Global genital warts treatment market is estimated to be valued at USD 2.12 Bn in 2026 and is expected to reach USD 3.11 Bn by 2033, exhibiting a compound annual growth rate (CAGR) of 5.6% from 2026 to 2033.
Discover market dynamics shaping the industry: Request Free Sample
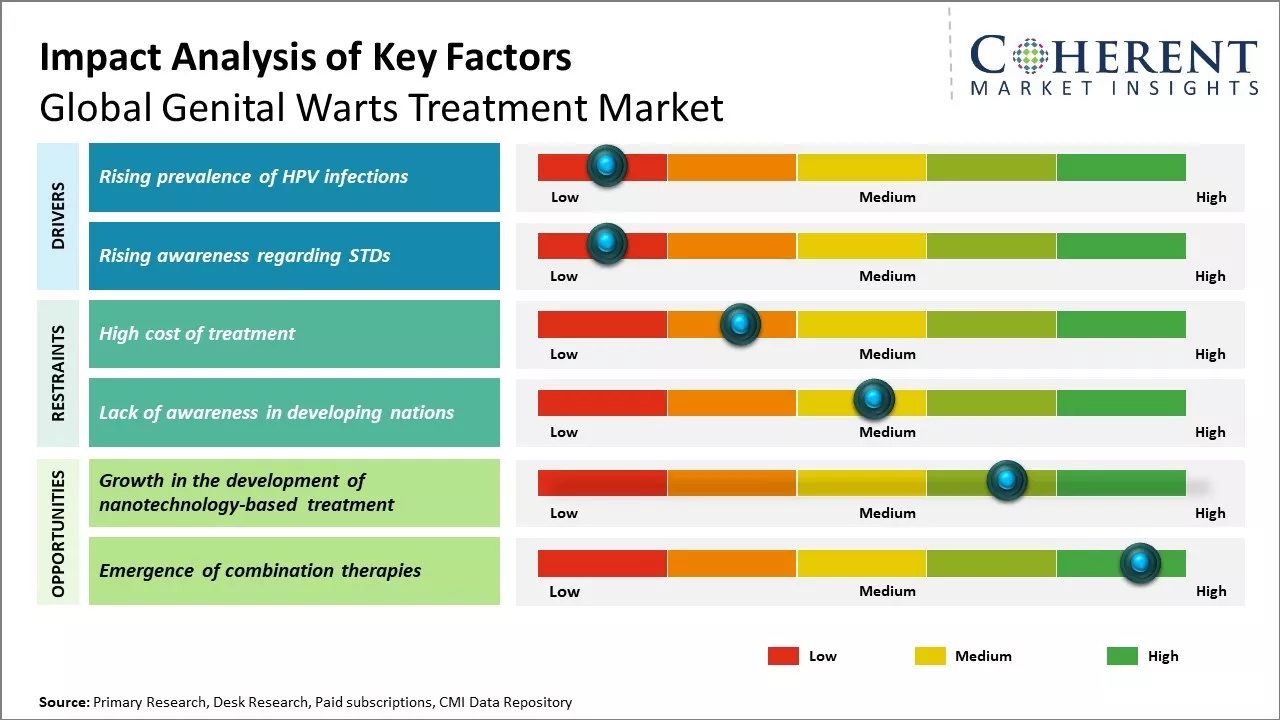
Increasing awareness among people about sexually transmitted diseases and availability of effective drugs and medicines for treatment of genital warts can drive the market growth. Developing healthcare infrastructure in emerging nations can also boost demand for genital warts treatment. However, social stigma associated with the condition and preference of patients towards non-drug therapies can hamper the market growth. Increasing awareness campaigns by governments and non-profit organizations as well as development of novel treatment therapies can offer new opportunities for the players in the global genital warts treatment market during the forecast period.
Rising prevalence of HPV infections
Increasing prevalence of HPV infections across both developed and developing regions can drive the genital warts treatment market growth. HPV, or human papillomavirus, is the most common STI globally and a leading cause of genital warts. Several strains of HPV are known to cause genital warts, with types 6 and 11 being the predominant strains associated with wart development. As per medical experts, HPV infection rates have surged in recent decades due to changing sexual behaviors such as early initiation of sexual activity, multiple sexual partners, and lack of protective barriers. Moreover, diagnosis of HPV infection has become more efficient with new diagnostic tools broadening the potential patient pool. At least 80% of sexually active individuals can acquire HPV infection at some point in their lives. Individuals with compromised immunity are at higher risk of getting infected. While many HPV infections resolve on their own, a substantial fraction of infected individuals develop visible genital warts that require medical intervention. Since there is no cure for HPV itself, the long-term treatment of recurring symptoms can boost repeat usage of wart remover therapies and surgical procedures. Significant monetary investments are also being made towards HPV vaccination but it will be many years before their effect in reducing transmission becomes evident.
Get actionable strategies to beat competition: Request Free Sample
Rising awareness regarding STDs
Rising awareness about sexual health and sexually transmitted diseases (STDs) can drive the global genital warts treatment market growth. With more education and accessibility of information, people are now more knowledgeable about STDs like human papillomavirus (HPV) that can cause genital warts. Various health organizations and government bodies have launched awareness campaigns to promote safe sex practices and highlight the importance of early detection and treatment of HPV and associated conditions. For instance, the World Health Organization (WHO) partnered with National Health Ministries across regions to strengthen sexual health services and counselling. As per WHO in 2022, as part of its 'Reaching Out' program launched in 2020, WHO aims to build the capacity of primary health workers to provide counselling for STDs, offer testing facilities and ensure access to affordable treatment options. Such initiatives have raised awareness about HPV as one of the most common STDs worldwide and genital warts as a visible symptom. People diagnosed with genital warts are likely to opt for recommended treatments to get relief from symptoms and reduce the risk of HPV transmission. Growing medicalization of sexual wellness and an expanding patient pool diagnosed with genital warts have encouraged pharmaceutical companies to intensify research and development of advanced treatment solutions. Market leaders are involving clinical studies to evaluate the effectiveness of newer therapeutics like Imiquimod for genital warts.
Key Takeaways from Analyst:
Global genital warts treatment market growth is driven by rising prevalence of human papillomavirus (HPV) infections worldwide. Growing awareness campaigns regarding safe sex practices and HPV vaccination boosts demand for genital warts treatment drugs and therapies. However, social stigma associated with discussing sexually transmitted diseases can hamper the market growth.
North America currently dominates the genital warts treatment market, owing to advanced healthcare infrastructure and higher awareness levels. However, Asia Pacific is expected to witness the fastest growth over the forecast period due to improved access to healthcare in developing countries of the region.
Pharmaceutical companies are investing more in research and development of novel non-invasive therapies to address the unmet needs of patients. This offers new opportunities for technology advancements in the field of genital warts treatment. Market players are also introducing combination therapies that shorten treatment duration and provide better cure rates.
Increased generic competition after major drugs lose patent protection can pose challenges to prominent market players. However, sustained expansion of healthcare coverage in emerging markets is a notable growth-inducing factor.
Market Challenges: High cost of treatment
One of the major factors hampering the growth of global genital warts treatment market is high cost of treatment. Genital warts, also known as human papillomavirus (HPV) infection, requires prolonged treatment using prescription medications, cryotherapy, or surgery in severe cases. All of these treatment options are quite expensive for a common person. Prescription medications for genital warts include topical creams containing drugs like podofilox or imiquimod. A single tube of these creams which lasts for 4-6 weeks costs US$ 50 to US$ 150 without insurance in the U.S. Cryotherapy which involves freezing off the warts using liquid nitrogen is also an extensively used method. However, multiple sittings are required which increases the overall cost. Even a single sitting of cryotherapy performed by a dermatologist cost over US$ 100 in developed countries. Furthermore, for symptomatic genital warts which are large, raised or resistant to topical treatments, surgeries like electrocautery or laser ablation are required. But the average cost of a single session of surgical removal ranges from US$ 300 to US$ 500. This makes surgery an impractical option for most patients given the high out-of-pocket expenditure needs to be paid. The high cost of treatment leads to decreased adherence to the prescribed line of treatment. In 2018, according to the data from International Agency for Research on Cancer, nearly 20% of patients in developing countries of Africa and South-East Asia discontinued the medication halfway mostly due to financial constraints. This leads to persistence of the disease which further transmits among population. It also negatively impacts the patient outcomes and overall disease management efforts.
Market Opportunities: Growth in the development of nanotechnology-based treatment
The development of nanotechnology-based treatment offers immense potential for global genital warts treatment market growth. Nanotechnology allows development of novel drug delivery systems that can efficiently target affected areas and cells. Nanoparticles can be designed to specifically recognize genital warts and deliver antiviral drugs or immunotherapy directly at the sites of infection. This targeted treatment ensures optimal drug concentrations are achieved locally while minimizing systemic exposure and side effects. Some promising nanotechnology platforms being researched include Lipid-based nanoparticles, Inorganic nanoparticles and Dendrimers. Lipid nanoparticles encapsulating siRNA or small molecule drugs have shown ability to penetrate warts and silence viral gene expression. Gold and silica nanoparticles conjugated with genetic materials or immune stimulants are under investigation. Dendrimers synthesized from branched monomers can entrap antiviral compounds internally and slowly release them. Working on these advanced mechanisms of delivery hold potential to achieve higher treatment efficacy and response rates compared to conventional topical medications. As per WHO, over 30 nanomedicine products and vaccines are in different trial phases for HPV related diseases globally. Researchers from Massachusetts Institute of Technology reported progress in an animal study published in February 2022 assessing an anti-HPV nanovaccine based on self-assembling polymers. If human trials prove successful, it may radically transform genital warts management. With persistent efforts, first generation of nanotechnology drugs for HPV are anticipated to be available in late 2020s or early 2030s. Their widespread adoption can drive the market growth through more effective clinical outcomes and patient acceptability.
Discover high revenue pocket segments and roadmap to it: Request Free Sample
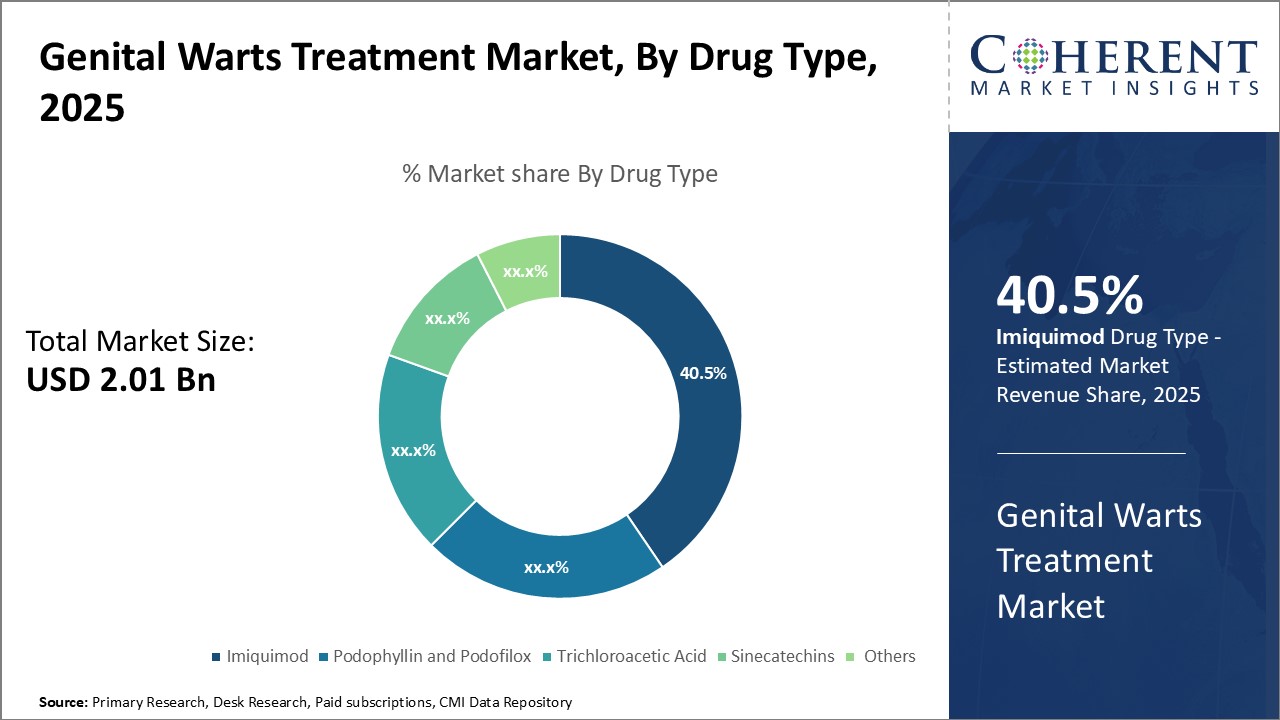
By Drug Type- Imiquimod's efficacy and safety drives the segment growth
In terms of drug type, imiquimod segment is estimated to contribute the highest market share of 41.5% in 2026, owing to its superior efficacy and safety profile as compared to other treatment options. As an immune response modifier, Imiquimod works by activating immune cells in the skin and body to fight the human papilloma virus (HPV) that causes genital warts. This targeted mode of action makes it highly effective at clearing warts while posing minimal risk of side effects. Imiquimod is also easy to apply as a topical cream, allowing for convenient at-home treatment without invasive procedures or lifestyle interruptions. Its favorable safety and usability characteristics have made Imiquimod the preferred first-line therapy recommended by doctors. Continuous innovation to further strengthen Imiquimod formulations, such as newer delivery technologies, can drive the segment growth.
To learn more about this report, Request Free Sample
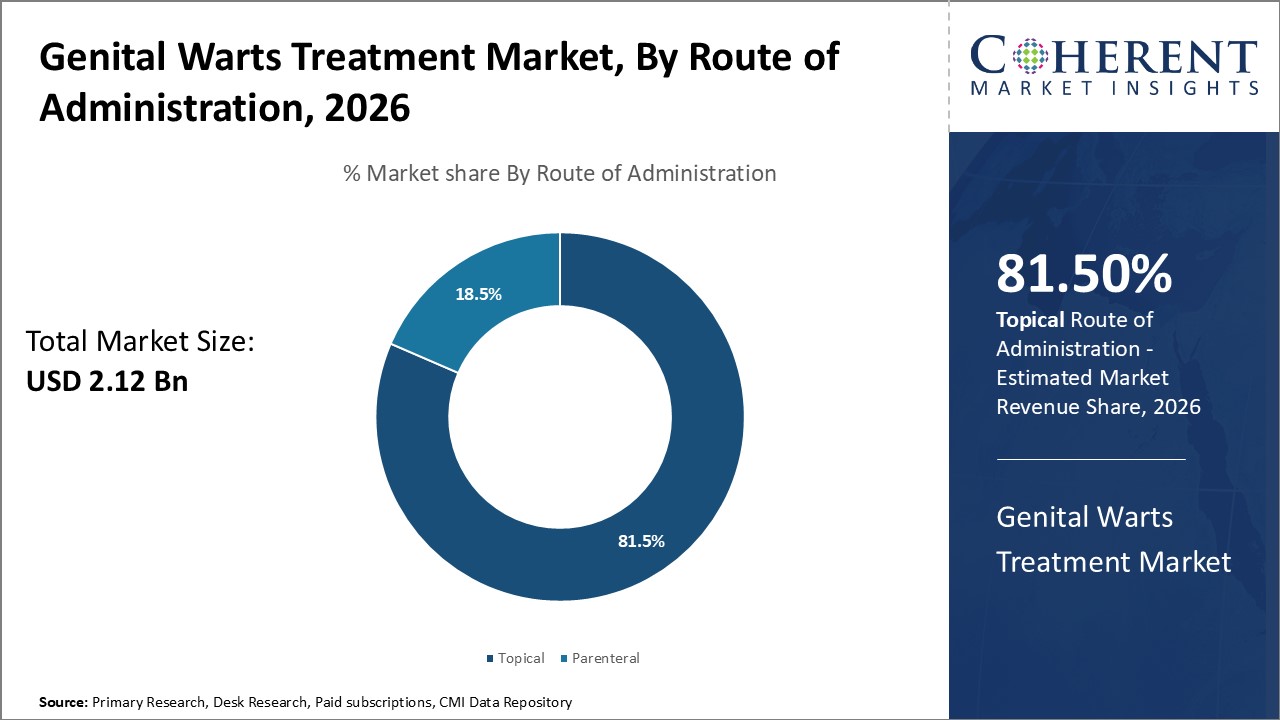
By Route of Administration - Topical administration dominates due to non-invasive delivery and enhanced comfort
In terms of route of administration, topical segment is estimated to contribute the highest market share of 81.5% in 2026, due to the significant advantages it offers over parenteral alternatives. Being applied externally on the skin, topical treatments does not require insertion of needles or catheterization like injections. This non-invasive delivery method assuages patients' pain and fear of procedures, improving compliance. It also avoids potential side effects associated with systemic absorption of drugs into the bloodstream. Topical medications tend to have localized effects, targeting genital tissues more precisely without subjecting the entire body to medication. The convenience and comfort of rubbing or spreading creams also fits well with the sensitive nature of genital infections. These attributes have made topical the ideal choice for most genital wart patients.
By Distribution Channel- Retail pharmacies are most accessible point of care for genital wart patients
In terms of distribution channel, retail pharmacies segment is estimated to contribute the highest market share of 31.5% in 2026, owing to their widespread accessibility and availability. As genital warts often require discreet treatment, patients prefer purchasing medications from local pharmacists these are familiar with rather than visiting hospitals. Community pharmacies are also more numerous than hospitals across all urban and rural settings. Their storefront operations allow for quick drop-in purchases without prior appointments. The anonymity and self-service nature of retail pharmacies addresses patients' privacy needs. These also offer delivery services for those unwilling to visit in person. Insurance coverage is more reliably applicable for retail prescriptions versus mail orders. These unparalleled convenience factors have established retail pharmacies as the go-to access point for most genital wart patients.

Need a Different Region or Segment? Request Free Sample
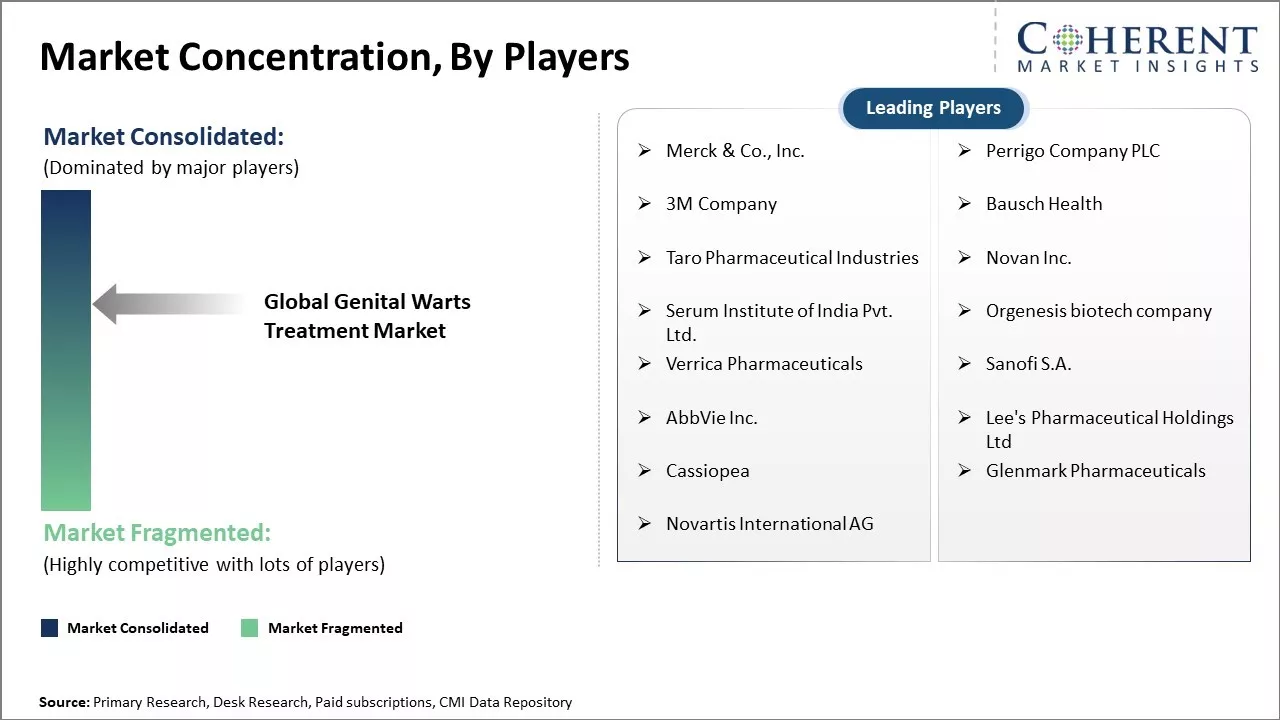
North America dominates the global genital warts treatment market with an estimated market share of 40.7% in 2026, due to strong healthcare infrastructure and high awareness levels about sexually transmitted diseases. The region has a high concentration of leading pharmaceutical companies that are involved in research and development of new drugs and therapies for genital warts. The U.S. is home to many top pharmaceutical players like Merck & Co., Pfizer Inc., and GlaxoSmithKline that have their own genital warts treatment portfolio.
Moreover, North America has witnessed faster adoption of advanced treatment options like cryotherapy due to better access and availability of such therapies. Hospitals and dermatology clinics in the region are well-equipped with state-of-art cryosurgery devices for genital warts removal. This has ensured quick and effective care for patients. However, patent expirations of major drugs may challenge the regional market growth.
Asia Pacific region has emerged as the fastest growing market for genital warts treatment. Factors such as rising incidence of HPV infections due to rapid urbanization and changing lifestyles drives the market growth. Countries like China, India and Japan have experienced significant increase in genital warts cases, thus, boosting demand for treatment solutions.
Genital Warts Treatment Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 2.12 Bn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.6% | 2033 Value Projection: | USD 3.11 Bn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
Merck & Co., Inc., Perrigo Company PLC, 3M Company, Bausch Health, Taro Pharmaceutical Industries, Novan Inc., Serum Institute of India Pvt. Ltd., Orgenesis biotech company, Verrica Pharmaceuticals, Sanofi S.A., AbbVie Inc., Lee's Pharmaceutical Holdings Ltd, Cassiopea, Glenmark Pharmaceuticals, Novartis International AG |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
*Definition: Global genital warts treatment market refers to the collective market for products and therapies used in the treatment of genital warts, a common sexually transmitted infection caused by certain strains of the human papillomavirus (HPV). This market encompasses various treatment options such as topical medications, surgical interventions, and other therapeutic approaches aimed at managing and eliminating genital warts. Factors driving this market include the prevalence of HPV infections, increasing awareness about sexually transmitted diseases, advancements in treatment options, and efforts to reduce the incidence of HPV-related cancers.
Primary Research Interviews:
Databases:
Magazines:
Journals:
Newspapers:
Associations:
Public Domain Sources:
Share
Share
Vipul Patil is a dynamic management consultant with 6 years of dedicated experience in the pharmaceutical industry. Known for his analytical acumen and strategic insight, Vipul has successfully partnered with pharmaceutical companies to enhance operational efficiency, cross broader expansion, and navigate the complexities of distribution in markets with high revenue potential.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients