation need across the Europe region. Increasing demand for regenerative therapies reflects unmet clinical needs and growing orthopedic interventions.
Source: Springer Nature; WHO
Source: NCBI
Osteoarthritis is projected to account for the largest share of cognitive systems spending in 2026, representing approximately 50.0% of the total volume. The osteoarthritis (OA) segment is the dominant application segment in the Europe Allogeneic Human Chondrocyte Market due to the increasing prevalence of Osteoarthritis among the aging population and the high demand for effective, long-term solutions for joint pain and mobility. OA, particularly in the knee, is a leading cause of disability in Europe, with millions of people affected annually. The limitations of traditional treatments, such as pain management and invasive surgeries, have driven interest in allogeneic human chondrocyte therapies. These therapies offer the potential for regenerative healing, addressing cartilage degeneration and restoring joint function, making them a highly sought-after treatment option for Osteoarthritis patients. The growing focus on minimally invasive, cost-effective regenerative therapies also enhances the segment's dominance. A 2026 systematic review estimated that the pooled prevalence of knee osteoarthritis in Europe is approximately 10% (95% CI: 7–14%), making it one of the most common joint disorders across the continent. Knee Osteoarthritis prevalence varied by region, reaching up to 19% in Eastern Europe, highlighting its significant burden among adults. Hip Osteoarthritis affected around 6% of the European population in the same analysis. Furthermore, in 2025, according to the data published by the Oxford University Press, population‑level data show that older adults in Germany (aged over 60) have a hip and knee OA prevalence of roughly 21.8%, with higher rates in women at 23.9%.
Moreover, in 2026, according to the data published by the National Institute for Health and Care Excellence, in the UK, it is estimated that 10 million people are affected by Osteoarthritis, with 6 million women and 4 million men experiencing the condition. Of these, 5.4 million people are impacted by knee Osteoarthritis specifically, making it one of the most common joint diseases. Additionally, 3.2 million people suffer from Osteoarthritis in other joints, such as the hip and hand.
To learn more about this report, Request Free Sample
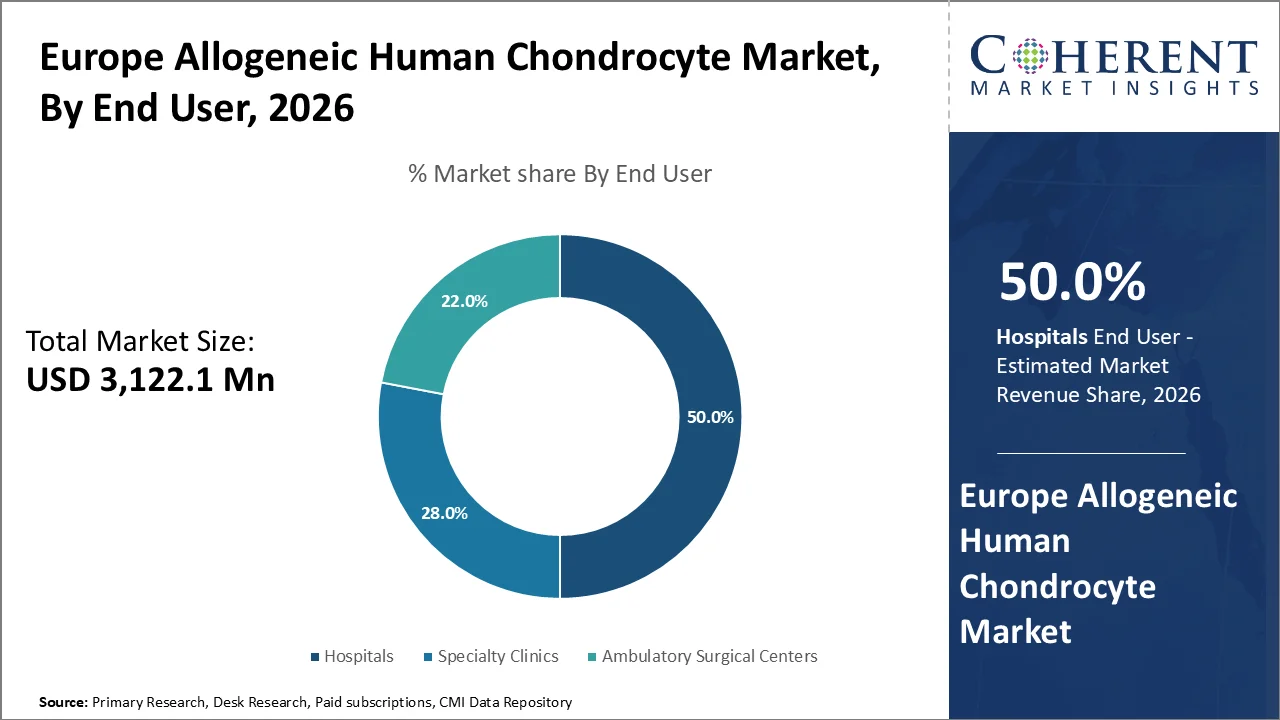
Based on end user, hospitals dominate the market, accounting for a significant 50.0% share in 2026. Hospitals are equipped with advanced infrastructure and skilled personnel to perform complex, high-risk procedures like allogeneic chondrocyte implantation, which requires specialized facilities for cell-based therapies. They also provide a comprehensive care pathway, from diagnosis and surgery to post-operative rehabilitation, making them the preferred setting for regenerative therapies. Additionally, hospitals often benefit from government reimbursement models that cover the costs of innovative treatments, further driving patient access. The high volume of patients with musculoskeletal disorders, particularly osteoarthritis, in hospital settings strengthens this segment’s position as the main driver for the adoption of chondrocyte-based therapies.
A 2026 Annals of the Rheumatic Diseases analysis highlights that workers with rheumatic and musculoskeletal disorders (which include osteoarthritis) have significantly reduced healthy working life expectancy compared to those without, illustrating the ongoing and increasing clinical impact of these conditions on populations across Europe. For instance, in 2024, according to the data published by the Organisation for Economic Co-operation and Development, on average across EU countries in 2022, there were 202 hip replacements per 100 000 population and 134 knee replacements per 100 000 population performed as elective surgical procedures. According to a NOCA report from April 2026, OA is the main cause of thousands of hip and knee replacements performed in Ireland each year, with 94% of hip replacement patients and 97% of knee replacement patients citing OA as the reason for surgery.
The increasing prevalence of musculoskeletal system disorders, especially osteoarthritis, is a major driver of the Europe allogeneic human chondrocyte market growth. With over 22% of the European population affected by musculoskeletal disorders, the demand for effective treatments is surging. In particular, osteoarthritis, which affects 10% of the adult population, leads to significant joint degeneration, creating a large patient pool in need of regenerative therapies like allogeneic chondrocyte implantation. With early intervention becoming key, the rising incidence of joint-related issues is accelerating the adoption of cutting-edge therapies across Europe. A 2026 WHO report highlights musculoskeletal disorders as a leading cause of disability in the region. For instance, in 2024, the prevalence of people aged 16 and over self‑reporting a long‑term MSK condition in England was 17.9% (~almost 1 in 5 adults).
Furthermore, according to the data published by the Carrothers Orthopaedics, Orthopedic clinic in Cambridge, England an Osteoarthritis update reported that in 2025, about 4.6 million people in England had knee osteoarthritis and 2.76 million had hip osteoarthritis showing strong need for regenerative therapies.
Increasing falls and knee cartilage injuries are expected to drive the Europe Allogeneic Human Chondrocyte Market by expanding the patient pool needing cartilage repair and regenerative therapies. For instance, in 2026, according to the data published by the European Association for Injury Prevention and Safety Promotion, in Europe, falls remain a major injury burden among older adults; around one-third of people aged over 65 fall every year, rising to 50% among those aged 80+, while nearly 3.8 million older Europeans receive hospital treatment after falls annually. Knee trauma from falls, sports, and accidents often damages articular cartilage, which has limited natural healing capacity. Furthermore, in March 2026, according to the data published by the National Center for Biotechnology Information, clinical evidence shows cartilage defects are common during knee arthroscopy, with studies reporting cartilage lesions in nearly 60% of arthroscopy patients. This increases demand for chondrocyte-based repair solutions across Europe.
Innovation is transforming the Europe Allogeneic Human Chondrocyte Market by shifting cartilage repair from complex, patient-specific procedures toward scalable and ready-to-use regenerative platforms. Allogeneic chondrocyte progenitor therapy is being studied in Europe to simplify adoption, improve patient access, and reduce treatment burden compared with conventional autologous chondrocyte implantation. Cell banking, donor screening, GMP-based expansion, and controlled manufacturing are helping companies create more consistent chondrocyte batches for wider orthopedic use. Scaffold-based constructs, injectable cell carriers, and 3D tissue-engineered matrices are also improving cell retention, cartilage integration, and surgeon handling during knee repair. EMA’s ATMP framework supports structured evaluation of cell- and tissue-based cartilage therapies, while earlier EU cartilage repair approvals validated the regulatory pathway for advanced chondrocyte products. These innovations are improving standardization, reducing procedure complexity, and strengthening adoption across European orthopedic centers.
For instance, as of April 2026, University of Birmingham is leading a Phase II clinical trial initiated in 2021, focused on allogeneic chondrocyte progenitor therapy for knee cartilage repair. This research, funded by the UK Medical Research Council, aims to streamline cartilage repair treatments, bypassing the traditional autologous two-stage process. The trial could improve accessibility and treatment outcomes for knee cartilage injuries across the NHS and European healthcare systems.
Furthermore, for instance, Synartro AB, a Swedish clinical‑stage life science company, is advancing its lead investigational therapy SYN321 a locally administered intra‑articular injection designed to provide sustained release of diclofenac directly into the knee joint for treatment of symptomatic knee osteoarthritis (OA). The first‑in‑human Phase 1/2a clinical trial of SYN321 was initiated in August 2025 following regulatory and ethical approvals by Swedish authorities, with patient dosing completed and study follow‑up finishing in early 2026. In April 2026, Synartro announced positive preliminary data from the Phase 1/2a study, showing that SYN321 was generally well tolerated across evaluated dose levels, with low systemic exposure and early signals of clinical benefit.
|
Current Event |
Description and its Impact |
|
EU HTA Regulation Supporting Faster Market Access for ATMPs |
|
|
SoHO Regulation Strengthening Cell and Tissue Safety Standards |
|
|
Rising Funding Activity in Europe |
|
|
Increasing Government Initiative |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
The prevalence of osteoarthritis (OA) in Europe is substantial, with an estimated 10% prevalence of knee OA and 6% for hip OA across European populations, based on pooled data from studies involving millions of individuals. This high disease burden underlines the need for advanced cartilage repair solutions like allogeneic human chondrocyte therapies.
Osteoarthritis prevalence shows strong regional differences within Europe: knee OA ranges from about 7% in Northern Europe up to 19% in Eastern Europe, while hip OA ranges from approximately 2% to 7% depending on the region. These variations highlight uneven clinical demand and adoption rates for therapies across the continent.
Across Western Europe alone, around 57 million people live with OA, a figure that has risen significantly over the past decades, showing increasing clinical awareness and need for regenerative solutions to manage pain and disability in aging populations.
Europe is moving toward allogeneic “off-the-shelf” chondrocyte therapies because they avoid patient-specific cell harvesting and may reduce the burden of two-stage autologous chondrocyte implantation. For instance, in 2026, the UKRI-funded ACT2 Phase II project states that it will “manufacture and implant an allogeneic chondrocyte progenitor ATMP” for cartilage repair and aims to improve NHS adoption and patient access compared with ACI. This supports the trend toward ready-to-use cartilage repair products in Europe.
Cartilage repair is increasingly linked with arthroscopic and shorter-stay procedures because hospitals are trying to reduce inpatient pressure. Eurostat reported around 46.9 million day-case discharges in the EU in 2023, compared with 68.2 million inpatient hospital discharges, showing the strong role of same-day care models in Europe. This supports future adoption of minimally invasive chondrocyte-based procedures where clinically suitable.
Europe’s healthcare systems are shifting appropriate surgical activity from inpatient hospitals to day-care settings. Eurostat defines surgical procedures as being performed as inpatient cases, day cases, or outpatient cases, confirming that Europe already tracks and supports these care models. This is important for allogeneic human chondrocyte therapies because standardized, ready-to-use products can fit better into shorter orthopedic workflows than complex autologous cell procedures.
Advanced tissue engineering is becoming central to cartilage repair innovation. In 2025, researchers working on allogeneic chondrocyte scale-up highlighted that allogeneic chondrocyte therapies are being developed as an alternative to autologous therapies for cartilage defects. The research focuses on juvenile and adult donor-derived chondrocytes, culture expansion, and scalable manufacturing, supporting the shift toward engineered cartilage repair platforms.
Clinical research activity is increasing because ATMPs require strong clinical evidence before market adoption. EMA’s updated ATMP clinical trial guideline, published in January 2025, states that ATMPs include somatic cell therapy and tissue-engineered products, and that well-conducted clinical trials are essential to determine their benefit-risk profile. This directly supports continued R&D investment in chondrocyte-based cartilage repair therapies across Europe.
Manufacturing scale-up is a key trend because allogeneic products need consistent donor-cell sourcing, expansion, and potency control. The UKRI MICA project focuses on identifying optimal healthy allogeneic chondrocyte sources and optimizing GMP chondrocyte expansion protocols using the Quantum system. This shows how European research is targeting scalable, reproducible, and lower-burden manufacturing for future allogeneic cartilage therapies.
Germany account 30.0% market share in 2026, supported by advanced healthcare infrastructure and regulatory support. Germany is home to one of the most advanced healthcare systems in Europe, with a strong presence of specialized orthopedic centers and hospital networks that support the clinical adoption of cutting-edge regenerative therapies, including allogeneic human chondrocyte therapies. Osteoarthritis (OA) is highly prevalent in Germany, with over 6.5 million people suffering from OA, particularly knee OA, which leads to a growing demand for cartilage repair solutions. The country’s aging population further increases the prevalence of OA, driving the demand for more effective treatments.
Germany shows a high burden of osteoarthritis (OA) relative to its population, which drives demand for cartilage repair and regenerative therapies including allogeneic human chondrocyte approaches. A 2025 clinical study reported that hip and knee OA prevalence in Germany’s population aged over 60 was about 21.8%, with hip and knee joint OA particularly common among women (23.9%). This elevated prevalence contributes to frequent clinical consultations, increased orthopedic interventions, and a greater need for advanced cartilage‑preserving treatments such as allogeneic chondrocyte therapies. According to the data published by the European Federation of Salaried Doctors (FEMS), 85% of healthcare spending is covered by compulsory (statutory) and government schemes, higher than the EU average of 81%, meaning most services are publicly financed.
The U.K. is poised to be as the fastest-growing region through 2026-2033, due to its high knee osteoarthritis burden and strong clinical focus on regenerative care. The high prevalence of osteoarthritis (OA) in the population, with over 10 million people affected, creates a large patient pool demanding regenerative therapies. Strong research and clinical trial activity in collaboration with the NHS and biotech companies accelerates the development and adoption of allogeneic chondrocyte products. Additionally, the supportive regulatory environment and increasing healthcare investment further fuel growth and early market adoption. According to the NICE Clinical Knowledge Summary on osteoarthritis (2026), an estimated 5.4 million people in the U.K. are affected by knee osteoarthritis a key driver of clinical demand for cartilage repair therapies such as allogeneic chondrocyte approaches. Knee OA is the most common site of symptomatic OA in the U.K., occurring in roughly 1 in 10 adults, which supports faster adoption of innovative treatments. This substantial patient population and emphasis on research propel the U.K.’s growth in advanced chondrocyte applications.
Some of the major key players in Europe Allogenic Human Chondrocyte Market are Kolon TissueGene, Inc., and ISTO Technologies Inc.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 3,122.1 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 10.4% | 2033 Value Projection: | USD 6,161.9 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Kolon TissueGene, Inc., and ISTO Technologies Inc. |
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Nikhilesh Ravindra Patel is a Senior Consultant with over 8 years of consulting experience. He excels in market estimations, market insights, and identifying trends and opportunities. His deep understanding of the market dynamics and ability to pinpoint growth areas make him an invaluable asset in guiding clients toward informed business decisions. He plays a instrumental role in providing market intelligence, business intelligence, and competitive intelligence services through the reports.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients