Hepatic Encephalopathy Market is estimated to be valued at USD 475.5 Mn in 2026 and is expected to reach USD 705.5 Mn in 2033, exhibiting a compound annual growth rate (CAGR) of 5.8% from 2026 to 2033. Increase in product launches by key market players is expected to drive the market growth during the forecast period. Moreover, an increase in prevalence of disease such as jaundice and others is also expected to drive the market growth.
Analysts’ Views on Global Hepatic Encephalopathy Market:
Increasing research and development activities for the treatment of liver diseases is expected to drive the global hepatic encephalopathy market growth over the forecast period. For instance, in March 2020, Bausch Health Companies Inc, is a Canadian multinational specialty pharmaceutical company, announced positive results for the Phase II study for the combination therapy, which included rifaximin in combination with the lactulose. The trial was carried out to study the effectiveness of combination therapy for the treatment of Overt Hepatic Encephalopathy (OHE).
Figure 1. Global Hepatic Encephalopathy Market Share (%), By Drug Class, 2026
To learn more about this report, Request Free Sample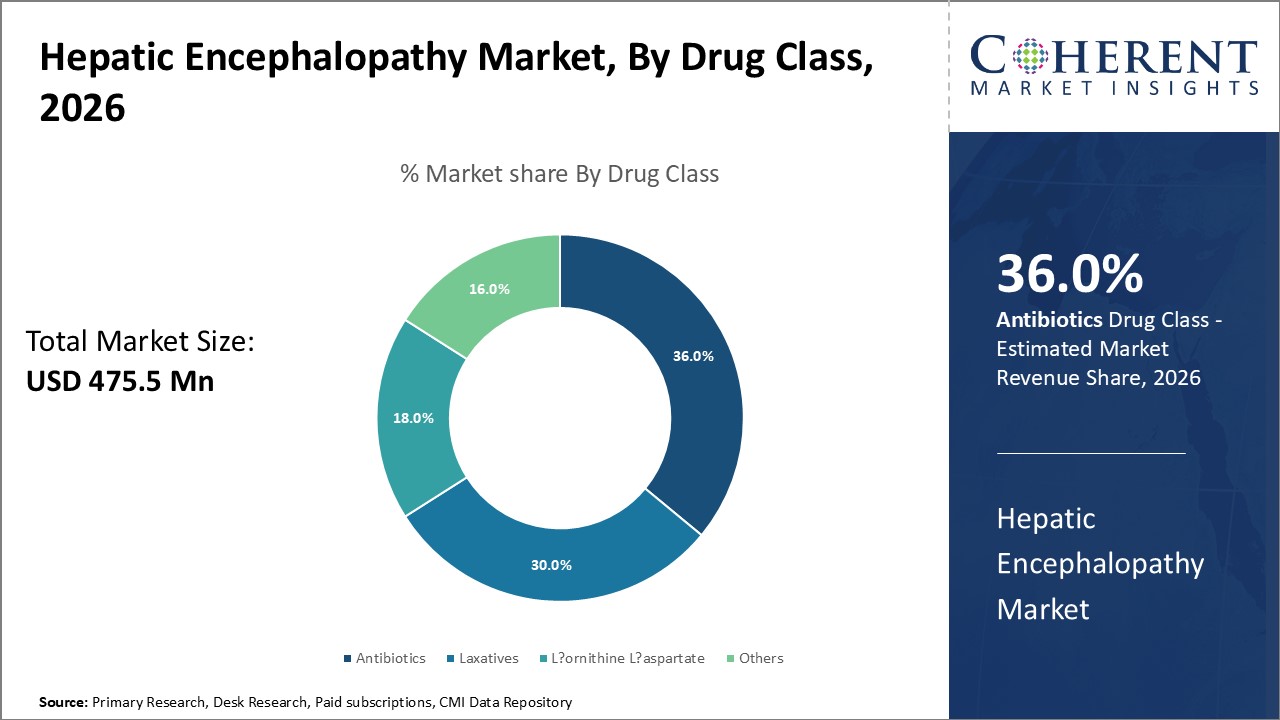
Global Hepatic Encephalopathy Market - Driver
Increasing New Therapies Associated With Liver Disease
An increase in the diagnosis of liver disease is expected to increase the demand for new therapies by key players operating in the market, which in turn is expected to drive the global hepatic encephalopathy market growth during the forecast period. The therapy involve methods besides non-absorbable disaccharides and non-absorbable antibiotics for example polyethylene glycol, l-ornithine l-aspartate, non-ureic nitrogen scavengers and albumin therapy.
Increasing submissions of abbreviated new drug application (ANDA)
Market players are involved in submitting the abbreviated new drug application (ANDA), which is an important step for launching new drugs in the market. For instance, in February 2016, Allergan plc, is an American, Irish-domiciled pharmaceutical company, announced that the U.S. Food and Drug Administration (FDA) accepted its abbreviated new drug application (ANDA) for gaining approval to market generic rifaximin tablets, 550 mg, which is indicated for the treatment of overt hepatic encephalopathy (HE) recurrence in adults.
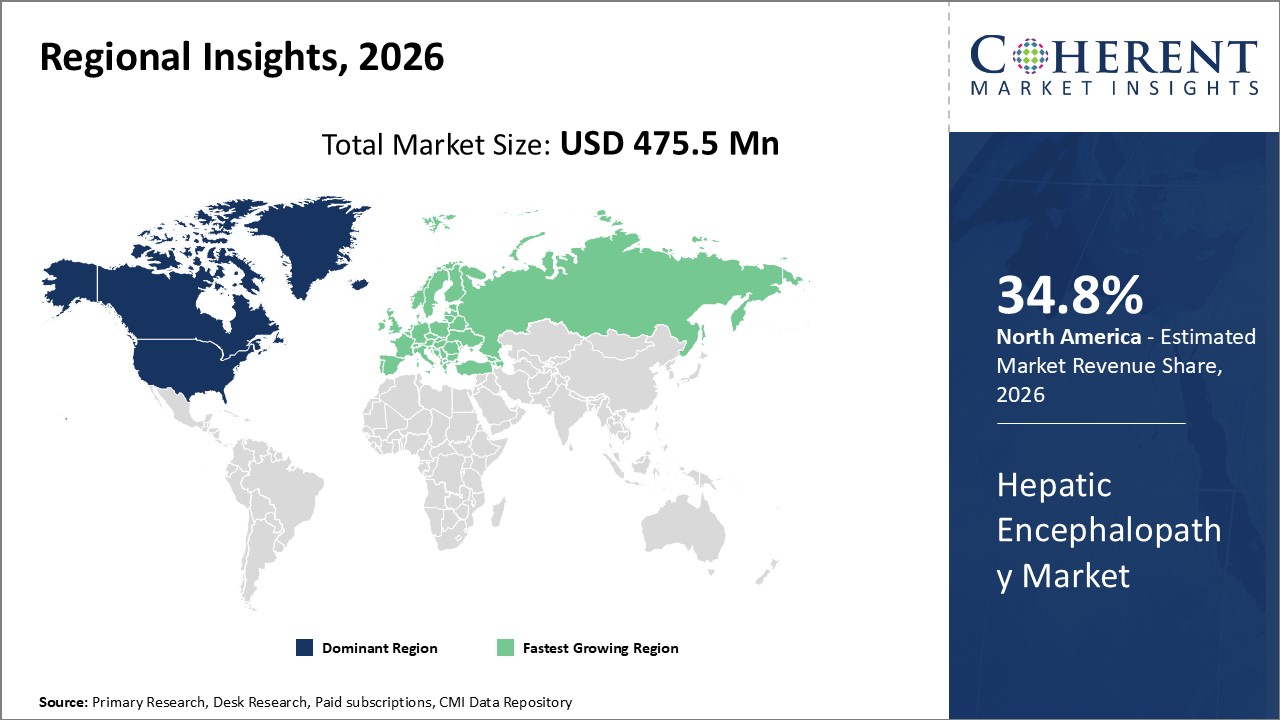
Figure 2. Global Hepatic Encephalopathy Market Share (%), By Region, 2026
To learn more about this report, Request Free Sample
Global Hepatic Encephalopathy Market - Regional Analysis
Among all regions, North America is expected to dominate the global market over the forecast period. This is attributed to North America holding a 34.8% market share and due to presence of major players such as ASKA Pharmaceutical Co., Ltd., Cosmo Pharmaceuticals N.V, Lupin Limited., Kaleido Biosciences., and others are contributing for development of Hepatic Encephalopathy market.
The European region is expected to be second dominating region over the forecast period, due to increase in cirrhosis and liver failure. For instance, September, 2021 according to the National Center of Biotechnology and Information and clinical liver disease (CLD), cirrhosis is a leading cause of mortality and morbidity across the world. It is the 11th leading cause of death and 15th leading cause of morbidity, accounting for 2.2% of deaths and 1.5% of disabilityâ€adjusted life years worldwide in 2016. 1 CLD caused 1.32 million deaths, approximately twoâ€thirds among men and oneâ€third among women.
Global Hepatic Encephalopathy Market
Solutions for managing liver diseases have proven to be highly beneficial in this regard since they enable healthcare providers to easily combine SGLT1 and SGLT2, patient management, and care management solutions with solutions for healthcare payers, such as claim management and payment management systems.
Such factors are contributing to a rise in mortality or some of theleading causes of death in the U.S., such as hepatitis A, B and Hepatitis C and liver failure.
Hepatic Encephalopathy Market Report Coverage
| Report Coverage | Details | ||
|---|---|---|---|
| Base Year: | 2025 | Market Size in 2026: | USD 475.5 Mn |
| Historical Data for: | 2020 To 2024 | Forecast Period: | 2026 To 2033 |
| Forecast Period 2026 to 2033 CAGR: | 5.8% | 2033 Value Projection: | USD 705.5 Mn |
| Geographies covered: |
|
||
| Segments covered: |
|
||
| Companies covered: |
ASKA Pharmaceutical Co., Ltd., Cosmo Pharmaceuticals N.V, Lupin Limited., Kaleido Biosciences, Kannalife Sciences Inc., Bausch Health Companies Inc., Ferring Pharmaceuticals Inc., Mallinckrodt Pharmaceuticals, Umecrine Cognition AB., Norgine B.V., Abbott Laboratories, QR Science and Technology development Co. |
||
| Growth Drivers: |
|
||
| Restraints & Challenges: |
|
||
Uncover macros and micros vetted on 75+ parameters: Get instant access to report
Global Hepatic Encephalopathy Market Segmentation:
The global hepatic encephalopathy market report is segmented into drug class, route of administration, distribution channel, and Region
Based on Drug Class, the market is segmented into hepatic encephalopathy, antibiotics, laxatives, l-ornithine l-aspartate and others. Out of which, the antibiotics is segment is expected to dominate the global hepatic encephalopathy market during the forecast period owing to the increasing product launches and approvals by the major market players.
Based on Route of Administration, the market is segmented into oral, intravenous, rectal and others. Out of which, the intravenous, segment is expected to dominate the market over the forecast period. Increase in product launches by key market players is likely drive the growth of segment. For instance, in August, 2026, the Food and Drug Administration (FDA) approved injectable HepatAmine (9% Amino Acid Injection) to treat the symptoms of hepatic encephalopathy in patients with Cirrhosis or Hepatitis.
Based on Distribution Channel, the market is segmented into hospital pharmacies, retail pharmacies and online pharmacies. Out of which, the hospital pharmacies is expected to dominate the market over the forecast period. For instance, increasing prevalence of Hepatitis A, B and C and liver Cirrhosis leads to increase in patient visits in hospitals and thereby drive the growth of segment
Based on Region, the market is segmented into North America, Latin America, Europe, Asia pacific, Middle East and Africa out of the North America segment is dominated market over the forecast period. Market players operating in the region are focused on various strategies such as product launches and agreement with other key market players, which is expected to drive the market growth in this region
Global Hepatic Encephalopathy Market Cross Sectional Analysis:
The global hepatic encephalopathy market is growing in Asia Pacific region, owing to the increasing incidence of liver diseases such as Hepatitis A, B and C, liver failure and others. This represents more product approvals for stimulators and inhibitors across the Asia Pacific region. For instance, on March 3, 2023, according to pharmaceutical technology drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data
The characteristics of the medicine, the company, and its clinical trials are crucial to the PTSR and chance of approval for that drug.
Global Hepatic Encephalopathy Market: Key Developments
In September, 2022, GENFIT, a late-stage biopharmaceutical company dedicated to improving the lives of patients with liver diseases, announced that they have acquired Clinical-stage Biopharmaceutical company, Versantis, with VS-01-ACLF, a Phase 2 ready program based on first-in-class scavenging liposomes technology, VS-01-UCD, a pediatric program focused on urea cycle disorder (UCD), and VS-02-HE, an early-stage program focused on hepatic encephalopathy (HE).
On April 7, 2023, Merck, a German multinational science and technology company, and Eisai, a Japanese pharmaceutical company, released information regarding Phase 3 Trials for the treatment of Advanced Melanoma and Metastatic Colorectal Cancer using the orally available multiple receptor tyrosine kinase inhibitors of KEYTRUDA (pembrolizumab) and LENVIMA (lenvatinib) respectively.
On March 3, 2023, Bausch Health Companies Inc. is a Canadian multinational specialty pharmaceutical company, has announced Rifaximin SSD (soluble solid dispersion) tablets plus lactulose is under the clinical trial phase III for treatment of overt hepatic encephalopathy.
Global Hepatic Encephalopathy Market: Restraint
High cost associated the treatment of hepatic encephalopathy
High cost of the treatment of overt hepatic encephalopathy (OHE) is one of the factor that is restrain the growth of the market in forecast period. For instance, according to the National Center for Biotechnology Information, in 2022, hepatic encephalopathy is the second most common reason for hospitalization in patients suffering from liver cirrhosis, in the U.S., after ascites. According to the same source, the national expenditures for the U.S., related to hospitalizations for hepatic encephalopathy have been estimated to range from around US$ 1 billion per year to above US$ 7 billion per year.
Global Hepatic Encephalopathy Market: Key Players
Major players operating in the global hepatic encephalopathy market include ASKA Pharmaceutical Co., Ltd., Cosmo Pharmaceuticals N.V, Lupin Limited., Kaleido Biosciences, Kannalife Sciences Inc., Bausch Health Companies Inc., Ferring Pharmaceuticals Inc., Mallinckrodt Pharmaceuticals, Umecrine Cognition AB., Norgine B.V., Abbott Laboratories, QR Science and Technology development Co.
*Definition: Hepatic encephalopathy (HE) is a reversible syndrome observed in patients with advanced liver dysfunction. This syndrome is characterized by a wide spectrum of neuropsychiatric abnormalities resulting from the accumulation of neurotoxic substances in the brain.
Share
Share
Ghanshyam Shrivastava - With over 20 years of experience in the management consulting and research, Ghanshyam Shrivastava serves as a Principal Consultant, bringing extensive expertise in biologics and biosimilars. His primary expertise lies in areas such as market entry and expansion strategy, competitive intelligence, and strategic transformation across diversified portfolio of various drugs used for different therapeutic category and APIs. He excels at identifying key challenges faced by clients and providing robust solutions to enhance their strategic decision-making capabilities. His comprehensive understanding of the market ensures valuable contributions to research reports and business decisions.
Ghanshyam is a sought-after speaker at industry conferences and contributes to various publications on pharma industry.
Joining thousands of companies around the world committed to making the Excellent Business Solutions.
View All Our Clients