The Flumazenil Market is estimated to be valued at USD 21.7 Mn in 2026 and is expected to reach USD 28.8 Mn by 2033, exhibiting a compound annual growth rate (CAGR) of 6.2% from 2026 to 2033.
Flumazenil drives the market as a benzodiazepine antagonist that actively reverses sedation and treats overdose cases. Hospitals, emergency departments, and anesthesia recovery units increasingly demand it due to rising benzodiazepine use and growing emphasis on patient safety. Expanding surgical procedures, diagnostic interventions, and critical care services further boost its adoption. Advances in drug delivery technology and integration into clinical guidelines improve its accessibility, positioning Flumazenil as a vital agent in modern healthcare systems worldwide.
|
Current Events |
Description and its impact |
|
Regulatory and Policy Developments in Key Regions |
|
|
Advances in Pharmaceutical Technology and Drug Delivery |
|
|
Supply Chain and Manufacturing Factors |
|
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
5ml hold the largest market share of 52.2% in 2026. Healthcare providers drive demand for 5 ml Flumazenil formulations due to their versatility and ease of use in clinical settings. Hospitals and emergency units use this format to rapidly reverse sedation and manage overdoses, benefiting from precise dosing for both adults and children. Rising numbers of surgeries, endoscopies, and anesthesia procedures increase the need for ready-to-use vials. Its enhanced safety, straightforward administration, and alignment with emergency protocols make the 5 ml Flumazenil vial a preferred choice, sustaining consistent market adoption.
Anti-sedation expected to hold the largest market share of 41.7% in 2026. Healthcare providers drive the Flumazenil market through its anti-sedation applications, using it to rapidly reverse benzodiazepine-induced sedation. Hospitals, surgical centers, and emergency units actively administer it to enhance patient safety, accelerate recovery, and manage sedation-related complications. Rising procedural volumes in anesthesia, endoscopy, and outpatient interventions increase its demand. By implementing improved clinical protocols, training staff, and utilizing efficient delivery formats, medical teams ensure timely administration, establishing Flumazenil as a vital anti-sedation agent that enables safer and more efficient patient care across healthcare settings. For instance, Chinese researchers found that blocking the GABAA receptor protects intestinal stem cells from chemotherapy and radiotherapy toxicity. Their September 20 study in the Journal of Experimental Medicine suggests that the FDA-approved sedative antagonist flumazenil may reduce treatment-related gastrointestinal side effects like diarrhea and vomiting.
Hospital Pharmacies acquired the prominent market share of 38.8% in 2026. Hospital pharmacies drive the Flumazenil market by actively ensuring its availability for emergency and critical care needs. They manage inventory to provide rapid sedation reversal, overdose treatment, and anesthesia recovery across departments. Rising procedural volumes, strict adherence to clinical guidelines, and a strong focus on patient safety increase their demand for the drug. By using streamlined procurement systems, bulk purchasing, and integrating Flumazenil into hospital protocols, pharmacies maintain a consistent supply, establishing it as a vital medication for timely and effective patient care.
To learn more about this report, Request Free Sample
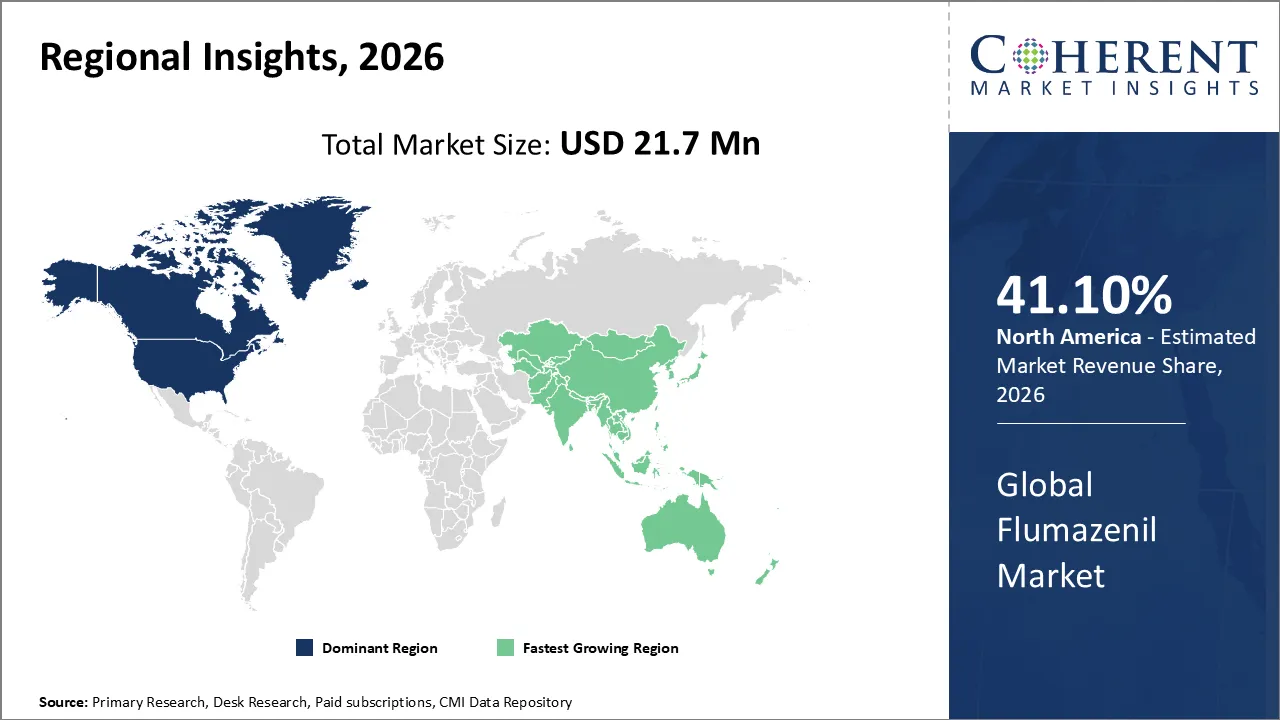
North America dominates the overall market with an estimated share of 41.10% in 2026. Healthcare providers in North America drive the Flumazenil market by increasingly using it for rapid sedation reversal and overdose management in hospitals and emergency care settings. Rising benzodiazepine use and higher volumes of surgical and outpatient procedures boost its adoption. Hospitals and clinics actively integrate Flumazenil into clinical protocols and emergency preparedness plans to prioritize patient safety. Advanced healthcare infrastructure, efficient distribution systems, and continuous staff training further promote its use, establishing Flumazenil as a vital agent for managing sedation and improving recovery across the region. For instance, Acacia Pharma Group plc launched BYFAVO (remimazolam) in the United States, making it available for order and delivery through major wholesalers and specialty distributors. The drug supports patient care during procedures such as surgery and chemotherapy.
Hospitals and emergency care centers in the Asia Pacific actively drive the Flumazenil market by using it for rapid sedation reversal and overdose management. Increasing focus on patient safety and rising numbers of surgeries, endoscopies, and anesthesia procedures boost its demand. Healthcare facilities integrate Flumazenil into clinical protocols while training staff and strengthening emergency preparedness. Expanding hospital infrastructure, improved distribution networks, and growing benzodiazepine use further enhance its adoption, establishing Flumazenil as a vital agent for effective sedation management and safer patient outcomes throughout the region.
Healthcare providers and hospitals across the United States actively propel the Flumazenil market by administering it to rapidly reverse benzodiazepine-induced sedation and treat overdoses. Rising numbers of surgeries, outpatient procedures, and anesthesia recoveries drive its increased use. Medical facilities incorporate Flumazenil into clinical protocols and emergency preparedness strategies to prioritize patient safety. Robust hospital infrastructure, efficient distribution systems, and continuous staff training further enhance its adoption, establishing Flumazenil as a vital agent for effective sedation management and accelerated patient recovery throughout U.S. healthcare settings.
Hospitals and emergency care providers in China actively drive the Flumazenil market by administering it to reverse benzodiazepine-induced sedation and manage overdose cases efficiently. Rising numbers of surgeries, endoscopies, and anesthesia procedures increase demand for the drug. Healthcare facilities incorporate Flumazenil into clinical protocols while enhancing staff training and emergency preparedness. Expanded hospital infrastructure, improved distribution networks, and greater focus on patient safety further promote its use, establishing Flumazenil as a vital agent for effective sedation management and safer outcomes throughout China’s healthcare system.
Healthcare providers increasingly rely on Flumazenil to counteract benzodiazepine-induced sedation in surgeries, outpatient procedures, and emergency care. The need for rapid recovery and reduced patient monitoring drives hospitals and anesthesia centers to prioritize its availability. Continuous expansion of procedural volumes, coupled with a focus on patient safety and improved clinical outcomes, ensures that Flumazenil remains a preferred agent for reversing sedation efficiently across diverse healthcare settings.
Flumazenil’s use in overdose management and critical care has strengthened its role in emergency departments. Hospitals and clinics incorporate it into standardized emergency protocols to improve response time and patient outcomes. Rising awareness of benzodiazepine-related emergencies and growing investment in emergency preparedness programs encourage widespread adoption, positioning Flumazenil as a vital component in hospital formulary lists and acute care treatment pathways.
Rising numbers of surgeries, endoscopies, and anesthesia-dependent outpatient procedures create significant opportunity for Flumazenil. Clinics and ambulatory centers require rapid sedation reversal to optimize patient throughput and recovery times. By offering easy-to-use formats, pre-filled vials, and precise dosing options, Flumazenil can expand its presence beyond hospitals into outpatient facilities, positioning itself as a preferred agent for safe and efficient sedation management across a variety of clinical environments.
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 21.7 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 6.2% | 2033 Value Projection: | USD 28.8 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Hoffmann La Roche, Akorn-Strides, LLC, Sandoz Canada Incorporated, Fresenius Kabi USA, LLC, Mylan Pharmaceuticals, Genentech, Inc., General Injectables & Vaccines Inc, Sina Health Inc, West Ward Pharmaceutical, HF Acquisition Co. LLC, DBA HealthFirst, Hikma Farmaceutica, Pfizer Laboratories Div Pfizer Inc., Physicians Total Care, Inc., and Bedford Pharmaceuticals. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients